Documenti di Didattica
Documenti di Professioni
Documenti di Cultura
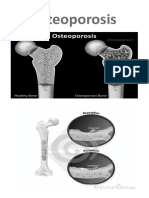
Chronic Glucocorticoid Therapy-Induced Osteoporosis in Patients With Persistent Asthma and Chronic Obstructive Pulmonary Disease (CPOD)
Caricato da
zulfantri19830 valutazioniIl 0% ha trovato utile questo documento (0 voti)
25 visualizzazioni28 pagineThis document discusses chronic glucocorticoid therapy-induced osteoporosis in patients with persistent asthma and chronic obstructive pulmonary disease. It covers risk factors for glucocorticoid-induced bone loss including daily dose, duration, and total lifetime dose. It also discusses molecular mechanisms, cellular effects, importance of glucocorticoid-induced osteoporosis, diagnosis using biomarkers and DEXA scans, and treatment recommendations including calcium, vitamin D, bisphosphonates, and lifestyle modifications.
Descrizione originale:
ppok kosrtikosteroid
Titolo originale
Copd Asma Kortikost
Copyright
© © All Rights Reserved
Formati disponibili
PPT, PDF, TXT o leggi online da Scribd
Condividi questo documento
Condividi o incorpora il documento
Hai trovato utile questo documento?
Questo contenuto è inappropriato?
Segnala questo documentoThis document discusses chronic glucocorticoid therapy-induced osteoporosis in patients with persistent asthma and chronic obstructive pulmonary disease. It covers risk factors for glucocorticoid-induced bone loss including daily dose, duration, and total lifetime dose. It also discusses molecular mechanisms, cellular effects, importance of glucocorticoid-induced osteoporosis, diagnosis using biomarkers and DEXA scans, and treatment recommendations including calcium, vitamin D, bisphosphonates, and lifestyle modifications.
Copyright:
© All Rights Reserved
Formati disponibili
Scarica in formato PPT, PDF, TXT o leggi online su Scribd
0 valutazioniIl 0% ha trovato utile questo documento (0 voti)
25 visualizzazioni28 pagineChronic Glucocorticoid Therapy-Induced Osteoporosis in Patients With Persistent Asthma and Chronic Obstructive Pulmonary Disease (CPOD)
Caricato da
zulfantri1983This document discusses chronic glucocorticoid therapy-induced osteoporosis in patients with persistent asthma and chronic obstructive pulmonary disease. It covers risk factors for glucocorticoid-induced bone loss including daily dose, duration, and total lifetime dose. It also discusses molecular mechanisms, cellular effects, importance of glucocorticoid-induced osteoporosis, diagnosis using biomarkers and DEXA scans, and treatment recommendations including calcium, vitamin D, bisphosphonates, and lifestyle modifications.
Copyright:
© All Rights Reserved
Formati disponibili
Scarica in formato PPT, PDF, TXT o leggi online su Scribd
Sei sulla pagina 1di 28
Chronic Glucocorticoid Therapy-Induced
Osteoporosis in Patients with Persistent
Asthma and Chronic Obstructive Pulmonary
Disease (CPOD)
Edu Tehupeiory
SUB-BAGIAN REUMATOLOGI BAGIAN ILMU PENYAKIT
DALAM FAKULTAS KEDOKTERAN UNIVERSITAS
HASANUDDIN
MAKASSAR
INTRODUCTION
• Long-term glucocorticoid (GC) therapy has been
decreasing morbidity and mortality in of chronic
inflammatory disease.
• Persistent asthma and COPD.
• The important and often unrecognized side
effects is secondary osteoporosis.
Risk Factors
• The risk of GC – Induced bone loss is correlated
with : daily dose, duration and total cummulative
lifetime dose of GC treatment.
• Oral prednisone increase the risk of bone loss
and fracture.
• High doses of inhaled GCs may also increased
the risk of Osteopenia / Osteoporosis.
CORTICOSTEROID (CS)
1. Moleculer Biology of CS Action
- CS exert their effects, anti-inflammatory and
immunosuppressive primarily through
interaction with ubiquitously receptors.
- In the circulation : free form and association
with cortisol-binding globulin.
