Documenti di Didattica
Documenti di Professioni
Documenti di Cultura
Interpretando RX Pediatrico
Caricato da
suzukishareDescrizione originale:
Titolo originale
Copyright
Formati disponibili
Condividi questo documento
Condividi o incorpora il documento
Hai trovato utile questo documento?
Questo contenuto è inappropriato?
Segnala questo documentoCopyright:
Formati disponibili
Interpretando RX Pediatrico
Caricato da
suzukishareCopyright:
Formati disponibili
PAEDIATRIC RESPIRATORY REVIEWS (2000) 1, 4150 doi:10.1053/prrv.2000.0018, available online at http://www.idealibrary.
com on
Interpretation of the paediatric chest X-ray
R. Arthur
The X-ray and Ultrasound Department,The Clarendon Wing, The General Infirmary at Leeds, Belmont Grove, LS2 9NS, UK
KEYWORDS chest X-ray, pattern recognition, radiographical technique, pulmonary infiltrates, obstructive emphysema
Summary Interpretation of the paediatric chest X-ray may appear intimidating at first but knowledge of a few basic rules and an understanding of how the radiographic appearance may be influenced by age and technique will help the clinician arrive at the correct diagnosis in many cases. A structured and logical approach to X-ray interpretation is described, drawing attention to the abnormal signs that should be sought. A limited differential diagnosis is given for some of the more frequent radiological abnormalities seen in children. 2000 Harcourt Publishers Ltd
INTRODUCTION
The chest X-ray is the most frequently performed radiographic examination in children and correct interpretation can be crucial to reaching the correct diagnosis and avoiding inappropriate treatment due to failure to appreciate normal variation and the influence of technical factors on the radiographic appearance. A methodical approach as suggested below is important in the evaluation of a chest X-ray to ensure that important clues to the diagnosis are not overlooked: i) Check the patients name, date of examination and side marking. ii) Note the projection (supine or erect, AP or PA), phase of respiration and the presence of any rotation. Identify any artefactual shadows. iii) Identify abnormal radiological signs by systematically reviewing all regions including: the trachea, carina and major bronchi, the mediastinal outlines and hilar regions, cardiac size and contour, pulmonary vascularity, the size and translucency of the lungs, the position of the major fissures, the clarity and height of the diaphragms, the costo-phrenic angles, the soft tissues and the thoracic skeleton. iv) Suggest the most likely diagnosis or list possible differential diagnoses based on the observed radiological signs and clinical features.
Correspondence to: Tel: 0113 392 3784; Fax: 0113 392 3775; E-mail: rarthu@ulth.northy.nhs.uk 15260550/00/010041 + 10 $35.00/0
INFLUENCE OF TECHNIQUE AND AGE ON RADIOLOGICAL INTERPRETATION
Projection
For most clinical indications, a single frontal projection of the chest is appropriate, but a lateral view may be helpful to demonstrate an abnormality in the mediastinum, at the lung bases or to localize a lesion identified on the frontal projection. The supine antero-posterior (AP) projection is used for most babies whereas the toddler is generally examined in an erect AP projection until the child can cooperate sufficiently. The heart size and mediastinal width will be exaggerated by both the supine position and the AP projection. Pleural fluid and pneumothoraces are more difficult to detect in the supine position and a horizontal beam projection will be necessary to demonstrate air fluid levels e.g., in a hydropneumothorax following trauma.
Phase of respiration
The diaphragms are projected over the 5th to 7th anterior rib ends in a well inspired examination. In the young infant an expiratory phase film will exaggerate the heart size and broncho-vascular markings and misinterpretation may lead to an erroneous diagnosis of cardiac failure or bronchopneumonia and the high position of the diaphragms will obscure abnormalities at the lung bases (Fig. 1).
2000 Harcourt Publishers Ltd
42
R. ARTHUR
Figure 1 a) Technically poor expiratory phase chest X-ray with prominent broncho-vascular markings and an apparently large heart; b) repeat examination. Note the normal heart size; the consolidation at the left base is now more clearly visualized on this well inspired X-ray (arrows).
Positioning
Rotation is the most common cause of inequality in the translucency of the two lungs and needs to be differentiated from increased transradiency resulting from other causes particularly air trapping. Rotation can be assessed by checking the symmetry of the ribs and the distance of the medial borders of the clavicles from the spinous process of the vertebrae.
Artefacts
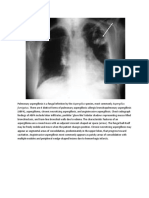
X-rays taken on the wards are particularly prone to artefacts. A round translucency caused by the hole in the perspex of the incubator, and skin folds between the baby and the cassette which mimic a pneumothorax are well recognized. In older children, hair either plaited or dressed with ornaments and lotions may cause a variety of artefacts projected over the upper lobes and mediastinum (Fig. 2).
can be recognized by its characteristic sail shape or wavy margins resulting from the interdigitation of the soft thymic tissue in the horizontal tissue or intercostal spaces (Fig. 3). The thymus gradually becomes less evident between the ages of 28 years after which it cannot be visualized on the frontal chest X-ray. At times the normal thymus can appear very large, and may need to be differentiated from a mediastinal mass or an area of pulmonary consolidation. A lateral decubitis view of the chest may accentuate the scalloped appearance of the thymic outline, and help to clarify the nature of any anterior mediastinal shadow. Additional imaging using ultrasound scanning and, in exceptional cases, CT/MRI scanning may also be helpful.
