Documenti di Didattica
Documenti di Professioni
Documenti di Cultura
Cheat Sheet (Bio) 1
Caricato da
Kelvin KohDescrizione originale:
Copyright
Formati disponibili
Condividi questo documento
Condividi o incorpora il documento
Hai trovato utile questo documento?
Questo contenuto è inappropriato?
Segnala questo documentoCopyright:
Formati disponibili
Cheat Sheet (Bio) 1
Caricato da
Kelvin KohCopyright:
Formati disponibili
Cystic fibrosis.
A defect in the cystic fibrosis transmembrane conductance regulator
(CFTR) gene causes cystic fibrosis (CF). A protein made by this gene controls the
movement of the water and salt in and out of the body's cells. Genes in ppl with CF
incorrectly code proteins. This causes thick, sticky mucus and salty sweat.
Neurofibromatosis. Neurofibromatosis is caused by point mutations in the
Neurofibromin 1 or 2 gene.
Sickle-cell anemia. is caused by a point mutation in the -globin chain of haemoglobin,
causing the hydrophilic amino acid glutamic acid to b replaced with the hydrophobic
amino acid valine at the 6th position.
Disease Caused by Mutation
Cancer. Point mutations in multiple tumor suppressor proteins cause cancer.
A novel assay, Fast parallel proteolysis (FASTpp), might help swift screening of specific
stability defects of specific proteins in individual cancer patients. FASTpp measures the
quantity of protein that resists digestion under various conditions. A thermostable
protease is used, which cleaves specifically at
exposed hydrophobic residues.The FASTpp assay combines the thermal unfolding,
specificity of a thermostable protease for the unfolded fraction with the separation power
of SDS-PAGE. Due to this combination, FASTpp can detect changes in the fraction folded
over a large physico-chemical range of conditions including temperatures up to 85C, pH
6-9, presence or absence of the whole cytosolic proteome.
Specific diseases caused by insertions/deletions
Tay-Sachs Disease. Tay-Sachs Disease is a fatal disease affecting the central nervous
system.
Symptoms do not appear until approximately 6 months of age. The child becomes blind,
deaf, unable to swallow, atrophied, and paralytic.
Mutations in the -hexosaminidase A (Hex A) gene are known to affect the onset of TaySachs.
CancerInsertion/deletion mutations cause colorectal cancer and other cancers with
microsatellite instability.
While environmental factors contribute to the progression of prostate cancer, genetic
component also will.
There are over 500 mutations on chromosome 17 that seem to play a role in the
development of breast and ovarian cancer in the BRCA1 gene, many of which are
Insertion/deletion.
SNP and DISEASE
One study even identified two genes in which particular variants can slow the onset of
AIDS, demonstrating the potential of this approach for understanding why people vary in
their susceptibility to infectious diseases.
New technologies that are slashing the costs of sequencing and genome analyses will
make possible the simultaneous genome-wide search for SNPs and other DNA alterations
in individuals.
Proteomics
Proteomics is the large-scale study of proteins, particularly their structures and
functions.
Proteins are vital parts of living organisms, as they are the main components of the
physiological metabolic pathways of cells.
The proteome consists of the entire complement of proteins, including the modifications
made to a particular set of proteins, produced by an organism or system.
This will vary with time and distinct requirements, or stresses, that a cell or organism
undergoes.
Number of Proteins in Human
Analyzing genome sequences alone will not lead to new therapies to fight human
diseases.
Whereas the human genome has approximately 35,000 genes and theoretically the
ability to encode up to 35,000 corresponding proteins.
The occurrence of alternative RNA splicing and posttranslational modifications (PTM),
such as phosphorylations, acetylations, and glycosylations, or protein cleavages may
increase the expression of proteins to 500,0001,000,000.
The proteins reflect more accurately the intrinsic genetic mechanisms of the cell and
their impact on the microenvironment, since they are the effectors and characterize
Proteomics in Biomedical Research
Biomarkers are biomolecules that is associated with an increased risk of the disease and
serve as indicators of biological and pathological processes or physiological and
pharmacological responses to a drug.
Proteins that are impt indicators of physiological or pathological states may contribute to
the early diagnosis of disease, which may provide a basis for identifying the underlying
mechanism of disease development.
These differentially expressed proteins in serum have become an impt in monitoring the
state for disease.
Comprehensive proteome of human serum fluid with high accuracy and availability has
the potential to open new doors for disease biomarker discovery and for disease
diagnostics.
Proteomics in Cancer Diagnostics
Allied to genomics, proteomics technologies is valuable for identifying new markers that
improve screening, early diagnosis, prognosis and prediction of therapeutic response or
toxicity, as well as the identification of new therapeutic targets.
Studies on the proteome in cancer have used tissue samples and biological fluids
including serum, plasma, saliva, and cerebrospinal fluid in search for the detection of
diagnostic, predictive, and prognostic biomarkers.
Among the proteomics tools, mass spectrometry (MS) is one of the most used
techniques for identifying unknown proteins. The mass spectrometer is an analytic
instrument capable of converting neutral molecules into gaseous ions and separating
them according to their mass-to-charge (m/z) ratio by using an electromagnetic field.