- Free form diffuses through plasma
membrane bind the cytoplasmic CS
receptor.
- The formation of steroid receptor complex
followed by translocation of the complex to
the nucleus.
- Corticosteroids regulate gene expression by :
transcriptional, post transcriptional, and post-
translational mechanism.
2. Cellular Effects of CS
- CS influence the function of variety of organs
and their component cells.
- Effects of CS on particular cell type depends
on concentration and type of CS
(synthetic / naturally occuring and the receptor
subtype to which steroid binds).
• The outcome varies with the phase in the cell
cycle and stage of differentiation or activation of
the cells.
GC - Dose
• In patients treated with oral GC more than
1 year, 86 % demonstrated a decrease in bone
mineral (dose not stated).
• Daily dose :
Low - dose : 33 % decrease BMD
Medium - dose : 71 % decrease BMD
High - dose : 80 % decrease BMD
Clinical Importance of
GC induced OP
• Most common form of drug – related OP in men
and women.
• Occurs at any age, in both gender, across races.
• Up to 50 % of patients on chronic steroid therapy
sustain OP fractures and/or develop
osteonecrosis.
PATHO – PHYSIOLOGY of
GC Induced OP
• Corticosteroids induce OP primarily through
effects on bone synthesis or formation.
• Bone remodeling is regulated via the interaction
of systemic hormones and local cytokine.
• An inbalance between osteoblast activity.
• Metabolic abnormalities contribute increased
bone absorption.
*) Secondary hyperparathyroidism.
*) Decrease adrenal production of androgene.
• The severity of these side effects parallels other
effects.
• Incidence is uncertain, approx 30 – 90 % and
the severity of OP is closely related to dose and
duration of GC therapy.
• Bone loss is greatest during the first 6 to 12
months of therapy and most marked in
trabecular bone.
• Additional factors that accelerate the risk
of fracture
- Smoking, alcohol intake
- Sedentary lifestyle, diet low calcium and
vit – D
- Menopause
• Fundamentaly GC are catabolic hormones,
acting through the GC receptor.
• Reduce the transcription of mRNAs for the
various constituent of bone matrix such as type
1 collagen
• Corticostiroid have important effects on bone
through indirect suppression of gonadal and
adrenal androgen production and secretion.
• Inhibit intestinal calcium absorption promote
renal calcium excretion.
• This net calcium loss stimulates PTH secretion
secondary hyperparath, which additional
contributes to bone loss.
• Plasma levels of PTH increased in most patients
taking high dose of GC.
• The major effect of corticosteroids is the
INHIBITION OF BONE FORMATION
DIAGNOSIS
• Early detection of bone loss prior to clinical
manifestafiosis is paramount.
• Conventional radiology not sensitive to detect
early bone loss.
• Changes less than 30% in bone density, cannot
be detected by conventional radiographic
imaging.
• Biochemical markers.
- Bone formation :
(osteocolein, alkalin phosphatase, bone
APh, procollagen type I carboxy-terminal
propeptide (PICP))
- Bone resorption :
(hydroxyproline, pyridoline, deoxypyridinoline)
• Biochemical markers in monitoring response to
therapy.
• Dual Energy X-ray Absorptiometry (DXA) scans
of the lumbar spine, hip, or wrist provide reliable
assessment of BMD and predict the risk of future
fracture.
• DEXA “Gold Standard” for diagnosis
osteoporosis.
THERAPIES FOR THE TREATMENT OF
GC-INDUCED OSTEOPOROSIS.
• Attemps to reduce the risk of bone loss :
- Use the lowest effective oral/inhaled GC dose.
- Optimal use of alternative non-GC anti-inflam
matory drugs.
• Modification Lifestyle :
- Smoking cessation.
- Reduced/stop alcohol intake.
- Prevention of falls.
Treatment Recommendation
1. - For the prevention of GC-Induced bone loss in
adult patient beginning or currently recieving
long-term GC (oral and/or inhaled) assessment
of lumbar spine or femoral (neck BMI).
- Standard lateral thoracic and lumbar spine
radiographs should be obtained.