Age
The gradual change in the normal appearance of the chest X-ray from infancy to adulthood must be appreciated. In a baby, the chest configuration is more triangular shaped and relatively deeper in the AP diameter. Air bronchograms frequently seen projected through the cardiac shadow in the neonate and young infant may not be significant but should be considered pathological when seen more peripherally. The anterior aspects of the diaphragms are higher, the costo-phrenic angles are relatively shallower in the infant, and the lower zones may be obscured, particularly in a poorly penetrated examination.
The thymus gland
The thymus gland gives rise to a prominent anterior mediastinal shadow in infancy which is quite variable in size and
Figure 2 Linear translucencies due to shadows of dressed hair adjacent to the trachea mimicking mediastinal air (arrowheads).
INTERPRETATION OF THE PAEDIATRIC CHEST X-RAY
43
Figure 3 Sail-shaped outline of the thymus gland demonstrating wavy right lateral border (arrows). Figure 4 Large anterior/middle mediastinal mass obscuring the arch of the aorta.The endotracheal and nasogastric tubes are displaced to the left (arrow heads) by the mass which is also causing loss of aeration of the right upper lobe (arrow). Normal thymus gland outline noted on the left.
INTERPRETATION OF ABNORMAL RADIOLOGICAL SIGNS
Pattern recognition forms the cornerstone of successful X-ray interpretation. Although some X-ray findings are pathognomonic for specific conditions, in most cases the diagnostic process depends upon correlating the X-ray abnormalities with the age of the child, the clinical history and examination, and the results of any previous X-rays and laboratory investigations. In some situations the diagnosis may only be confirmed on follow-up, by monitoring response to therapy or following biopsy.
SYSTEMATIC REVIEW OF THE CHEST X-RAY
Trachea and main bronchi and hilar regions
In the infant the trachea is quite mobile and may buckle anteriorly and to the right on expiration. However below the thoracic inlet the intrathoracic trachea should always appear straight on a lateral chest X-ray, even in expiration, and any anterior or posterior displacement of the intrathoracic trachea should raise suspicion of a mediastinal mass. A penetrated view may help to improve visualization of the airway in the AP projection particularly to demonstrate the carina and the major bronchi. A lateral view from the larynx to the carina is advisable where there is a history suggestive of upper airway obstruction. Subglottic narrowing of the trachea is most commonly seen in acute laryngo-tracheo-bronchitis (i.e., croup), but extrinsic compression by a vascular ring or an impacted
oesophageal foreign body, and narrowing due to a congenital tracheal stenosis or an intraluminal tumour must also be considered in the differential diagnosis, particularly when the trachea is narrowed at or below the level of the thoracic inlet. Widening of the trachea may be seen in children following prolonged intubation, or those with chronic cough e.g., cystic fibrosis and in Mounier-Kuhn syndrome, i.e., congenital tracheomegaly. Enlargement of the hilar shadows may be due to hilar gland lymphadenopathy, most commonly related to viral pneumonia or chronic infection e.g., in cystic fibrosis. When markedly enlarged lymph node infiltration is noted, tuberculosis and sarcoidosis should be considered. A lateral view of the hilum is useful to confirm the nature of the hilar enlargement since other pathology e.g., pulmonary consolidation may be projected over the hilum on a frontal film and mimic hilar lymphadenopathy.
The superior mediastinum
The superior mediastinal outline should be assessed for both size and shape. The characteristics of any abnormal shadow should be noted, particularly whether the widening is bilateral or unilateral. If a naso-gastric tube is in-situ its position should be noted as an indicator of the line of the oesophagus (Fig. 4). Classically the superior mediastinum has been divided into the anterior, middle and posterior compartments, with the great vessels, trachea, oesophagus
44
R. ARTHUR
Figure 7 Translucent right hemithorax is due to air trapping resulting from an inhaled peanut in the right main bronchus. Note decreased pulmonary vascular markings on the right. Figure 5 Peripheral pruning of the pulmonary artery branches (arrow) indicating pulmonary arterial hypertension secondary to multiple pulmonary emboli precipitated by a previous ventriculo-atrial shunt, which was subsequently replaced by a ventriculo-peritoneal shunt (arrowheads).
and paratracheal lymph nodes comprising the middle compartment. An anterior mediastinal mass is suspected when the trachea is deviated posteriorly e.g., in lymphoma or with a terato-dermoid tumour. Loss of visualization of the aortic knuckle indicates that the mass lies adjacent to the aortic arch and arises from the anterior or middle mediastinum. Lateral deviation of the trachea or separation of the trachea and oesophagus (i.e., position of NG tube) point to a middle mediastinal mass e.g., bronchogenic cyst. Masses arising in the posterior mediastinum e.g., a neurogenic tumour, may show areas of calcification, result in splaying or even destruction of the posterior rib ends. Localization of any radiographic abnormality into one of these compartments will help to focus the anatomical origin and nature of the abnormality.1 Acute infection or steroid therapy may result in transient thymic atrophy but if the superior mediastinal outline is persistently narrow the possibility of an absent thymus gland e.g., in Di George syndrome, or congenital heart disease should be considered.
