Documenti di Didattica
Documenti di Professioni
Documenti di Cultura
Kinetic Reactor Design Chapter 3 Lecture 4
Caricato da
Khairul Imran AzmanDescrizione originale:
Copyright
Formati disponibili
Condividi questo documento
Condividi o incorpora il documento
Hai trovato utile questo documento?
Questo contenuto è inappropriato?
Segnala questo documentoCopyright:
Formati disponibili
Kinetic Reactor Design Chapter 3 Lecture 4
Caricato da
Khairul Imran AzmanCopyright:
Formati disponibili
CHAPTER 3
Rate Laws and Stoichiometry
Lecture 4
Tell me and I'll forget; show me and I may remember;
involve me and I'll understand.
Chinese Proverb
CCB 3043 Kinetics and Reactor Design
Topics
Lecture 1: Basic definitions - Part 1
Types of reaction and relative rate of reaction
Lecture 2: Basic definitions - Part 2
Rate constant and activation energy
Lecture 3: Stoichiometric tables
Batch system
Consideration for constant volume system
Lecture 4:
Stoichiometric tables
Flow systems
Consideration for changing volume systems
CCB 3043 Kinetics and Reactor Design
Flow systems
D
a
d
C
a
c
B
a
b
A + +
F
A0
F
B0
F
C0
F
D0
F
I0
F
A
F
B
F
C
F
D
F
I
*PFR or CSTR
F
A0
F
B0
F
C0
F
D0
F
I0
F
A
F
B
F
C
F
D
F
I
D
a
d
C
a
c
B
a
b
A + +
CCB 3043 Kinetics and Reactor Design
Batch systems
at t = 0
N
A0
N
B0
N
C0
N
D0
N
I0
at t = t
N
A
N
B
N
C
N
D
N
I
CCB 3043 Kinetics and Reactor Design
Species Sym Initial Change Remaining
A A F
A0
- F
A0
X F
A
= F
A0
(1-X)
B B F
B0
= F
A0
O
B
-(b/a) F
A0
X F
B
= F
A0
[O
B
-(b/a)X]
C C F
C0
= F
A0
O
C
(c/a)F
A0
X F
C
= F
A0
[O
C
+(c/a)X]
D D F
D0
= F
A0
O
D
(d/a)F
A0
X F
D
= F
A0
[O
D
+(d/a)X]
Inert I F
I0
= F
A0
O
I
- F
I0
= F
A0
O
I
Total
F
T0
F
T
= F
T0
+ [d/a + c/a
b/a a]F
A0
X
F
T
= F
T0
+ oF
A0
X
Flow systems
CCB 3043 Kinetics and Reactor Design
Where:
Flow systems
v
F
C
1
a
b
a
c
a
d
y
y
C
C
F
F
i
i
0 A
0 i
A0
i0
A0
i0
i
=
+ =
= = =
Valid for constant
volume system
CCB 3043 Kinetics and Reactor Design
Flow system
Note that:
For liquid phase system (no phase change,
constant volume);
For gas phase system, the above is not true;
need to account for volume expansion
( ) X C C
v v
A A
=
=
1
0
0
CCB 3043 Kinetics and Reactor Design
Volume change with reaction (gas
phase): Batch Reactors
Occur when a system does not have an equal no of product
and reactant, (usually involve gas phase system) e.g.;
N
2
+ 3H
2
2NH
3
Combustion reaction
Need to take into account the expansion factor, c
X N
N N
T
T T
0
0
X; conversion any at or
reactor the to fed moles of number total
conversion complete for moles of number in total change
=
=
c
c
o c
0
0
0
1
A
T
A
y
N
N
a
b
a
c
a
d
=
|
.