Tandem mass spectrometry (MS/MS) offers information about specific ions. In this
approach, distinct ions are selected based on their m/z from the first round of MS and are
fragmented by a number of methods of dissociation, such as colliding the ions with a
stream of inert gas, as in collision-induced dissociation or higher energy collision
dissociation. Other methods of ion fragmentation include electron-transfer dissociation
and electron-capture dissociation .
These fragments are then separated based on their individual m/z ratios in another round
of MS. MS/MS is commonly used to sequence proteins and oligonucleotides, as the
fragments can be used to match predicted peptide or nucleic acid sequences that are
found in databases. These sequence fragments can then be organized in silico into fulllength sequence predictions.
A sample is injected into the mass spectrometer, ionized and accelerated and then
analyzed by mass spectrometry (MS1). Ions from the MS1 spectra are then selectively
fragmented and analyzed by mass spectrometry (MS2) to give the spectra for the ion
fragments.
Sugars, amino acids and nucleotides can polymerize to form macromolecules called
polysaccharides, proteins and nucleic acids. When sugars, amino acids and nucleotides
polymerize, water is released. In the process of hydrolysis, a water molecule reacts with
the bond linking the monomers. A monomer is broken off, resulting in a shorter polymer.
Sugars are defined by the presence of an carbonyl group and multiple hydroxyl groups.
Sugars like glucose can exist in both linear and ring forms. Like many organic molecules,
sugars are chiral molecules- they can exist as right-handed (D) or left handed (L)
isomers. Right-handed (D) forms predominate in cells.
When glucose forms a ring, the hydroxyl group
attached to the
number 1 carbon is locked into one of
two alternate positions: either
below the plane of the
ring, or above it. These two ring forms of glucose are
called alpha () (down) and beta () (up), respectively:
Examples
of
sugar
polymers:
Starch
is
polymerized
glucose, in which -glucose monomers are
polymerized via a
Production of glucose 6-phosphate
Glucose 6-phosphate is produced by phosphorylation of glucose on the sixth
carbon.
This is catalyzed by the enzyme hexokinase in most cells, and, in higher
animals, glucokinase in certain cells, most notably liver cells. One molecule of
ATP is consumed in this reaction.
The major reason for the immediate phosphorylation of glucose is to prevent
diffusion out of the cell. The phosphorylation adds a charged phosphate grp so
the glucose 6-phosphate cannot easily cross the cell membrane.
Two Forms of Starch
Around 30%, tightly
packed structure,
more resistant to
Digestion. Amylose can
Exist in Helical Forms.
Around 70%,highly
branched structure,
being formed of 2,000
to 200,000 glucose
units can be quickly
degraded
Amylopectin on the other hand is a branched-chain polysaccharide where in
addition to the -1,4-glycosidic bonds there is the occasional -1,6-glycosidic
bonds. Branching occurs about every 24-30 glucose units. Helical structure of
amylopectin is disrupted by branching.
Glycogen
A multibranched polysaccharide of glucose that is a form of energy storage in
animals and fungi.
In humans, glycogen is made and stored in the cells of the liver and the
muscles, and functions as the secondary longterm energy storage (primary
energy stores being fats).Glycogen is the analogue of starch, having a similar
structure to amylopectin, but more branched and compact. Glycogen is found in
the form of granules in the cytoplasm in many cell types, and plays an impt role
in the glucose cycle. Glycogen forms an energy reserve that can be quickly
mobilized to meet the need for glucose, but is less compact than the energy
reserves of triglycerides.
Glycogen is a branched biopolymer consisting of linear chains of glucose
residues with further chains branching off every 10 glucoses. Glucoses are linked
together linearly by (14) glycosidic bonds. Branches are linked to the chains
and are branched off by (16) glycosidic bonds.
Cellulose
Made of -glucose. To form glycosidic links, each -glucose molecule is rotated
180o compared to the one next to it. Has straight, unbranched chains that run
parallel to one another. Hydrogen bond links the chains. The -glycosidic link
between glucose molecules in cellulose results in a polymer that forms a long
linear strand. The hydroxyl groups of one cellulose molecule are free to H bond
with the hydroxyls of adjacent molecules. In plants, the long strands of cellulose
bundle together to form microfibrils. Bundles of microfibrils form plant cell walls.
So many hydrogen bonds help to strength cellulose
This makes cellulose a good structural material, hence its use in plant cell walls
to aid rigidity
cellulose does this by grouping together to form microfibrils
Cellulose prevents cell bursting, so they are turgid when full with water. This
helps support stems
Other
important
structural
polysaccharides
are
chitin
and
peptidoglycan
Both composed of polymers of amino sugars, such as N-acetyl-glucosamine
(chitin) or [N-acetyl-glucosamine plus N-acetyl-muramic acid] (Peptidoglycan).
A mesh of peptidoglycan chains, crosslinked by covalent bonds, make up the
tough and flexible bacterial cell wall. (antibiotics poison the bacterial enzymes
that synthesize cell wall)
Chitin (mono monomer) (Parallel strands joined by hydrogen bonds)
Chitin is a long-chain polymer of a Nacetylglucosamine, a derivative of glucose.