The heart and great vessels
The mediastinum and cardiac contour are relatively large in the infant and the transverse diameter of the normal heart may approach 60% of the thoracic transverse diameter. The atrial and visceral situs should be established by inspection of the bronchial anatomy and position of the stomach bubble and the side of the ascending and descending aorta should be noted. A right sided aortic arch is commonly associated with congenital heart disease and vascular rings are seen more frequently in association with a right sided aortic arch.2 Any increase in cardiac size should prompt a close inspection of the lungs to assess pulmonary vascularity. The branches of the
Figure 6 Low flat diaphragms and hyperinflated lungs in cystic fibrosis. Note the prominent hilar shadows and multiple ring shadows (arrows) in the right upper and left lower lobes.
INTERPRETATION OF THE PAEDIATRIC CHEST X-RAY
45
pulmonary arteries should not be visible in the peripheral third of the lung and if seen suggest increased pulmonary blood flow. Non-visualization of pulmonary vessels more centrally suggests reduced pulmonary blood flow. Pulmonary arterial hypertension is indicated by the presence of peripheral pruning of the pulmonary arteries i.e., dilatation of the proximal arteries with a distinct reduction in calibre of the central and peripheral pulmonary arteries (Fig. 5). There are many causes of cardiomegaly including congenital heart disease, cardiomyopathy, congestive cardiac failure and pericardial effusion. Whilst abnormalities in cardiac contour may be useful to indicate which cardiac chambers may be enlarged the appearances are often non specific and full assessment by echocardiography is recommended.
Unequal translucency of the lungs always merits serious consideration. Patient rotation, probably the commonest cause, should be excluded by obtaining a repeat straight radiograph, to avoid overlooking unilateral obstructive emphysema (Fig. 7). It may be difficult to determine which side is abnormal when the two lungs are of different densities, but consideration of the following points, as described by Swischuk3 should help identify the abnormal side. i) Pulmonary vascularity The side with decreased vascularity is abnormal The side with increased or normal vascularity is usually normal ii) Variation in appearance between inspiratory and expiratory films The side which changes least on expiration is usually abnormal iii) The size of the hemithorax A small completely opaque hemithorax is abnormal Both compensatory and obstructive emphysema may be associated with a large translucent hemithorax, with the contra-lateral lung showing increased opacity (Fig. 8). Increased translucency due to obstructive emphysema e.g., from an inhaled foreign body or congenital lobar emphysema is usually associated with attenuation of the pulmonary vascularity and the disparity between the two sides will become more pronounced on an expiratory phase film with the abnormal side remaining over-inflated. On the other hand compensatory emphysema due to collapse or hypoplasia of the contra-lateral lung will usually
Lungs/pleural cavities: patterns of disease
Abnormalities in the lungs and pleural cavities are often indicated by focal or generalized areas of either increased or decreased translucency in the lungs, ring shadows and pulmonary nodules.
Increased translucency
Generalized increased translucency of the thorax in association with low flattened diaphragms may be seen in a healthy child having made a large inspiratory effort but is commonly associated air trapping e.g., in asthma, bronchiolitis and cystic fibrosis (Fig. 6). Upper airway obstruction due to tracheal obstruction e.g., a vascular ring or tracheal foreign body should also be considered.
A B
Figure 8 a) Congenital lobar emphysema of the right upper lobe causing incomplete collapse of the right middle and lower lobes and the left lung. Note the reduced pulmonary vascularity in the abnormal emphysematous right upper lobe. b) Right lung shows compensatory emphysema due to collapse of the left upper lobe. Note prominent pulmonary vascularity on the right indicating the normal side (white arrow).
46
R. ARTHUR
when air is loculated anteriorly the only abnormality may be increased clarity of the heart border as the lung edge may not be visible. Air in the mediastinum may be suspected by the appearance of a central area of increased translucency and increased clarity of the cardiac outline. Mediastinal air is easier to detect when the air is noted to outline the lobes of the thymus or when the air tracks along the soft tissues of the neck to give rise to streaky linear translucencies in the root of the neck. A horizontal lateral view may be useful to confirm an anterior pneumothorax and mediastinal air. Air may also leak into the pericardium, peritoneum and the systemic circulation. Leaks into the pulmonary interstitium occur particularly in ventilated neonates with idiopathic respiratory distress syndrome, where the typical bubbly appearance of pulmonary interstitial emphysema develops.
Figure 9 Adult respiratory distress syndrome: A right sided pneumothorax has been drained. On the left a loculated anterior pneumothorax (arrow) and subpulmonary pneumothorax (arrowheads) persist despite attempted drainage with two chest drains.