- If no fracture and BMD is normal : adequate
calcium and vitamin-D (800 IU/d)
- These supplements impede the proggression
to osteopenia.
- After one month of supplementation, 24-h urine
calcium should be assessed Hypercalciuria.
- If present, hypercalciuria require treatment with
calcium-sparing diuretic (e.g : indopamide 1.25
mg/d trichlormethiazide 2.0 mg/d)
2. Patients with long-term GC documented bone
loss and/or osteoporotic fracture :
- Supplement calcium & vitamin-D.
- HRT (Not approved in USA).
- Bisphosphonate therapy.
Risedronate in the Prevention of CIO:
Conclusions
In patients initiating oral corticosteroids,
risedronate prevents bone loss with a trend in
reducing vertebral fractures
Risedronate protects against loss of both
cortical and trabecular bone
Risedronate was well-tolerated and exhibited
a favorable bone safety profile
Cohen at al, Arthritis and Rheumatism, 1999
3. - After initiation of treatment BMD assessment
every 12 month.
- If BMD remains stable (increasing relative to
baseline or decreasing by less than 5 %), no
therapeutic changes .
- If BMD declines by > 5 %, pharmacologic
changes.
- Consultation with a bone metabolism specialist.
to therapy.
SUMMARY
Reducing prevalance of GC-Induced bone loss in
patients with asthma and COPD, require the
following.
1. The appropriate use of NSAID to limit
exposuren to GC.
2. Understanding of the adverse effects of
chronic oral and inhaled GC therapy.
3. Early identification of asthma and COPD
patients
4. Utilization of agents attenuate bone resorption
and promote bone formation.
5. Ability to quickly, precisely, and inexpensively
assess improvements in BMD in respons to
therapy.
Potrebbero piacerti anche
- Diagnosis and Treatment Planning in ImplantsDocumento26 pagineDiagnosis and Treatment Planning in ImplantsGayathri Gangadharan100% (3)
- Endocrine Disorders and Their Effects in OrthodonticsDocumento29 pagineEndocrine Disorders and Their Effects in OrthodonticsSurabhi SaxenaNessuna valutazione finora
- Drug Induced OsteoporosisDocumento8 pagineDrug Induced OsteoporosislueglemeNessuna valutazione finora
- Adverse Effects of Corticosteroids On BoneDocumento6 pagineAdverse Effects of Corticosteroids On BoneDiego Rivas-VazquezNessuna valutazione finora
- Clinical Features and Evaluation of Glucocorticoid-Induced Osteoporosis - UpToDateDocumento15 pagineClinical Features and Evaluation of Glucocorticoid-Induced Osteoporosis - UpToDateatp.nutricionydeporteNessuna valutazione finora
- Host ModulationDocumento17 pagineHost ModulationIbrar HumayunNessuna valutazione finora
- Secondary OsteoporosisDocumento22 pagineSecondary OsteoporosisBenny Chris TantoNessuna valutazione finora
- Sem Osteoporosis (Edit)Documento31 pagineSem Osteoporosis (Edit)Rhomizal MazaliNessuna valutazione finora
- Doosis KalsiumDocumento8 pagineDoosis KalsiumNurul Kamilah SadliNessuna valutazione finora
- Nursing Pharmacology 2Documento26 pagineNursing Pharmacology 2milkah mwauraNessuna valutazione finora
- UK Guideline 2020 220709Documento8 pagineUK Guideline 2020 220709rnwkrnrnNessuna valutazione finora
- B Is Phospho NatesDocumento34 pagineB Is Phospho NatesGayathri RaveendranNessuna valutazione finora
- 5 GlucocorticoidesDocumento16 pagine5 Glucocorticoideshernan gutierrezNessuna valutazione finora
- 3 OsteoporosisDocumento6 pagine3 Osteoporosisiraqeana100100Nessuna valutazione finora