The main component of the cell walls of fungi, the exoskeletons of arthropods
such as
crustaceans and insects, the radulae of molluscs, and the beaks and internal
shells of cephalopods.
The structure of chitin is comparable to the polysaccharide cellulose, forming
crystalline
nanofibrils. In terms of function, it may be compared to the protein keratin.
It form covalent -1,4 linkages (similar linkages between glucose units forming
cellulose).
Chitin is cellulose with one hydroxyl group replaced with an acetyl amine group.
Peptidoglycan (Parallel strands joined by peptide bonds)
also known as murein, is a polymer consisting of sugars and amino acids that
forms a mesh-like layer outside the plasma membrane of most bacteria, forming
the cell wall.
The sugar component consists of alternating residues of -(1,4) linked Nacetylglucosamine and N-acetylmuramic acid. Attached to the N-acetylmuramic
acid is a peptide chain of three to five amino acids. The peptide chain can be
crosslinked to the peptide chain of another strand forming the 3D mesh-like layer.
Peptidoglycan serves a structural role in the bacterial cell wall, giving structural
strength, as well as counteracting the osmotic pressure of the cytoplasm.
peptidoglycan helps maintain the structural strength of the cell.
Peptidoglycan is also involved in binary fission during bacterial cell
reproduction.
The peptidoglycan layer is substantially thicker in Gram-positive bacteria than
in Gram-negative bacteria, with the attachment of the S-layer.
Peptidoglycan forms around 90% of the dry weight of Gram-positive bacteria
but only 10% of Gram-negative strains.
Meso-diaminopimelic acid (DAP) for Gram Positive
Nucleic Acids
Two classes of nucleic acids: deoxyribonucleic acid (DNA) and ribonucleic acid
(RNA)
Cells use DNA to determine and control the synthesis of proteins with the help
of mRNA.
Other shorthand notation for DNA sequence:
5- TCA 3
Two complementart strands of DNA can specifically pair with each other,
beacuse the bases form specific hydrogen bond.
Double helix structure
DNA contains major and minor grooves and many DNA-binding, gene regulatory
proteins prefer to bind nucleotides located in the major groove.
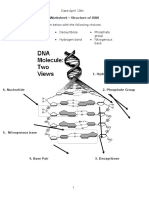
1. DNA molecule consists of two polynucleotide chains in a double helix
configuration.
2. The two strands are anti-parallel.
3. The sugar-phosphate backbone is on the outside of the helix, bases are on the
inside.
4. A always pairs with T; G always pairs with C. The sequence of one strand (5
3) dictates the
sequence of the other strand.
5 GCATGCAATGCCGAATG 3
3 CGTACGTTACGGCTTAC 5
5. 2nm wide diameter: perfect for purine-pyrimidine bond.
6. Base pairs are 3.4 apart: a complete 360 turn of the helix is 34 , which
equals 10 base pairs.
7. The helix has a major groove and a minor groove.
8. When heated or when deviating from physiological conditions, hydrogen bonds
between the two DNA strands are cleaved and the strands are separated from
each other to form single string DNA (ssDNA).
RNA
The structure of RNA is similar to that of DNA except:
1. The nucleotide subunits have ribose, rather than deoxyribose as the sugar
2. Uridine is substituted for thymidine
3. RNA is generally found as a single-stranded molecule in cells.
3-D structure of RNA
GCAU instead of GCAT
Due to the additional OH group on the ribose sugar, steric hindrance is too
great to allow for the formation of a double strand. So, RNA exists as a single
stranded molecule.
RNA can loop back to form internal self base-paired structures, called stemloop structures
Transfer RNA Contains a Modified Base from Uridine
It is found in tRNA, found with thymidine and cytosine in the TC arm and is one
of the invariant regions of tRNA. It is expected to play a role in association with
aminoacyl transferases during their interaction with tRNA, and hence in the
initiation of translation. Recent studies suggest it may offer protection from
radiation.
Amino Acids:
RNA molecules can form complex structures with
pockets and clefts on their
Classification
surface. Also the purine and pyrimidine nitrogenous bases contain
chemically
based
on R
reactive functional groups that can catalyze chemical
reactions.
group
Proteins Proteins are synthesized beginning with the amino
terminal
amino
Basic
amino acids
acid and finishing with the carboxy terminal amino acid. And when writing
Acidic out
the amino acid sequence of a protein, the convention
the amino terminus on
amino is
acids
the left , the carboxy terminus on the right.
Aliphatic
amino acids
Aromatic
amino acids
Hydroxyl containing
amino
acids
Sulfur containing amino acids
Secondary amino acids
What makes a strong acid?
HCL is a much stronger acid than acetic acid:
Ka = 107
MeCOOH
Ka = 1.74 x 10-5
This is to do with the strength (stability) of the conjugate base, Cl- is not strong enough to
deprotonate H3O+, but acetate is. In other words, the chloride ion is inherently more stable than
the acetate ion.
An acids pKa depends on the stability of its conjugate base.