Increased pulmonary opacification
Increased pulmonary opacification, may be caused by the presence of pulmonary infiltrates, pulmonary collapse, pulmonary hypoplasia and agenesis, pleural fluid and by tumour infiltration. A mass lesion or diffuse thickening of the chest wall may also give rise to apparent increased opacification of the underlying lung. Pulmonary infiltrates may be alveolar (air space) or interstitial and are associated with characteristic radiological appearances.4 Air space shadowing is characterized by areas of increased opacity in the lungs which may be hazy or dense, inhomogeneous or coalesce to form confluent areas of pulmonary shadowing in which air bronchograms may be apparent. Air space shadowing is typically seen in association with infection where the infiltrate is commonly segmental or lobar (Fig. 10) or due to pulmonary oedema and opportunistic infection where the changes are generally bilateral (Fig. 11a). Interstitial infiltrates develop following thickening of the pulmonary interstitium or alveolar walls due to inflammation, fibrosis, infiltration or increased interstitial fluid. A number of patterns of pulmonary opacification may result. A predominantly linear pattern with peribronchial cuffing due to thickening of the bronchial walls is associated with acute interstitial pulmonary oedema (Fig. 11b) or infection e.g., with mycoplasma pneumoniae (Fig. 12). Other interstitial patterns, more commonly associated with chronic interstitial disease processes include reticulo-nodular, nodular, miliary shadowing and particularly, in endstage disease, a honeycomb appearance. A full discussion of the differential diagnoses of these patterns can be found in the literature.5 Pleural fluid may produce a generalized hazy increased opacification throughout the hemithorax when the X-ray is taken in the supine position. Lung markings may be visible through the fluid indicating the pleural nature of the shadowing. Large pleural effusions may displace the
Figure 10 Lobar pneumonia, demonstrating air bronchogram, and loss of mediastinal contour indicating the infection is in the left upper lobe (Silhouette sign).
become less marked on expiration and will be associated with normal or increased pulmonary vascularity. A small translucent hemithorax is most commonly associated with pulmonary hypoplasia and ipsilateral hypoplasia of the pulmonary artery e.g., following repair of congenital diaphragmatic hernia, but may be seen in the SwyerJames-Mcleod syndrome where bronchiolitis obliterans develops following a severe pneumonia. Air leaks The diagnosis of a pneumothorax may be obvious by visualization of the lung edge in association with increased translucency of the thorax (Fig. 9). However,
INTERPRETATION OF THE PAEDIATRIC CHEST X-RAY
47
Figure 11 Pulmonary infiltrates. a) Air space shadowing with prominent air bronchograms (arrowheads); b) interstitial shadowing with prominent linear pattern, fluid in the horizontal and oblique fissures (arrowheads) and a small right pleural effusion.
Figure 12 Bilateral interstitial pulmonary infiltrates due to Mycoplasma pneumonia. Figure 13 A completely opaque hemithorax due to a large pleural effusion with shift of the mediastinum to the right.
mediastinum to the contra-lateral side unless there is collapse of the ipsi-lateral lung (Fig. 13). An ultrasound examination will determine whether the pleural fluid is loculated or amenable to percutaneous drainage, and may also help to identify other pathology e.g., tumour, or congenital abnormality in the underlying lung. A thoracic tumour may also present as a large opaque hemithorax which may be difficult to differentiate from a large pleural effusion without further imaging. The mediastinum is usually shifted to the contra-lateral side and
the trachea may be bowed away from the mass. The presence of foci of calcification or evidence of bone destruction or erosion are important diagnostic clues if present. The differential diagnosis of bilateral and complete opacification of the hemithoraces depends on the age of the child although poor inspiration or tracheal obstruction may result in a generalized increase in pulmonary shadowing at
48
R. ARTHUR
Figure 14 Bilateral overinflation and widespread pulmonary infiltrates in an intubated infant with severe bronchiolitis with secondary bacterial infection.
Figure 15 Collapse of the right middle and lower lobes (arrows). A Dinky car steering wheel which had been mislaid 3 months previously was removed from the intermediate bronchus at bronchoscopy!
any age. The most common causes for the neonate and older children are given in Table 1 below. Widespread multifocal and inhomogeneous opacities have wide differential diagnosis. In the neonate this includes meconium aspiration syndrome, infection and aspiration (Fig. 14). In older children, aspiration, infection particularly TB, atypical and opportunistic infections and tumour infiltration should be considered. The diagnosis will usually depend upon the correlation of the radiological findings with clinical history and examination and laboratory investigations. Focal opacities confined to a lobar or segmental distribution are most frequently due to areas of consolidation secondary to infection or atelectasis and the classical patterns of collapse and lobar consolidation are well described in standard texts.6 The presence of an air bronchogram within the opacity confirms the presence of air space shadowing of the common bacterial pneumonias e.g., pneumococcal
Figure 16 Round pneumonia simulating a mass lesion in the right lower lobe. Table 1 Bilateral pulmonary opacification Neonate Idiopathic respiratory distress syndrome Pulmonary haemorrhage Cardiac failure Group B streptococcal infection Pulmonary hypoplasia Bilateral pleural effusions Older child Acute respiratory distress syndrome Pulmonary oedema Neuro-muscular disease Opportunistic infection infection Bilateral pleural effusions Chronic lung disease
pneumonia. The affected lobe can usually be deduced from the frontal X-ray using the silhouette sign where the mediastinal or heart border is obscured by an area of adjacent consolidation as the air soft tissue contrast required for visualisation is lost (Fig. 10). Atypical pneumonias e.g., Mycoplasma pneumonia, often result in a more interstitial pattern and are frequently bilateral (Fig. 12). Pulmonary collapse is associated with the development of a region of increased opacity associated with loss of lung
INTERPRETATION OF THE PAEDIATRIC CHEST X-RAY
49
Figure 17 Multiple ring shadows continuous with the bowel gas pattern in an infant with congenital diaphragmatic hernia. Note the mediastinal shift (arrowheads), and chest drain inserted on the right to drain a pneumothorax.