- ADRENOCORTICOIDSDocumento32 pagineADRENOCORTICOIDSJoyce VillanuevaNessuna valutazione finora
- Do NotDocumento5 pagineDo NotDominique MendozaNessuna valutazione finora
- 3 Musculo Skeletal Disorders - The Missing FactorsDocumento63 pagine3 Musculo Skeletal Disorders - The Missing Factorsshirish gundalaNessuna valutazione finora
- Osteoporosis: Mohd Fikri Bin MustapaDocumento54 pagineOsteoporosis: Mohd Fikri Bin MustapaiqiessesNessuna valutazione finora
- Fosamax Plus FinalDocumento5 pagineFosamax Plus FinalRobin LimNessuna valutazione finora
- Medication Induced CushingDocumento3 pagineMedication Induced Cushinggermany23Nessuna valutazione finora
- Chapter 14: Bone Disease and Calcium Abnormalities in Elderly Patients With CKDDocumento5 pagineChapter 14: Bone Disease and Calcium Abnormalities in Elderly Patients With CKDHarun NasutionNessuna valutazione finora
- Masculoskeletal Disorders PharmacotherapyDocumento101 pagineMasculoskeletal Disorders PharmacotherapyWaaqoo Guutuu Waaqoo GuutuuNessuna valutazione finora
- OsteoporosisDocumento24 pagineOsteoporosisMousa SuhailNessuna valutazione finora
- Week 5 Endokrin WO - Michael GeraldilDocumento5 pagineWeek 5 Endokrin WO - Michael GeraldilMichael GNessuna valutazione finora
- Osteoarthritis: A Holistic Approach: Andrew Barr and Philip G ConaghanDocumento3 pagineOsteoarthritis: A Holistic Approach: Andrew Barr and Philip G ConaghanHendry BarcaNessuna valutazione finora
- Prevention and Treatment of Systemic Glucocorticoid Side EffectsDocumento10 paginePrevention and Treatment of Systemic Glucocorticoid Side EffectsMahmoud DiaaNessuna valutazione finora
- OsteoporosisDocumento43 pagineOsteoporosisMuna Hassan Mustafa100% (1)
- Dietetics 3 OA GoutDocumento22 pagineDietetics 3 OA Goutkanza MughalNessuna valutazione finora
- OsteoporosisDocumento47 pagineOsteoporosisSasha VaidyaNessuna valutazione finora
- J of Bone Mineral Res - 2016 - Zanchetta - Impaired Bone Microarchitecture Improves After One Year On Gluten Free Diet ADocumento8 pagineJ of Bone Mineral Res - 2016 - Zanchetta - Impaired Bone Microarchitecture Improves After One Year On Gluten Free Diet Adiego.alejandro.iraheta.cruzNessuna valutazione finora
- General - OsteoporosisDocumento9 pagineGeneral - OsteoporosisAsma NazNessuna valutazione finora
- Side Effects of Systemic Corticosteroids in ChildrenDocumento6 pagineSide Effects of Systemic Corticosteroids in ChildrenChamara AtukoralaNessuna valutazione finora
- Prevention and Treatment of Glucocorticoid-Induced OsteoporosisDocumento13 paginePrevention and Treatment of Glucocorticoid-Induced OsteoporosisharryparralesNessuna valutazione finora
- Osteoporosis in Elderly MenDocumento5 pagineOsteoporosis in Elderly MenFathia RachmatinaNessuna valutazione finora
- HTTPDocumento3 pagineHTTPapi-26071302Nessuna valutazione finora
- Osteoporosis: by Ali Naqvi, Jeff Hughes, and Shuktika NandkekolyarDocumento19 pagineOsteoporosis: by Ali Naqvi, Jeff Hughes, and Shuktika NandkekolyarAdelaide SharfinaNessuna valutazione finora
- Long-Term and Sequential Treatment For Osteoporosis: EndocrinologyDocumento14 pagineLong-Term and Sequential Treatment For Osteoporosis: EndocrinologyjoaovcfNessuna valutazione finora
- EnglishDocumento4 pagineEnglishTodirenche LarisaNessuna valutazione finora
- Rheumatologic Disorders: OsteoporosisDocumento7 pagineRheumatologic Disorders: Osteoporosisمحمد رحمان محمد الدليميNessuna valutazione finora