- The stronger the acid HA, the weaker its conjugate base A- The stronger the base A-, the weaker the conjugate acid HA.
For example:
- HI with pKa of -10, is a strong enough acid to protonate most functional groups. Its conjugate
base, I- is not really basic.
- Methyl lithium is a powerful base, which behaves as CH3-. The conjugate acid is CH4, which isnt
acidic with pKa = 48.
Peptide Bond Formation
Condensation rxn Between NH2 of n residue and COOH of n+1 residue. Rigid, inflexible.
Loss of 1 water molecule.
The peptide bond has a barrier to rotation. The resonance structure explains this, and bond length
comparisons are consistent with partial double bond character. As a consequence, the atoms are all
constrained to lie in the same plane. The peptide bond is planar. But the planar conformation can be
accommodated in two alternate forms denoted as trans and cis. The more stable trans form. The
cis form is less stable because of its greater steric repulsion between the C atoms and their
attached groups.
Peptides and Proteins
Peptides and protein made up from long chains of amino acids via peptide bonds.
There are two types of protein structures: fibrous (elongated proteins not soluble in water and
providing structural support), and globular (spherical proteins soluble in water and have specific
function in the immune system and metabolism).
The structural proteins
Primary Structure The sequence of amino acids The peptide bond is rigid and can not move due
to its partial double bond character of C-N bond. To write peptide and protein always from Nterminal to C-terminal.
Secondary Structure Regular elements such as -helices and - sheets, which are formed
between relatively small parts of the protein sequence. They are determined by the local
conformation of the polypeptide backbone. -helix Most abundant; ~35% of residues in a protein
Repetitive secondary structure 1.5 rise in 100 rotation C=Oof i forms H bonds with N-H of
residue i+4 Intra-strand H bonding C=O groups are parallel to the axis; side chains point away
from the axis Polar ends present at surfaces Amphipathic All N-H and CO are H-bonded, except
first N-H and last CO
-sheet
Other major structural element Basic unit is a -strand Usually 5-10
Residues Can be parallel or anti-parallel based on the relative directions
of interacting -strands Pleated appearance
Another important interaction is the formation of hydrogen bonds
between the carbonyl oxygen and amide hydrogens on adjacent regions
of the peptide backbone:
-sheet (with primary structure)
(a) antiparallel and (b) parallel -sheet. Blue and white
beads represent the positively charged (Arg) and
hydrophobic residues, respectively, and the polar
residue (Tyr) and Gly residues are denoted by green
beads. Solid lines indicate the disulfide bonds between
Cys residues, and dotted lines indicate the backbone
hydrogen bond (H-bond).
-pleated sheet
The chains are folded so that they lie alongside each other. All that means is that next-door chains
are heading in opposite directions. Given the way this particular folding happens, that would seem
to be inevitable.
The generalized structure of an amino acid: Amino acids are chiral
Some of the amino acids have hydrophobic side chains; others have hydrophilic side chains. The
molecules (can exist as right or left handed forms). But whereas in the case of
different AA like to interact with each other, and the protein chain folds to maximize these
sugar, the right-handed form predominates in cells, in the case of amino acids, it interactions. Also one important way a protein folds is such that the hydrophobic AA will be in the
is the left-handed form that is found in cells.
interior of the folded protein, and the hydrophilic AA will be on the surface. In addition to the
Amino Acids
backbone hydrogen bonds that permit the formation of secondary structures, other interactions
Names for amino acids are abbreviated to either three symbol or a one symbol between the side chains of the various AA govern the overall the folded structure of the protein.
short form.
20 amino acids found in living organisms.
Building blocks of peptide and protiens
Linear chain of amino acids forms peptide/protein.
Peptides - Small peptides with fewer than about ten amino acids are called
oligopeptides
and peptides with more than ten amino acids are termed polypeptides.
Proteins Chain of amino acids with molecular weights of more than 10,000
(50100 amino acids) are usually termed proteins.
R group varies, thus, can be classified based on R-group.
Glycine is the simplest amino acid. Side chain R=H.
Unique because Gly -carbon is achiral.
Chiral: when a molecule is not superimposable on its mirror image
Zwitterionic character, pK and pI
Tertiary Structure
At the pH under physiological conditions (pH 6-7), the amino group (pK
Describe the complete three-dimensional structure of whole polypeptide chain. Include the
8.7~10.7) is ionized to NH3+ and the carboxyl group (pK 1.8~2.5) is ionized to relationship of different domains formed by the proteins secondary structure and the interactions
COO-. So, at physiological pH,. amino acids are zwitterionic
Level
Description
Stabilized by:
of the amino
acid substituent R group.
pK is the dissociation constant for H+.