Figure 18 Scimitar syndrome. Small hemithorax associated with right basal opacity partially obscuring the diaphragmatic contour. Note the Scimitar vein (arrowheads).
volume as seen by alteration in the position of the greater fissures and/or hilar shadows (Fig. 15). Where the loss of volume is sufficiently large there may also be shift of the mediastinum towards the collapsed lung and elevation of the ipsi-lateral diaphragm. It is important to differentiate focal areas of consolidation from those of collapse, since collapse may be associated with a foreign body inhalation or other extrinsic obstruction for which bronchoscopy or surgery will be indicated. However, collapse particularly when subsegmental may be difficult to differentiate from the air space shadowing of infection especially in children with asthma. In these situations the clinical features will help decide whether a pulmonary infiltrate is due to mucus plug obstruction or underlying infection.
Multifocal nodules may be due to multiple metastases, but multifocal infection, granulomas, laryngeal papillomatosis and multiple emboli must also be considered in the differential diagnosis. Cavitation within solitary or multiple pulmonary nodules is most frequently associated with infection, and when multiple raises the possibility of septic emboli.
Ring shadows
Ring shadows refer to thin walled air filled cystic lesions in the lungs which may be solitary or multiple, and may be bilateral e.g., in bronchiectasis. The detection of the thin wall is important in the differentiation from other cavitating nodules particularly abscesses. Pneumatocoeles are the most frequent cause of ring shadows seen in children, and commonly affect the middle and lower lobes. They may be solitary or multiple and are often of varying sizes. They are particularly associated with staphylococcal and streptococcal pneumonia. Infected pneumatocoeles may contain fluid and exhibit air fluid levels on erect films. Congenital lesions which may present as a solitary thin walled cystic lesion include bronchogenic cysts or extralobar sequestrations, whereas complex masses comprising multiple ring shadows suggest the diagnosis of congenital diaphragmatic hernia or cystic adenomatoid malformation of the lung7 (Fig. 17). The former diagnosis may be obvious due to the intrathoracic position of the stomach and continuity of the thoracic ring shadows with the abdominal bowel gas. The differential diagnosis for generalized cystic lung change (i.e. the bubbly lung), is quite different from that
Pulmonary nodules
Pulmonary nodules are discrete opacities seen within the lungs. These may be solitary or multiple, of varying sizes, and may show evidence of calcification or cavitation. There is a wide differential diagnosis but by far the most common cause of a solitary pulmonary opacity seen in childhood is round pneumonia often associated with pneumococcal infection. In the acute phase air bronchograms are not usually visible and they may be erroneously misinterpreted as possible focal lesions due to tumour (Fig. 16). The clinical findings are usually suggestive of infection and the opacity and clinical status rapidly respond to antibiotic therapy.
50
R. ARTHUR
for more focal ring shadows. In the neonate pulmonary interstitial emphysema, bronchopulmonary dysplasia and Mickity Wilson disease must be considered. In older children cystic fibrosis, extensive cavitating Hodgkins disease, disseminated fungal infection and honey comb lung may result in this pattern of abnormality.
Diaphragms
Marked elevation in the diaphragm may be due to loss of lung volume, paralysis, eventration, congenital diaphragmatic hernia or subpulmonary effusion. A flattened diaphragm generally indicates overinflation of a lung but may also be due to a loculated subpulmonary pneumothorax (Fig. 9). Loss of clarity of the diaphragm may be related to adjacent pulmonary collapse or consolidation but the possibility of a congenital extralobar sequestration should be considered particularly if the opacity is slow to clear following treatment for any infection. The association of a small hemithorax and basal opacity on the right should raise a suspicion of Scimitar syndrome and a search made for the linear density of the Scimitar vein (Fig. 18).
Having completed the systematic review of the chest it is always worth reviewing those areas where abnormality is easily overlooked e.g., behind the clavicles, the mediastinum and retro cardiac regions of the lung bases. A second look at an X-ray is often useful when the appearances are difficult to interpret or there is a discrepancy with the clinical impression. Discussion with the radiologist may help to find just one more radiological clue to arrive at the correct diagnosis in the more difficult cases.
PRACTICE POINTS
X-ray appearances are influenced by technique and the age of the patient. The thymus is the commonest cause for a
widened mediastinal shadow. Increased translucency of the hemithorax consider an inhaled foreign body. Hazy increased opacity of a hemithorax, on a supine X-ray may be due to a pleural effusion. Round pneumonia is the commonest solitary pulmonary mass lesion in childhood.