- Secondary OsteoporosisDocumento6 pagineSecondary OsteoporosisSitti FatimahNessuna valutazione finora
- General: Guideline TitleDocumento27 pagineGeneral: Guideline TitleltgcanlasNessuna valutazione finora
- Musculoskeletal SystemDocumento60 pagineMusculoskeletal Systempetite_chien15Nessuna valutazione finora
- Glucocorticoid-Induced Osteoporosis: Insights For The ClinicianDocumento10 pagineGlucocorticoid-Induced Osteoporosis: Insights For The ClinicianLucía del ValleNessuna valutazione finora
- Strategies For Minimizing Corticosteroid Toxicity: A Review: Roosy Aulakh and Surjit SinghDocumento7 pagineStrategies For Minimizing Corticosteroid Toxicity: A Review: Roosy Aulakh and Surjit SingherikafebriyanarNessuna valutazione finora
- Osteoporosis: Dr. Lubna DwerijDocumento26 pagineOsteoporosis: Dr. Lubna DwerijNoor MajaliNessuna valutazione finora
- 21223lbl PDFDocumento11 pagine21223lbl PDFMelia Fatrani RufaidahNessuna valutazione finora
- Glucocorticoid-Induced Osteoporosis: Insights For The ClinicianDocumento10 pagineGlucocorticoid-Induced Osteoporosis: Insights For The Clinicianarif fatkhurNessuna valutazione finora
- Management of Glucocorticoid-Induced Osteoporosis - Nature Reviews Rheumatology 6, 82 (2010) - Doi10.1038-Nrrheum.2009.259 - 000455Documento7 pagineManagement of Glucocorticoid-Induced Osteoporosis - Nature Reviews Rheumatology 6, 82 (2010) - Doi10.1038-Nrrheum.2009.259 - 000455srini durveshNessuna valutazione finora
- Chapter 25 GlucocorticoidsDocumento4 pagineChapter 25 GlucocorticoidsRachaelNessuna valutazione finora
- Regaining Ground in Osteoporosis: Pharmacists Leading The ChargeDocumento15 pagineRegaining Ground in Osteoporosis: Pharmacists Leading The ChargeJudith GraalNessuna valutazione finora
- Regaining Ground in Osteoporosis: Pharmacists Leading The ChargeDocumento15 pagineRegaining Ground in Osteoporosis: Pharmacists Leading The ChargeJudith GraalNessuna valutazione finora
- Metabolic Bone Disorders: By: DR - Shaheen Ahmed Dept of Oral SurgeryDocumento33 pagineMetabolic Bone Disorders: By: DR - Shaheen Ahmed Dept of Oral SurgeryhumaNessuna valutazione finora
- Glicocorticoide e MineralizaçãoDocumento9 pagineGlicocorticoide e Mineralizaçãomatheus galvãoNessuna valutazione finora
- OsteoporosisDocumento22 pagineOsteoporosisPrachi DSaNessuna valutazione finora
- Gotthardt 2017Documento9 pagineGotthardt 2017Emile VeenstraNessuna valutazione finora
- SR 2Documento33 pagineSR 2hussein alnasryNessuna valutazione finora
- HormonesDocumento9 pagineHormonesRamadhan AkmalNessuna valutazione finora
- Prevention and Treatment of Glucocorticoid-Induced Osteoporosis - UpToDateDocumento29 paginePrevention and Treatment of Glucocorticoid-Induced Osteoporosis - UpToDateatp.nutricionydeporteNessuna valutazione finora
- OsteoporosisDocumento9 pagineOsteoporosisRenuka Sivaram71% (7)
- Translational Endocrinology of Bone: Reproduction, Metabolism, and the Central Nervous SystemDa EverandTranslational Endocrinology of Bone: Reproduction, Metabolism, and the Central Nervous SystemGerard KarsentyNessuna valutazione finora
- Erc - 2005Documento58 pagineErc - 2005zulfantri1983Nessuna valutazione finora
- Medical Doctor in Faculty of Medicine, University of Udayana, - Pulmonologist in Faculty of Medicine, University of Indonesia, JakartaDocumento24 pagineMedical Doctor in Faculty of Medicine, University of Udayana, - Pulmonologist in Faculty of Medicine, University of Indonesia, Jakartazulfantri1983Nessuna valutazione finora