|
\
|
+
=
CCB 3043 Kinetics and Reactor Design
CONSTANT VOLUME
BATCH FLOW
v
F
C
1
a
b
a
c
a
d
y
y
C
C
F
F
i
i
0 A
0 i
A0
i0
A0
i0
i
=
+ =
= = =
V
N
C
1
a
b
a
c
a
d
y
y
C
C
N
N
i
i
0 A
0 i
A0
i0
A0
i0
i
=
+ =
= = =
Reactor
Volume
Volumetric
flowrate
CCB 3043 Kinetics and Reactor Design
Volume change with reaction (gas
phase): Batch Reactors
Gas system; (Equation of state):
RT ZN PV
T
=
P = Total pressure, 1 atm
V = Volume, dm
3
Z = Compressibility factor
N
T
= Total number of moles
R = Gas constant,
0.08206 dm
3
.atm/mol.K
T = Temperature, K
CCB 3043 Kinetics and Reactor Design
At t = 0:
0 0 0 0 0
RT N Z V P
T
=
Volume change with reaction (gas
phase): Batch Reactors
RT ZN PV
T
=
0 0 0
0
0
0 0 0 0 0
T
T
T
T
N
N
Z
Z
T
T
P
P
V V
RT N Z V P
RT ZN PV
|
|
.
|
\
|
|
.
|
\
|
=
=
=
: rearrange
Divide
(1)/(2):
Remember? c=(N
T
-N
T0
)/N
T0
X
And for gas phase system, Z
0
~Z
( ) X
T
T
P
P
V V c +
|
.
|
\
|
= 1
0
0
0
Volume of gas for variable
volume batch reaction
CCB 3043 Kinetics and Reactor Design
Volume change with reaction (gas
phase): Batch Reactors
At any conversion;
o c
c
0
0
0
0
0
1
A
T
A
T
T T
y
N
N
a
b
a
c
a
d
X N
N N
=
|
.
|
\
|
+
=
=
X N
a
b
a
c
a
d
N N
From
A T T 0 0
1
|
.
|
\
|
+ =
: Table tric Stoichiome
F
T
= F
T0
+ [d/a + c/a b/a a]F
A0
X
OR
N
T
= N
T0
+ [d/a + c/a b/a a]N
A0
X
CCB 3043 Kinetics and Reactor Design
Volume change with reaction: BATCH
For batch system:
( )
0
0
0
T
T
X 1
P
P
V V c + |
.
|
\
|
=
o c
c
0
0
0
1
A
T
A
y
N
N
a
b
a
c
a
d
=
|
.
|
\
|
+
=
CCB 3043 Kinetics and Reactor Design
Volume change with reaction:
FLOW SYSTEM
|
.
|
\
|
|
|
.
|
\
|
=
T
T
P
P
F
F
C C
T
j
T j
0
0
0
D
a
d
C
a
c
B
a
b
A + +
|
|
.
|
\
|
|
|
.
|
\
|
=
0
0
0
0
T
T
P
P
F
F
T
T
u u
( )
|
.
|
\
|
|
|
.
|
\
|
+
+ O
=
T
T
P
P
X
X v C
C
j A
j
0
0
0
1 c
Can be used for membrane reactors
and multiple reactions
F
T
= sum of molar
flowrate from each
species
C
T
= sum of
concentration from
each specied
v
j
= stoichiometric
coefficient
v
A
= -1, v
B
= -b/a, v
C
= c/a, v
D
= d/a
Refer to
pg 113
( )
|
|
.
|
\
|
+ =
0
0
0
1
T
T
P
P
X c u u
CCB 3043 Kinetics and Reactor Design
VOLUME CHANGE
( ) X
T
T
P
P
V V c +
|
.
|
\
|
= 1
0
0
0
( )
|
|
.
|
\
|
+ =
0
0
0
1
T
T
P
P
X c u u
BATCH FLOW
P constant, T constant (isothermal):
( ) X V V c + = 1
0
( ) X c u u + = 1
0
BATCH FLOW
CCB 3043 Kinetics and Reactor Design
Volume change with reaction:
FLOW SYSTEM
Try to derive
those equations!!
(Hint: Use the
I deal gas law)
Refer to page
111(Fogler)
Read page 111 to
115, to make you
understand better
Where do we get those equations from???