Primary
The sequence
amino acids
Peptide bondsstable within a restricted range of
The specific
folding of of
a protein
is only thermodynamically
pI (isoelectric point) - It is a specific pH value at which aa exhibits no net
Secondar
Formation
of a-helices
b-pleated H-bonding
between
peptide groups along the peptide
environmental
parameters,
e.g.,and
Temperature,
pH, ionic
strength
charge.
yQuaternary
sheets
backbone
Structure
It can be estimated via the Henderson- Hasselbalch equation pI = (pKNH3+
Tertiary
Overall
three-dimensional
shape arrangement
Bonds and other
interactions
Quaternary
structure
is the 3-Dimensional
of multiple
folded between
protein orR-groups,
coiling or
of a polypeptide
between
R-groups
and the peptide
backbone
+pKCOOH), where pKi and pKj are the dissociation constants of the ionization
protein molecules
in a multi-subunit complex by
hydrogen
bond, electrostatic
attraction
and sulfide
Quaterna
groups involved.
bridge. Shape produced by combination of Bonds and other interactions between R-groups, and
ry
polypeptides
between
peptide
backbones
of
different
polypeptides
At its isoelectric point, amino acid remains stationary under an applied electric When functional unit consist of two or more structural domains, we speak of the quaternary
field.
structure of the protein.
Acid-Base Properties
It is the linear sequence of amino acids in a protein that determines the 3-dimensional folded
pH and pKa
structure of that protein.
The pH of a solution is a measure of the acidity of the solution. It is defined as
Another way to state this concept is to say that proteins fold to their thermodynamically
Where ] is the concentration of hydronium ions in the solution.
most stable state; ie, each particular folded protein has maximized all its particular
Consequently, the pH of a solution depends on two things
combinations of possible hydrogen bonds, electrostatic interactions, hydrophobic interactions, etc,
-The concentration of the solution if we have two solutions of the same acid, the in its final folded shape:
more concentrated solution will have more free H3O+ ions and therefore a lower
pH.
-The acid in question if we have two equally concentrated solution of acid, the
solution of a strong acid will have a lower pH than that of a weak acid, because it
is fully dissociated and therefore produces more H3O+ ions. HCL for example, is
completely dissociated.
Therefore, we see that pH does not measure the strength of an acid, but the
acidity of a given solution.
The pH of water is 7. This means that a solution of pure water has 10-7 mol/dm3 To demonstrated that the linear sequence of amino acids in a protein determines the folded
of hydronium ions. This can only happen through the autoprotolysis of water:
structure of that protein. Using a chemical called urea, he unfolded a protein called Ribonuclease-A,
and then reduced its internal disulfide bonds with mercaptoethanol. (Disulfide bonds stabilize the
This mean that in water,
folded protein in its original shape.) When the urea and mercaptoethanol is removed, the
To be clearer about what a strong and weak acid is, we look at the reaction:
ribonuclease renatured back, and regained full enzymatic activity. Based on experiments, the
information for the complete, correct folding of a protein is in the linear AA sequence of that protein.
The position of the equilibrium is measured by the equilibrium constant
One of the impt functions of proteins is to serve as catalysts for chemical reactions necessary for
Now, in dilute solutions of acid, stays roughly constant at about 56 mol/dm3. we life. Proteins that functions as chemical catalysts are called enzymes. The complex surface of a
Potrebbero piacerti anche
- Children's Respiratory NursingDa EverandChildren's Respiratory NursingJanice MightenNessuna valutazione finora
- Bio Cheat SheetDocumento2 pagineBio Cheat Sheetboardermikie69100% (1)
- To Rejuvenate With The Blood Plasma Of Young PeopleDa EverandTo Rejuvenate With The Blood Plasma Of Young PeopleNessuna valutazione finora
- Biology NotesDocumento6 pagineBiology NotesElizaNessuna valutazione finora
- 210 Biology of CholeraDocumento4 pagine210 Biology of CholeraQuang Huy PhạmNessuna valutazione finora
- Zoology Notes: 017 Chapter 13Documento3 pagineZoology Notes: 017 Chapter 13humanupgrade100% (1)
- 16: Work, Power, and Energy: Key Physics Terms Key ConceptsDocumento1 pagina16: Work, Power, and Energy: Key Physics Terms Key ConceptsBlaine RogalskiNessuna valutazione finora