- Pathogenic Intestinal FlagellateDocumento15 paginePathogenic Intestinal Flagellatezulfantri1983Nessuna valutazione finora
- P WestermaniDocumento9 pagineP Westermanizulfantri1983Nessuna valutazione finora
- Pathogenic Free Living Amoeba: Parasitology Department Medical Faculty of Universitas Sumatera UtaraDocumento43 paginePathogenic Free Living Amoeba: Parasitology Department Medical Faculty of Universitas Sumatera Utarazulfantri1983Nessuna valutazione finora
- Alpha1 Antitrypsin DeficiencyDocumento12 pagineAlpha1 Antitrypsin Deficiencyzulfantri1983Nessuna valutazione finora
- Stop Mutation With Fixed-Dose Combination (FDCS)Documento30 pagineStop Mutation With Fixed-Dose Combination (FDCS)zulfantri1983Nessuna valutazione finora
- Resistensi Isolat Pneumokokus Invasif Studi Dari Negara AseaDocumento16 pagineResistensi Isolat Pneumokokus Invasif Studi Dari Negara Aseazulfantri1983Nessuna valutazione finora
- Levofloxacin Iv/Oral Versus Ceftriaxone Iv/ Cefuroxime Axetil Oral Therapy in Acute Exacerbation of Bronchiectasis in Persahabatan HospitalDocumento34 pagineLevofloxacin Iv/Oral Versus Ceftriaxone Iv/ Cefuroxime Axetil Oral Therapy in Acute Exacerbation of Bronchiectasis in Persahabatan Hospitalzulfantri1983Nessuna valutazione finora
- KPR 132 Echinostoma SPPDocumento4 pagineKPR 132 Echinostoma SPPzulfantri1983Nessuna valutazione finora
- "Therapeutic Drug Monitoring" Aminoglycoside & The Clinical ApplicationDocumento17 pagine"Therapeutic Drug Monitoring" Aminoglycoside & The Clinical Applicationzulfantri1983Nessuna valutazione finora
- Effect Radiation and Chemotherapy CancerDocumento13 pagineEffect Radiation and Chemotherapy Cancerzulfantri1983Nessuna valutazione finora
- KPR 132 Enterobius VermicularisDocumento1 paginaKPR 132 Enterobius Vermiculariszulfantri1983Nessuna valutazione finora
- The Role of Labacs in The Treatment of Copd: Hadiarto MangunnegoroDocumento36 pagineThe Role of Labacs in The Treatment of Copd: Hadiarto Mangunnegorozulfantri1983Nessuna valutazione finora
- 4.2 Immunologic DiseasesDocumento20 pagine4.2 Immunologic Diseaseszulfantri1983Nessuna valutazione finora
- Asap DR TjandraDocumento41 pagineAsap DR Tjandrazulfantri1983Nessuna valutazione finora
- Disturbances in Thought KJW 161Documento23 pagineDisturbances in Thought KJW 161zulfantri1983Nessuna valutazione finora
- Pharmacology of Disopyramide: Yosephine A. H. 020100058Documento20 paginePharmacology of Disopyramide: Yosephine A. H. 020100058zulfantri1983Nessuna valutazione finora
- Bronchitis: Synonyms and Related KeywordsDocumento27 pagineBronchitis: Synonyms and Related Keywordszulfantri1983Nessuna valutazione finora
- The Role of Non-Invasive Modalities in Diagnosis of Congenital Heart DiseaseDocumento33 pagineThe Role of Non-Invasive Modalities in Diagnosis of Congenital Heart Diseasezulfantri1983Nessuna valutazione finora
- Benefits of OTN in Transport SDNDocumento9 pagineBenefits of OTN in Transport SDNGhallab AlsadehNessuna valutazione finora
- Defect Origin (J) Phase Requirement Analysis Design Coding Unit TestingDocumento87 pagineDefect Origin (J) Phase Requirement Analysis Design Coding Unit Testingashish_jain_csNessuna valutazione finora
- DSR Codes - 1Documento108 pagineDSR Codes - 1lakkireddy seshireddyNessuna valutazione finora