CCB 3043 Kinetics and Reactor Design
Important notes for Rate Law and
Stoichiometry
IMPORTANT TABLES:
Table 3-3 (p101, Fogler): Stoichiometric
table for a BATCH system
Table 3-4 (p107, Fogler): Stoichiometric
table for a FLOW system
Table 3-5 (p114, Fogler): Concentrations in
a varible-volume gas FLOW system
CCB 3043 Kinetics and Reactor Design
Example 3.5: Determining the concentration of
species involved in a gas phase reaction
Consider the oxidation of SO
2
below. A mixture of 28%
SO
2
and 72% air is charged to a flow reactor in which
SO
2
is oxidized. Given that the total pressure of the
system is 1485 kPa and the temperature is constant at
227
o
C. Set up the stoichiometric table for the system and
evaluate the concentration of species presents in terms of
conversion. Plot the concentration profile of the species
with respect to conversion
2SO
2
+ O
2
2SO
3
CCB 3043 Kinetics and Reactor Design
Example 3.6: Calculating the equilibrium conversion
The reversible gas-phase decomposition of nitrogen tetroxide, N
2
O
4,
,
is carried out at constant temperature. The feed consists of pure N
2
O
4
at 340 K and 202.6 kPa (2 atm). The concentration equilibrium
constant K
c
, at 340 K is 0.1 mol/dm
3
.
a) Express the equilibrium conversion of N
2
O
4
in a constant-
volume batch reactor, in term of C
A0
and K
C
.
b) Express the equilibrium conversion of N
2
O
4
in a flow reactor, in
term of C
A0
and K
C
c) Assuming the reaction is elementary, express the rate of reaction
solely as a function of conversion for a constant-volume batch
and a flow system.
d) If k
A
is 0.5
min-1
and feed rate is 3 mol/min, determine the CSTR
volume necessary to achieve 80% of the equilibrium conversion
N
2
O
4
2NO
2
CCB 3043 Kinetics and Reactor Design
Checklist..
Mole balance
Rate Law
Stoichiometry
Do you have all these building blocks?
CCB 3043 Kinetics and Reactor Design
Potrebbero piacerti anche
- Revision QuestionsDocumento12 pagineRevision QuestionsLiew Wen Xuan0% (2)
- Stoichiometric TableDocumento22 pagineStoichiometric TableMark Antony LevineNessuna valutazione finora
- Chapter 3 Rev1 Rate Laws & StoichiometryDocumento35 pagineChapter 3 Rev1 Rate Laws & StoichiometryHakashiMirudoNessuna valutazione finora
- (P01, C01, C02, C2, C3) : Confidential EH/JUN 2014/CHE584/594Documento11 pagine(P01, C01, C02, C2, C3) : Confidential EH/JUN 2014/CHE584/594Addison JuttieNessuna valutazione finora
- 4310 Combustion Introduction Lecture 14Documento15 pagine4310 Combustion Introduction Lecture 14lorenzofrancescaNessuna valutazione finora
- Steady-State Non-Isothermal Reactor Design CompleteDocumento130 pagineSteady-State Non-Isothermal Reactor Design CompleteYesid Tapiero MartínezNessuna valutazione finora
- 08 Multiple ReactionsDocumento17 pagine08 Multiple ReactionsFikrie MuhdNessuna valutazione finora
- Chapter 3 Rates Law and StoichiometryDocumento60 pagineChapter 3 Rates Law and StoichiometryMalek Marry AnneNessuna valutazione finora
- Heat and Mass Transfer PropertiesDocumento63 pagineHeat and Mass Transfer Propertieskaushita banerjeeNessuna valutazione finora
- Chapter 9 ChE 126Documento94 pagineChapter 9 ChE 126Roque LeccioNessuna valutazione finora