- Rules For Naming Ionic Compounds Containing Polyatomic IonsDocumento2 pagineRules For Naming Ionic Compounds Containing Polyatomic Ionsrotsacreijav77777Nessuna valutazione finora
- As Unit 2 Edexcel Biology PDFDocumento2 pagineAs Unit 2 Edexcel Biology PDFBreakfree Michigan50% (2)
- Unit 5 Notes Biology AqaDocumento94 pagineUnit 5 Notes Biology Aqa李超然Nessuna valutazione finora
- OCR Biology F211 Cells, Exchange and Transport Revision NotesDocumento4 pagineOCR Biology F211 Cells, Exchange and Transport Revision Notes24wrightphilip100% (1)
- Overview: Cellular Messaging: © 2011 Pearson Education, IncDocumento69 pagineOverview: Cellular Messaging: © 2011 Pearson Education, IncbbyiknowNessuna valutazione finora
- Cytogenetics - Lesson 2 - Dna Structure and Dna ExtractionDocumento8 pagineCytogenetics - Lesson 2 - Dna Structure and Dna ExtractionAli TaguibaoNessuna valutazione finora
- DNA Structure and ReplicationDocumento12 pagineDNA Structure and ReplicationJhune Dominique GalangNessuna valutazione finora
- Stefan Boltzmann Law PDFDocumento3 pagineStefan Boltzmann Law PDFESAKKIMALA SNessuna valutazione finora
- MeiosisDocumento24 pagineMeiosisMuhammad HamzaNessuna valutazione finora
- Summary Notes - Topic 5 Edexcel (A) Biology A-LevelDocumento6 pagineSummary Notes - Topic 5 Edexcel (A) Biology A-LevelAhmad MohdNessuna valutazione finora
- Types of Intramolecular Forces of AttractionDocumento5 pagineTypes of Intramolecular Forces of AttractionGrazielle AlmazanNessuna valutazione finora
- 4-Bio Factsheets - Protein SynthesisDocumento4 pagine4-Bio Factsheets - Protein SynthesisAbir OmarNessuna valutazione finora
- MAS Cheat SheetDocumento6 pagineMAS Cheat SheetDark LordNessuna valutazione finora
- Edexcel Biology Unit 3 (Alternative) June 2010 Examiner ReportDocumento22 pagineEdexcel Biology Unit 3 (Alternative) June 2010 Examiner Reportgoharayub3909100% (1)
- SNC2D8 - Physics: Motion Review: Key TermsDocumento6 pagineSNC2D8 - Physics: Motion Review: Key TermsOTKILLNessuna valutazione finora
- Protein Synthesis SummaryDocumento2 pagineProtein Synthesis SummaryTrinitee Wesner100% (1)
- Test DocumentDocumento99 pagineTest Documentwoilau100% (1)
- Phylogenetic Trees Click Learn WorksheetDocumento4 paginePhylogenetic Trees Click Learn Worksheetfcm31450% (1)
- Enzymes (AS Level Bio)Documento23 pagineEnzymes (AS Level Bio)DrMufaddal RampurwalaNessuna valutazione finora
- CHAPTER 3 - Technique in Field InvestigationDocumento99 pagineCHAPTER 3 - Technique in Field Investigationnur ilyanaNessuna valutazione finora
- Summary Notes - Topic 6 Microbiology, Immunity and Forensics - Edexcel (IAL) Biology A-LevelDocumento11 pagineSummary Notes - Topic 6 Microbiology, Immunity and Forensics - Edexcel (IAL) Biology A-LevelSaeed AbdulhadiNessuna valutazione finora
- Chapter 11 - Thermal Properties of MatterDocumento6 pagineChapter 11 - Thermal Properties of MatterFirNessuna valutazione finora
- Zoology Notes On Nervous SystemDocumento3 pagineZoology Notes On Nervous SystemMorgan MessingNessuna valutazione finora
- Phylogenetic Tree Creation Morphological and Molecular Methods For 07-JohnsonDocumento35 paginePhylogenetic Tree Creation Morphological and Molecular Methods For 07-JohnsonCHRISTEROP100% (2)
- 03: Kinematics in One Dimension: Key Physics Terms Constant Velocity vs. Constant AccelerationDocumento1 pagina03: Kinematics in One Dimension: Key Physics Terms Constant Velocity vs. Constant AccelerationBlaine RogalskiNessuna valutazione finora
- Summary Notes - Topic 6 Microbiology, Immunity and Forensics - Edexcel (IAL) Biology A-LevelDocumento11 pagineSummary Notes - Topic 6 Microbiology, Immunity and Forensics - Edexcel (IAL) Biology A-LevelsalmaNessuna valutazione finora
- 056 - Autonomic Nervous SystemDocumento4 pagine056 - Autonomic Nervous SystemSan SiddzNessuna valutazione finora
- Torque and Rotation PhysicsDocumento20 pagineTorque and Rotation PhysicsSaf Tanggo DiampuanNessuna valutazione finora
- Physics Cheat SheetDocumento2 paginePhysics Cheat SheetSam L'Huillier75% (4)
- Molecular Biology Unit 2 (DNA & RNA)Documento40 pagineMolecular Biology Unit 2 (DNA & RNA)Fuz Nas100% (2)
- Plant Adaptations To Dry Environments PDFDocumento2 paginePlant Adaptations To Dry Environments PDFStarlight 101Nessuna valutazione finora
- PG 7 - 9 Calculation in Chemistry HandoutDocumento0 paginePG 7 - 9 Calculation in Chemistry Handout金时进Nessuna valutazione finora