- PositioningDocumento2 paginePositioningKishan AndureNessuna valutazione finora
- Foundation Design LectureDocumento59 pagineFoundation Design LectureJamaica MarambaNessuna valutazione finora
- No Client Too Far: Flexible Antenna Options TDMA GPS Sync ClientDocumento2 pagineNo Client Too Far: Flexible Antenna Options TDMA GPS Sync ClientFelix MartinezNessuna valutazione finora
- Industrial Revolution OutlineDocumento8 pagineIndustrial Revolution OutlineGeraldine GuarinNessuna valutazione finora
- EU - Guidance On GMP For Food Contact Plastic Materials and Articles (60p)Documento60 pagineEU - Guidance On GMP For Food Contact Plastic Materials and Articles (60p)Kram NawkNessuna valutazione finora
- Sop ExcelDocumento104 pagineSop ExcelRizky C. AriestaNessuna valutazione finora
- WL4000Documento1 paginaWL4000Laser PowerNessuna valutazione finora
- Oasis AirlineDocumento5 pagineOasis AirlineRd Indra AdikaNessuna valutazione finora
- Moisture and Total Solids AnalysisDocumento44 pagineMoisture and Total Solids AnalysisNicholas BoampongNessuna valutazione finora
- Practical Considerations in Modeling: Physical Interactions Taking Place Within A BodyDocumento35 paginePractical Considerations in Modeling: Physical Interactions Taking Place Within A BodyFábio1 GamaNessuna valutazione finora
- SFT PresentationDocumento16 pagineSFT Presentationapna indiaNessuna valutazione finora
- Burst Abdomen 3Documento12 pagineBurst Abdomen 3Satvik BansalNessuna valutazione finora
- Design of Cycle Rickshaw For School ChildrenDocumento23 pagineDesign of Cycle Rickshaw For School ChildrenAditya GuptaNessuna valutazione finora
- Legend Of Symbols: Chú Thích Các Ký HiệuDocumento9 pagineLegend Of Symbols: Chú Thích Các Ký HiệuKiet TruongNessuna valutazione finora
- 4 Pure BendingDocumento42 pagine4 Pure BendingOmarfirozNessuna valutazione finora
- Habitat Preference of Great Argus Pheasant ArgusiaDocumento11 pagineHabitat Preference of Great Argus Pheasant ArgusiaFaradlina MuftiNessuna valutazione finora
- Science7 q2 Mod6of8 Asexual Sexualrep v2Documento26 pagineScience7 q2 Mod6of8 Asexual Sexualrep v2Ishi OcheaNessuna valutazione finora
- Editor: Lalsangliana Jt. Ed.: H.Documento4 pagineEditor: Lalsangliana Jt. Ed.: H.bawihpuiapaNessuna valutazione finora
- Boundary ScanDocumento61 pagineBoundary ScanGéza HorváthNessuna valutazione finora
- Mechanism Design: A SeriesDocumento3 pagineMechanism Design: A Seriesamirmasood kholojiniNessuna valutazione finora
- Qa/Qc Mechanical Monthly Progress Report For June 2015: Area/System Description Status RemarksDocumento1 paginaQa/Qc Mechanical Monthly Progress Report For June 2015: Area/System Description Status RemarksRen SalazarNessuna valutazione finora
- Biasing Opamps Into Class ADocumento11 pagineBiasing Opamps Into Class AsddfsdcascNessuna valutazione finora
- Magical ExercisesDocumento5 pagineMagical ExercisesAnonymous ytxGqZNessuna valutazione finora
- Holowicki Ind5Documento8 pagineHolowicki Ind5api-558593025Nessuna valutazione finora
- Fo Transfer ProcedureDocumento8 pagineFo Transfer ProcedureadityasahayNessuna valutazione finora
- Section 08630 Metal-Framed SkylightDocumento4 pagineSection 08630 Metal-Framed SkylightMØhãmmed ØwięsNessuna valutazione finora
- Charles Haanel - The Master Key System Cd2 Id1919810777 Size878Documento214 pagineCharles Haanel - The Master Key System Cd2 Id1919810777 Size878Hmt Nmsl100% (2)