- Duhok Polytechnique University-Petrochemical Department 2018 / 2019 Catalysis DR Farhad M. Ali 2018/2019Documento6 pagineDuhok Polytechnique University-Petrochemical Department 2018 / 2019 Catalysis DR Farhad M. Ali 2018/2019MUHAMMAD AKRAMNessuna valutazione finora
- Tutorial For Chapter 23Documento9 pagineTutorial For Chapter 23Thurgah VshinyNessuna valutazione finora
- hw3 - Che324Documento3 paginehw3 - Che324Ahmed Ali0% (1)
- ChE 471 EXAM 1 2005Documento2 pagineChE 471 EXAM 1 2005HungDoNessuna valutazione finora
- Multiple Reaction System-1Documento35 pagineMultiple Reaction System-1ANZWAYNENessuna valutazione finora
- CHE3044F, 2013: Reactor Design 1: TUTORIAL 6Documento4 pagineCHE3044F, 2013: Reactor Design 1: TUTORIAL 6nmhatityeNessuna valutazione finora
- Distillation Chapter 2Documento35 pagineDistillation Chapter 2fatien zakaria100% (1)
- Tutorial 7 - Kinetics - SOLUTIONSDocumento5 pagineTutorial 7 - Kinetics - SOLUTIONSJustin Chuong NguyenNessuna valutazione finora
- 10 PDFDocumento23 pagine10 PDFTysir SarhanNessuna valutazione finora
- Stage & Continuous Gas-Liquid Separation Processes: Chapter ThreeDocumento116 pagineStage & Continuous Gas-Liquid Separation Processes: Chapter ThreeDoni RopawandiNessuna valutazione finora
- Tutorial 1 SolutionDocumento6 pagineTutorial 1 SolutionMihir Kumar MechNessuna valutazione finora
- CREII-Module-I - Lecture 4 PDFDocumento34 pagineCREII-Module-I - Lecture 4 PDFshubhamNessuna valutazione finora
- Solution: For A First-Order Reaction, The Following Rate Coefficients Were FoundDocumento16 pagineSolution: For A First-Order Reaction, The Following Rate Coefficients Were FoundDeepak SharmaNessuna valutazione finora
- Set3ans 10Documento5 pagineSet3ans 10amalinaishahNessuna valutazione finora
- Lecture 9 - Collection and Analysis of Rate DataDocumento13 pagineLecture 9 - Collection and Analysis of Rate DataSabrina AzharNessuna valutazione finora
- CHE244 Project GuidelinesDocumento5 pagineCHE244 Project GuidelinesEiman UzmiNessuna valutazione finora
- Lecture 6Documento48 pagineLecture 6tkjingNessuna valutazione finora
- Semester-6 3360503 CRE MCQ KRD PDFDocumento9 pagineSemester-6 3360503 CRE MCQ KRD PDFDhruv RanaNessuna valutazione finora
- Reaction KineticsDocumento37 pagineReaction KineticsNurshuhada NordinNessuna valutazione finora
- Set8ans 10Documento5 pagineSet8ans 10Agustina Evania DewiNessuna valutazione finora
- 8 3 Packed Bed ReactorsDocumento20 pagine8 3 Packed Bed ReactorsridhajamelNessuna valutazione finora
- Ugpa3033 Process Optimization and Simulation Tutorial 4Documento1 paginaUgpa3033 Process Optimization and Simulation Tutorial 4Tko Kai OnnNessuna valutazione finora
- Developing and Using Stio Tables NotesDocumento27 pagineDeveloping and Using Stio Tables NotesThabangNessuna valutazione finora
- UO 4 Solid Handling UnitDocumento17 pagineUO 4 Solid Handling UnitNoor FadzleenaNessuna valutazione finora
- CHE411 Fall 2010-Chemical Reaction Engineeirng-Ahmed A AbdalaDocumento206 pagineCHE411 Fall 2010-Chemical Reaction Engineeirng-Ahmed A AbdalaYayan IndrayaniNessuna valutazione finora
- Chapter 3 ConSol PPT by E.cusslerDocumento39 pagineChapter 3 ConSol PPT by E.cusslerheena_scottNessuna valutazione finora
- Gate 2006 PDFDocumento21 pagineGate 2006 PDFVammsy Manikanta SaiNessuna valutazione finora