- BiologyDocumento41 pagineBiologymina0% (1)
- A224 Why We Need Biodiversity MedicineDocumento3 pagineA224 Why We Need Biodiversity Medicineramloghun veerNessuna valutazione finora
- Chemical Equilibrium NotesDocumento4 pagineChemical Equilibrium NotesHaile CordaNessuna valutazione finora
- ZOOLOGY BasicsNotes PDFDocumento10 pagineZOOLOGY BasicsNotes PDFknarasimhareddyNessuna valutazione finora
- Cell Processes Cheat SheetDocumento3 pagineCell Processes Cheat SheetsarasNessuna valutazione finora
- State of Matter-Gas)Documento48 pagineState of Matter-Gas)bigsnailz100% (1)
- Matter and Its PropertiesDocumento10 pagineMatter and Its PropertiesGerald CatiponNessuna valutazione finora
- Cellular Respiration 1Documento28 pagineCellular Respiration 1Maria Flor PabeloniaNessuna valutazione finora
- Preliminary Instructions: Cu Ni CR Fe FeDocumento4 paginePreliminary Instructions: Cu Ni CR Fe FeEmmanuel Ryan100% (1)
- Nucleic Acids and Protein Synthesis RevisedDocumento60 pagineNucleic Acids and Protein Synthesis Revisedemanuel coatesNessuna valutazione finora
- Gel ElectrophoresisDocumento6 pagineGel Electrophoresisapi-385481351Nessuna valutazione finora
- ThermochemistryDocumento17 pagineThermochemistryBrian Smith100% (9)
- Molecular Biology of The Gene: Chapter ObjectivesDocumento17 pagineMolecular Biology of The Gene: Chapter Objectivesirene9tan9ailianNessuna valutazione finora
- Unit 6Documento10 pagineUnit 6Anonymous XU2T0mf100% (1)
- Body System ChecklistDocumento6 pagineBody System Checklistapi-422967453Nessuna valutazione finora
- Biology Summer Study Transport in PlantsDocumento9 pagineBiology Summer Study Transport in PlantsAya100% (1)
- Genetic Disorder and Cell MembraneDocumento7 pagineGenetic Disorder and Cell MembraneKumo FuyuNessuna valutazione finora
- Plasma EnzymesDocumento13 paginePlasma EnzymesBobskinnyNessuna valutazione finora
- Biology UNIT 1 Revision GuideDocumento53 pagineBiology UNIT 1 Revision GuideChristopher DoNessuna valutazione finora
- Biochem ReportDocumento10 pagineBiochem ReportLahari PylaNessuna valutazione finora
- AQA (B) AS Module 1: "Sharing Will Enrich Everyone With More Knowledge."Documento78 pagineAQA (B) AS Module 1: "Sharing Will Enrich Everyone With More Knowledge."NverLeaveUsNessuna valutazione finora
- Protein DNA InteractionsDocumento16 pagineProtein DNA InteractionsAvi VermaNessuna valutazione finora
- Biology 12 Unit 5 Dna Worksheet - Dna Strucuture 1Documento2 pagineBiology 12 Unit 5 Dna Worksheet - Dna Strucuture 1api-338578874100% (1)
- Proteins & EnzymesDocumento15 pagineProteins & EnzymesShruti Baid BafnaNessuna valutazione finora
- Quiz Protein and EnzymeDocumento4 pagineQuiz Protein and EnzymeAndrew DonaireNessuna valutazione finora
- Ncert Line by Line 28updated29-286-299Documento14 pagineNcert Line by Line 28updated29-286-299123Nessuna valutazione finora
- ProteinDocumento33 pagineProteinIhsan Badsha100% (1)
- Amino Acid Sequences and Evolutionary RelationshipsDocumento6 pagineAmino Acid Sequences and Evolutionary RelationshipsAbby Gail DigalNessuna valutazione finora
- Adn ArmableDocumento2 pagineAdn ArmableMarcela Ino100% (1)
- Ppcatalogue 2 PDFDocumento527 paginePpcatalogue 2 PDFrutwickNessuna valutazione finora
- Test On Molecular Biology NEETDocumento4 pagineTest On Molecular Biology NEETSadananda KumbhakarNessuna valutazione finora
- Pmwasabi VimentinDocumento4 paginePmwasabi VimentinAlleleBiotechNessuna valutazione finora
- Hand Out-Biochemistry-2022Documento3 pagineHand Out-Biochemistry-2022Rounak RajNessuna valutazione finora
- Protein: Bahan Ajar Kimia Xii/2 Ma Darul Ulum Muhammadiyah Galur TAHUN PELAJARAN 2013/2014Documento24 pagineProtein: Bahan Ajar Kimia Xii/2 Ma Darul Ulum Muhammadiyah Galur TAHUN PELAJARAN 2013/2014syukurajiNessuna valutazione finora
- Enzyme Activity: BY Deeparubini P Snekha S MDocumento20 pagineEnzyme Activity: BY Deeparubini P Snekha S MdeepaNessuna valutazione finora
- Protein As MicromoleculesDocumento7 pagineProtein As MicromoleculesPrincess JacintoNessuna valutazione finora
- C6 Nucleic AcidsDocumento57 pagineC6 Nucleic AcidsSoraya Al-obinayNessuna valutazione finora
- Letter: de Novo Protein Design by Citizen ScientistsDocumento19 pagineLetter: de Novo Protein Design by Citizen ScientistsLivsNessuna valutazione finora
- Chromatin RemodelingDocumento5 pagineChromatin RemodelingRohit GargNessuna valutazione finora