Thoracic skeleton and soft tissues
Abnormalities of the thoracic skeleton are important to identify since they may be a marker for a generalised bone disorder including the expanded anterior rib ends of a rickety rosary, short or broad ribs of a skeletal dysplasia and destructive bone lesions in malignancy and infection. The identification of rib fractures on the chest X-ray of a child on the intensive care unit may be the only clue that a head injury may have been inflicted non-accidentally. Generalized thickening of the chest wall may result in increased opacity of a lung whilst a unilateral congenital absence of the pectoral muscles can simulate air trapping due to the increased translucency of the thorax. Multiple soft tissue masses e.g., neurofibroma, may project as multiple opacities in the lungs. However, close inspection of the soft tissues should reveal the subcutaneous origin of the shadows.
REFERENCES
1. 2. 3. 4. Meza PM, Benson M, Slovis M. Imaging of mediastinal masses in children. Radiol Clin North Am 1993; 31: 583604. Burton EM, Brody AS. Cardiovascular system In: Essentials of pediatric radiology. Thieme, 1999; 239269 Swischuk LE, John SD. Differential diagnosis in pediatric radiology. Lippincott, Williams and Wilkins, 1995. Willich E, Richter E. The thorax. In: Ebel K-D, Blickman H, Willich E, and Richter E (eds). Differential diagnosis in pediatric radiology. Thieme, 1999; 3151. Boklic RE, Hilman BC. Interstitial lung disease in children. 1994; 41: 543567. Caffey. Pediatric x-ray diagnosis: an integrated imaging approach. 9th ed. Mosby Yearbook, 1992. Kravitz RM. Congenital malformations of the lung. Pediatr Clin North Am 1994; 41: 453470.
5. 6. 7.
Potrebbero piacerti anche
- Paediatric Chest X-RayDocumento10 paginePaediatric Chest X-Raypheeplukz100% (1)
- Jurnal 2Documento0 pagineJurnal 2Seftiana SaftariNessuna valutazione finora
- Neonatal Chest X-Ray-2019Documento11 pagineNeonatal Chest X-Ray-2019madhu rathoreNessuna valutazione finora
- Ped CXRDocumento17 paginePed CXRSupitcha ThongchaiNessuna valutazione finora
- Pneumothoraks Jurnal RadiologiDocumento9 paginePneumothoraks Jurnal RadiologiRachmi MerrinaNessuna valutazione finora
- Pediatric Chest ImagingDocumento211 paginePediatric Chest ImagingluisalejocmNessuna valutazione finora
- Congenital Lobar EmphysemaDocumento3 pagineCongenital Lobar Emphysemamadimadi11Nessuna valutazione finora
- Ultrasound of The Thorax (Noncardiac)Documento13 pagineUltrasound of The Thorax (Noncardiac)ludiegues752Nessuna valutazione finora
- Chest X-Ray Interpretation A Structured Approach Radiology OSCEDocumento1 paginaChest X-Ray Interpretation A Structured Approach Radiology OSCEValentina SepúlvedaNessuna valutazione finora
- Thoracicneoplasms Inchildren: Beverley NewmanDocumento32 pagineThoracicneoplasms Inchildren: Beverley NewmannadiaNessuna valutazione finora
- Liszewski 2017Documento16 pagineLiszewski 2017Zakia DrajatNessuna valutazione finora
- Neonatal Chest X Ray 2019Documento11 pagineNeonatal Chest X Ray 2019Ahmad ApriansahNessuna valutazione finora
- Introduction To Pediatric Radiology Pediatric Radiology Online Course by Dr. Ashraf Abotaleb PDFDocumento436 pagineIntroduction To Pediatric Radiology Pediatric Radiology Online Course by Dr. Ashraf Abotaleb PDFRicardo VallejoNessuna valutazione finora
- Case Analysis - Radiology Clerks (Oct 16-31, 2022) : NG, Perico, Piedad, PinedaDocumento4 pagineCase Analysis - Radiology Clerks (Oct 16-31, 2022) : NG, Perico, Piedad, PinedaImmah Marie PinedaNessuna valutazione finora
- Auntminnie SignsDocumento56 pagineAuntminnie SignsHey ReelNessuna valutazione finora
- Back To Basics - Must Know' Classical Signs in Thoracic RadiologyDocumento30 pagineBack To Basics - Must Know' Classical Signs in Thoracic Radiologyvalencia suwardiNessuna valutazione finora
- Topic 20. X-RayDocumento4 pagineTopic 20. X-RayMadeline WanhartNessuna valutazione finora
- Stridor in ChildrenDocumento17 pagineStridor in ChildrenAmal AlyousifNessuna valutazione finora
- Pi Is 00960217166003657Documento15 paginePi Is 00960217166003657Jesicca SNessuna valutazione finora
- Handa 2018Documento18 pagineHanda 2018rocio enriquezNessuna valutazione finora
- ICU Imaging: Joshua R. Hill, MD, Peder E. Horner, MD, Steven L. Primack, MDDocumento18 pagineICU Imaging: Joshua R. Hill, MD, Peder E. Horner, MD, Steven L. Primack, MDJackee_BlackNessuna valutazione finora