- CDB 4313 Heat Integration - Basic Heat IntegrationDocumento25 pagineCDB 4313 Heat Integration - Basic Heat Integrationharvin raoNessuna valutazione finora
- Professional Reference Shelf: Sothermal Eactor EsignDocumento5 pagineProfessional Reference Shelf: Sothermal Eactor EsignSourodip GhoshdastidarNessuna valutazione finora
- Reactor Design and Cost EstimationDocumento21 pagineReactor Design and Cost EstimationEkeh Victor Chibueze0% (1)
- Properties of Water PDFDocumento36 pagineProperties of Water PDFanon_348923763Nessuna valutazione finora
- 13 Reaction Kinetics (S)Documento32 pagine13 Reaction Kinetics (S)Mr TanNessuna valutazione finora
- Recycle. Process EngineeringDocumento20 pagineRecycle. Process EngineeringDarel WilliamsNessuna valutazione finora
- Transport Phenomena AssignmentDocumento3 pagineTransport Phenomena AssignmentKushal KayalNessuna valutazione finora
- 8.3 - Packed-Bed ReactorsDocumento20 pagine8.3 - Packed-Bed ReactorscarolinacmleiteNessuna valutazione finora
- Kmu346-22 HW IDocumento1 paginaKmu346-22 HW IDevendraa MuniandyNessuna valutazione finora
- Mass Transfer QuizzesDocumento10 pagineMass Transfer QuizzesNate JamesNessuna valutazione finora
- Adsorption & Ion Exchange ProblesmDocumento10 pagineAdsorption & Ion Exchange ProblesmDeepak KanjwaniNessuna valutazione finora
- Notes5 Simple ReactorsDocumento40 pagineNotes5 Simple ReactorsstorkjjNessuna valutazione finora
- SITI SOLEHAH BINTI OMAR (2015834112) /EH224 7A1 Heuristics of ReactorDocumento2 pagineSITI SOLEHAH BINTI OMAR (2015834112) /EH224 7A1 Heuristics of ReactorSolehah OmarNessuna valutazione finora
- 1.multiple ReactionsDocumento58 pagine1.multiple ReactionsDianah NajeebNessuna valutazione finora
- ChE426 Final Exam 2005Documento2 pagineChE426 Final Exam 2005احمد الدلالNessuna valutazione finora
- Advanced Reaction - Lecture 1Documento39 pagineAdvanced Reaction - Lecture 1nahomNessuna valutazione finora
- Tutorial Questions On Heat Ex ChangersDocumento3 pagineTutorial Questions On Heat Ex ChangersPuneet Garg100% (1)
- Chapter 6 Rev1 Multiple ReactionDocumento37 pagineChapter 6 Rev1 Multiple ReactionHakashiMirudoNessuna valutazione finora
- Ex0 Questions SolutionsDocumento7 pagineEx0 Questions SolutionsBiniyam haileNessuna valutazione finora
- Practical Chemical Thermodynamics for GeoscientistsDa EverandPractical Chemical Thermodynamics for GeoscientistsNessuna valutazione finora
- High Pressure Phase Behaviour of Multicomponent Fluid MixturesDa EverandHigh Pressure Phase Behaviour of Multicomponent Fluid MixturesNessuna valutazione finora
- Essay Rough Draft 19Documento9 pagineEssay Rough Draft 19api-549246767Nessuna valutazione finora
- Appendix - Pcmc2Documento8 pagineAppendix - Pcmc2Siva PNessuna valutazione finora
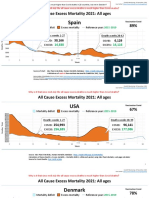
- Countries EXCESS DEATHS All Ages - 15nov2021Documento21 pagineCountries EXCESS DEATHS All Ages - 15nov2021robaksNessuna valutazione finora
- Chapter 1 ClassnotesDocumento35 pagineChapter 1 ClassnotesAllison CasoNessuna valutazione finora
- UTP Student Industrial ReportDocumento50 pagineUTP Student Industrial ReportAnwar HalimNessuna valutazione finora
- Syllabus DresserDocumento2 pagineSyllabus DresserVikash Aggarwal50% (2)