- Isolation and Characterization of Collagen From Chicken FeetDocumento5 pagineIsolation and Characterization of Collagen From Chicken FeetMohammed AdilNessuna valutazione finora
- Isoenzymes-Multienzyme Complex PDFDocumento20 pagineIsoenzymes-Multienzyme Complex PDFintaliallitaNessuna valutazione finora
- EnzymesDocumento25 pagineEnzymesAbdirazak AliNessuna valutazione finora
- Csir Ugc Net - Life Sciences - Free Sample TheoryDocumento12 pagineCsir Ugc Net - Life Sciences - Free Sample TheorySyamala Natarajan100% (1)
- Formal Report: Stability of ProteinsDocumento7 pagineFormal Report: Stability of ProteinsQueenie Tenedero100% (2)
- NEET UG Biology Biomolecules-2Documento20 pagineNEET UG Biology Biomolecules-2VyjayanthiNessuna valutazione finora
- Unit 2 Amino Acids and PeptidesDocumento23 pagineUnit 2 Amino Acids and PeptidesAndrei PusungNessuna valutazione finora
- Protein and The Element Sulfur: (No Need To Define)Documento3 pagineProtein and The Element Sulfur: (No Need To Define)Luis Gatchalian LacanilaoNessuna valutazione finora
- Unit 4: Gizmos Lab Simulation: RNA and Protein SynthesisDocumento7 pagineUnit 4: Gizmos Lab Simulation: RNA and Protein Synthesisjibityy junnnNessuna valutazione finora
- AminoaciziDocumento28 pagineAminoaciziBogdan AlexNessuna valutazione finora
- Dna Sequencing (Sanger's Method)Documento12 pagineDna Sequencing (Sanger's Method)Ajay KumarNessuna valutazione finora
- Lecture 5: The Chemical Basis of HeredityDocumento19 pagineLecture 5: The Chemical Basis of HeredityShigrid Ann DocilNessuna valutazione finora
- Periodic Tales: A Cultural History of the Elements, from Arsenic to ZincDa EverandPeriodic Tales: A Cultural History of the Elements, from Arsenic to ZincValutazione: 3.5 su 5 stelle3.5/5 (137)
- It's Elemental: The Hidden Chemistry in EverythingDa EverandIt's Elemental: The Hidden Chemistry in EverythingValutazione: 4 su 5 stelle4/5 (10)
- The Nature of Drugs Vol. 1: History, Pharmacology, and Social ImpactDa EverandThe Nature of Drugs Vol. 1: History, Pharmacology, and Social ImpactValutazione: 5 su 5 stelle5/5 (5)
- ICH Quality Guidelines: An Implementation GuideDa EverandICH Quality Guidelines: An Implementation GuideAndrew TeasdaleNessuna valutazione finora
- The Regenerative Grower's Guide to Garden Amendments: Using Locally Sourced Materials to Make Mineral and Biological Extracts and FermentsDa EverandThe Regenerative Grower's Guide to Garden Amendments: Using Locally Sourced Materials to Make Mineral and Biological Extracts and FermentsValutazione: 5 su 5 stelle5/5 (3)
- Chemistry for Breakfast: The Amazing Science of Everyday LifeDa EverandChemistry for Breakfast: The Amazing Science of Everyday LifeValutazione: 4.5 su 5 stelle4.5/5 (14)
- Chemistry: a QuickStudy Laminated Reference GuideDa EverandChemistry: a QuickStudy Laminated Reference GuideValutazione: 5 su 5 stelle5/5 (1)
- The Nature of Drugs Vol. 1: History, Pharmacology, and Social ImpactDa EverandThe Nature of Drugs Vol. 1: History, Pharmacology, and Social ImpactValutazione: 5 su 5 stelle5/5 (1)
- Handbook of Formulating Dermal Applications: A Definitive Practical GuideDa EverandHandbook of Formulating Dermal Applications: A Definitive Practical GuideNessuna valutazione finora
- Monkeys, Myths, and Molecules: Separating Fact from Fiction, and the Science of Everyday LifeDa EverandMonkeys, Myths, and Molecules: Separating Fact from Fiction, and the Science of Everyday LifeValutazione: 4 su 5 stelle4/5 (1)
- The Production of Volatile Oils and Perfumery Plants in the United StatesDa EverandThe Production of Volatile Oils and Perfumery Plants in the United StatesNessuna valutazione finora
- Practical High-Performance Liquid ChromatographyDa EverandPractical High-Performance Liquid ChromatographyNessuna valutazione finora
- Is That a Fact?: Frauds, Quacks, and the Real Science of Everyday LifeDa EverandIs That a Fact?: Frauds, Quacks, and the Real Science of Everyday LifeValutazione: 5 su 5 stelle5/5 (4)
- AP Chemistry Flashcards, Fourth Edition: Up-to-Date Review and PracticeDa EverandAP Chemistry Flashcards, Fourth Edition: Up-to-Date Review and PracticeNessuna valutazione finora
- The Nature of Drugs Vol. 2: History, Pharmacology, and Social ImpactDa EverandThe Nature of Drugs Vol. 2: History, Pharmacology, and Social ImpactNessuna valutazione finora
- Chemistry for Breakfast: The Amazing Science of Everyday LifeDa EverandChemistry for Breakfast: The Amazing Science of Everyday LifeValutazione: 4.5 su 5 stelle4.5/5 (90)