- Radiology in Paediatric Non-Traumatic Thoracic Emergencies: Insights Into Imaging October 2011Documento15 pagineRadiology in Paediatric Non-Traumatic Thoracic Emergencies: Insights Into Imaging October 2011amaliaNessuna valutazione finora
- Chest X-ray Interpretation for Radiographers, Nurses and Allied Health ProfessionalsDa EverandChest X-ray Interpretation for Radiographers, Nurses and Allied Health ProfessionalsNessuna valutazione finora
- Asthma and Lung MassDocumento4 pagineAsthma and Lung MassAzmachamberAzmacareNessuna valutazione finora
- Pediatrics StridorDocumento13 paginePediatrics StridorEmaNessuna valutazione finora
- Pediatric Surgery 2 (Respiratory and Gastrointestinal Conditions) PDFDocumento12 paginePediatric Surgery 2 (Respiratory and Gastrointestinal Conditions) PDFRenatoCosmeGalvanJunior100% (1)
- 4.pneumothorax Imaging - Practice Essentials, Radiography, Computed TomographyDocumento11 pagine4.pneumothorax Imaging - Practice Essentials, Radiography, Computed TomographyAchmadRizaNessuna valutazione finora
- KleinDocumento14 pagineKleinsaifulmangopo123Nessuna valutazione finora
- Chest Radio GraphsDocumento13 pagineChest Radio Graphsdranees12Nessuna valutazione finora
- Congenital Malformations, TrakeaDocumento3 pagineCongenital Malformations, TrakeaAntonia DinaNessuna valutazione finora
- CDH Modern MXDocumento11 pagineCDH Modern MXphobicmdNessuna valutazione finora
- Diaphragmatic Eventration: Shawn S. Groth,, Rafael S. AndradeDocumento9 pagineDiaphragmatic Eventration: Shawn S. Groth,, Rafael S. AndradeFranciscoJ.ReynaSepúlvedaNessuna valutazione finora
- First Page PDFDocumento1 paginaFirst Page PDFmadhu rathoreNessuna valutazione finora
- Section II - Chest Radiology: Figure 1ADocumento35 pagineSection II - Chest Radiology: Figure 1AHaluk Alibazoglu100% (1)
- Stridor YousefDocumento7 pagineStridor YousefYousef Hassan BazarNessuna valutazione finora
- The V-Line: A Sonographic Aid For The Confirmation of Pleural FluidDocumento4 pagineThe V-Line: A Sonographic Aid For The Confirmation of Pleural FluidAlpi ApriansahNessuna valutazione finora
- PIIS0749070413000729Documento25 paginePIIS0749070413000729Juan Carlos MezaNessuna valutazione finora
- Accuracy of The Physical Examination in Evaluating Pleural EffusionDocumento7 pagineAccuracy of The Physical Examination in Evaluating Pleural EffusionTeresa MontesNessuna valutazione finora
- Emphysema ImagingDocumento6 pagineEmphysema Imagingestues2Nessuna valutazione finora
- Blunt Thoracic TraumaDocumento3 pagineBlunt Thoracic TraumaNetta LionoraNessuna valutazione finora
- Jurnal Radio Inggris TalDocumento17 pagineJurnal Radio Inggris TalimamattamamiNessuna valutazione finora
- Advances in Trauma UltrasoundDocumento12 pagineAdvances in Trauma UltrasoundBruce Fredy Chino ChambillaNessuna valutazione finora
- Chest X-Ray CXR InterpretationDocumento13 pagineChest X-Ray CXR InterpretationduncanNessuna valutazione finora
- Gambaran Radiologis Pada Kegawatan Paru: Farah Fatma WatiDocumento42 pagineGambaran Radiologis Pada Kegawatan Paru: Farah Fatma WatisabariaNessuna valutazione finora
- Interpreting The Chest Radio Graph Friendly)Documento9 pagineInterpreting The Chest Radio Graph Friendly)Vagner BorgesNessuna valutazione finora
- Hanan Fathy Pediatric Nephrology Unit University of AlexandriaDocumento47 pagineHanan Fathy Pediatric Nephrology Unit University of AlexandriadrhananfathyNessuna valutazione finora
- Articulo 2Documento16 pagineArticulo 2Dante Alan López RosalesNessuna valutazione finora
- 2 1 Diseases of The Respiratory System Radiological Pathology DRDocumento43 pagine2 1 Diseases of The Respiratory System Radiological Pathology DRNatnael GetahunNessuna valutazione finora
- Ultrasound A CL 2021Documento17 pagineUltrasound A CL 2021Alejandra SanchezNessuna valutazione finora
- Rapid Diagnosis and Treatment of A Pleural Effusion in A 24-Year-Old ManDocumento4 pagineRapid Diagnosis and Treatment of A Pleural Effusion in A 24-Year-Old ManJonathan EdbertNessuna valutazione finora
- Lung Cancer in ChildDocumento4 pagineLung Cancer in Childtonirian99Nessuna valutazione finora
- Journal Radiologi 3Documento14 pagineJournal Radiologi 3WinayNayNessuna valutazione finora