- Snapdragon 435 Processor Product Brief PDFDocumento2 pagineSnapdragon 435 Processor Product Brief PDFrichardtao89Nessuna valutazione finora
- Role of Losses in Design of DC Cable For Solar PV ApplicationsDocumento5 pagineRole of Losses in Design of DC Cable For Solar PV ApplicationsMaulidia HidayahNessuna valutazione finora
- Mark Garside Resume May 2014Documento3 pagineMark Garside Resume May 2014api-199955558Nessuna valutazione finora
- Presentation 11Documento14 paginePresentation 11stellabrown535Nessuna valutazione finora
- Developing Paragraphs: Test Section - Writing Task 2Documento11 pagineDeveloping Paragraphs: Test Section - Writing Task 2Cengizhan AkdağNessuna valutazione finora
- V737 OverheadDocumento50 pagineV737 OverheadnewahNessuna valutazione finora
- Dialogue Au Restaurant, Clients Et ServeurDocumento9 pagineDialogue Au Restaurant, Clients Et ServeurbanuNessuna valutazione finora
- Nama: Yossi Tiara Pratiwi Kelas: X Mis 1 Mata Pelajaran: Bahasa InggrisDocumento2 pagineNama: Yossi Tiara Pratiwi Kelas: X Mis 1 Mata Pelajaran: Bahasa InggrisOrionj jrNessuna valutazione finora
- PyhookDocumento23 paginePyhooktuan tuanNessuna valutazione finora
- Theory GraphDocumento23 pagineTheory GraphArthur CarabioNessuna valutazione finora
- Dynamics of Machinery PDFDocumento18 pagineDynamics of Machinery PDFThomas VictorNessuna valutazione finora
- Quantitative Methods For Economics and Business Lecture N. 5Documento20 pagineQuantitative Methods For Economics and Business Lecture N. 5ghassen msakenNessuna valutazione finora
- Gemini Dollar WhitepaperDocumento7 pagineGemini Dollar WhitepaperdazeeeNessuna valutazione finora
- ISBN Safe Work Method Statements 2022 03Documento8 pagineISBN Safe Work Method Statements 2022 03Tamo Kim ChowNessuna valutazione finora
- Unit 7: Anthropology: Q2e Listening & Speaking 4: Audio ScriptDocumento6 pagineUnit 7: Anthropology: Q2e Listening & Speaking 4: Audio ScriptĐại học Bạc Liêu Truyền thông100% (1)
- DNA ReplicationDocumento19 pagineDNA ReplicationLouis HilarioNessuna valutazione finora
- De Thi Hoc Ki 1 Mon Tieng Anh Lop 5 Co File NgheDocumento10 pagineDe Thi Hoc Ki 1 Mon Tieng Anh Lop 5 Co File Nghetuyen truongNessuna valutazione finora
- Assessment of Students' Oral Communication in English ClassDocumento10 pagineAssessment of Students' Oral Communication in English ClassKeebeek S ArbasNessuna valutazione finora
- Read While Being Blind.. Braille's Alphabet: Be Aware and Active !Documento3 pagineRead While Being Blind.. Braille's Alphabet: Be Aware and Active !bitermanNessuna valutazione finora
- Functional DesignDocumento17 pagineFunctional DesignRajivSharmaNessuna valutazione finora
- Study 107 - The Doctrine of Salvation - Part 8Documento2 pagineStudy 107 - The Doctrine of Salvation - Part 8Jason MyersNessuna valutazione finora
- in Strategic Management What Are The Problems With Maintaining A High Inventory As Experienced Previously With Apple?Documento5 paginein Strategic Management What Are The Problems With Maintaining A High Inventory As Experienced Previously With Apple?Priyanka MurthyNessuna valutazione finora
- Technology Management 1Documento38 pagineTechnology Management 1Anu NileshNessuna valutazione finora
- Waterstop TechnologyDocumento69 pagineWaterstop TechnologygertjaniNessuna valutazione finora