- Congenital Pulmonary Airway MalformationDocumento5 pagineCongenital Pulmonary Airway MalformationKiagusRoyNessuna valutazione finora
- ABC ChestDocumento4 pagineABC ChestSintounNessuna valutazione finora
- TRAUMA Pulmones, Traquea y EsofagoDocumento14 pagineTRAUMA Pulmones, Traquea y EsofagosantigarayNessuna valutazione finora
- Imaging of EmphysemaDocumento8 pagineImaging of EmphysemaWijayanti EfendiNessuna valutazione finora
- An Atlas of Neonatal Brain Sonography, 2nd EditionDa EverandAn Atlas of Neonatal Brain Sonography, 2nd EditionValutazione: 5 su 5 stelle5/5 (5)
- Pulmonary Functional Imaging: Basics and Clinical ApplicationsDa EverandPulmonary Functional Imaging: Basics and Clinical ApplicationsYoshiharu OhnoNessuna valutazione finora
- Kumpulan POMRDocumento26 pagineKumpulan POMRIMN Wiranta PrasetyajiNessuna valutazione finora
- Y Tube Ipost Test OnlineDocumento11 pagineY Tube Ipost Test OnlineMohammed Manassra100% (1)
- Lecture 1 Chest TraumaDocumento19 pagineLecture 1 Chest Traumaj.doe.hex_870% (1)
- Emergency Lecture PDFDocumento57 pagineEmergency Lecture PDFDuane Liloc100% (1)
- Bontragers Textbook of Radiographic Positioning and Related Anatomy 9th Edition Lampignano Test BankDocumento16 pagineBontragers Textbook of Radiographic Positioning and Related Anatomy 9th Edition Lampignano Test BankMrsKellyHammonddqnya100% (9)
- EPOSTER HARIO-dikonversiDocumento1 paginaEPOSTER HARIO-dikonversiHario HendrokoNessuna valutazione finora
- Pluera and LungsDocumento105 paginePluera and LungsueumanaNessuna valutazione finora
- Ati Med SurgDocumento36 pagineAti Med SurgVanessaMUeller94% (17)
- Chest DrainageDocumento28 pagineChest DrainageCristy Guzman100% (1)
- Responsi Spontan PneumothoraxDocumento49 pagineResponsi Spontan PneumothoraxNizarAbdullahNessuna valutazione finora
- PneumothoraxDocumento2 paginePneumothoraxAfif MansorNessuna valutazione finora
- Respiratory PathologyDocumento42 pagineRespiratory PathologyMorgan PeggNessuna valutazione finora
- 4 Tactical Evacuation CareDocumento15 pagine4 Tactical Evacuation CareINSTRUCTOR INTERNACIONAL ANTONIO RAMIREZNessuna valutazione finora
- Primery Survey GadarDocumento11 paginePrimery Survey GadarapryNessuna valutazione finora
- Management of Pneumothorax With Oxygen Therapy A CDocumento4 pagineManagement of Pneumothorax With Oxygen Therapy A CAndreas Paian SiahaanNessuna valutazione finora
- Can Thiep Dau Duoi Sieu AmDocumento138 pagineCan Thiep Dau Duoi Sieu AmLinh DieuNessuna valutazione finora
- Minimally Invasive Thoracic SurgeryDocumento9 pagineMinimally Invasive Thoracic SurgeryLEONEL ALFREDO GUILLEN MARINNessuna valutazione finora
- Thoracocentesis: Nadja E. SigristDocumento4 pagineThoracocentesis: Nadja E. SigristDwi AnaNessuna valutazione finora
- Study GuideDocumento46 pagineStudy GuideAkia Cayasan BayaNessuna valutazione finora
- Chest Tube Insertion: Prepare By: Amrita:Thapa Roll No. 7Documento39 pagineChest Tube Insertion: Prepare By: Amrita:Thapa Roll No. 7sushma shresthaNessuna valutazione finora
- Legal Med. Report-QueenDocumento61 pagineLegal Med. Report-QueenQuinnee VallejosNessuna valutazione finora
- Case Report Surgical Management of Lumbal Compression FractureDocumento8 pagineCase Report Surgical Management of Lumbal Compression FractureYustinusrobbyNessuna valutazione finora
- ATI Repiratory Questions - Answer KeyDocumento11 pagineATI Repiratory Questions - Answer KeyLeelee SheppardNessuna valutazione finora
- Gunshot Wounds: Ivanova Zalina M.DDocumento18 pagineGunshot Wounds: Ivanova Zalina M.DVaishali Singh100% (1)
- Chapter 27: Alterations of Pulmonary Function Power-Kean Et Al: Huether and Mccance'S Understanding Pathophysiology, Second Canadian EditionDocumento13 pagineChapter 27: Alterations of Pulmonary Function Power-Kean Et Al: Huether and Mccance'S Understanding Pathophysiology, Second Canadian EditionmonicaNessuna valutazione finora
- Trauma ArrestDocumento12 pagineTrauma ArrestydtrgnNessuna valutazione finora
- Air Leak Syndrome: Signs and SymptomsDocumento2 pagineAir Leak Syndrome: Signs and SymptomsSheila Rose SalesNessuna valutazione finora
- Chest InjuriesDocumento8 pagineChest InjuriesNavjot BrarNessuna valutazione finora
- Belluomini - Clean Needle TechniqueDocumento81 pagineBelluomini - Clean Needle Techniquealbertomtch75% (4)
- Exam Code HAAD4Documento12 pagineExam Code HAAD4Carmela Marie Eriguel CuetoNessuna valutazione finora