Documenti di Didattica
Documenti di Professioni
Documenti di Cultura
IVD Guide
Caricato da
volkandemirDescrizione originale:
Titolo originale
Copyright
Formati disponibili
Condividi questo documento
Condividi o incorpora il documento
Hai trovato utile questo documento?
Questo contenuto è inappropriato?
Segnala questo documentoCopyright:
Formati disponibili
IVD Guide
Caricato da
volkandemirCopyright:
Formati disponibili
EuropeAid /114385/D/SV/CY Assessment and administration capacity building for the harmonisation with the New Approach directives
GUIDE
to the implementation of DIRECTIVE
98/79/EC
on
IVD
In Vitro Diagnostic Devices
Guide on IVD Directive
EuropeAid /114385/D/SV/CY
This Guide was printed with funds of the EuropeAid /114385/D/SV/CY project: Assessment and administration capacity building for the harmonisation with the New Approach directives The views expressed in this publication do not necessarily reflect the views of the European Commission or the Cyprus Ministry of Commerce, Industry and Tourism. Republic of Cyprus, Nicosia, May 2004
Guide on IVD Directive
EuropeAid /114385/D/SV/CY
INTRODUCTION
This information is intended as general guidance and should not be regarded as an authoritative statement of the law. Manufacturers and others should not rely on this information but should consult the legislation referred to, making their own decisions on matters affecting them in conjunction with their lawyers and other professional advisers.
What is a medical device?
A series of three Directives regulating the safety and marketing of medical devices throughout the European Union started to come into effect from 1 January 1993. For the purposes of the Directives, a medical device is defined as: "any instrument, apparatus, appliance material or other article, whether uses alone or in combination, including the software necessary for its proper application intended by the manufacturer to be used on human beings for the purpose of: diagnosis, prevention, monitoring, treatment or alleviation of disease, diagnosis, monitoring, treatment, or alleviation of or compensation for an injury or handicap, investigation, replacement or modification of the anatomy or of a physiological process, control of conception and which does not achieve its principal intended action in or on the human body by pharmacological, immunological or metabolic means, but which may be assisted in its function by such means".
Why do we need the directives?
Before the introduction of the medical devices Directives, each Member State in the European Union would control the safety and marketing of medical devices on its territory in different ways. The Directives benefit manufacturers by harmonising controls within a single system and avoid the need for manufacturers having to comply with 15 (or 25) different sets of rules. Purchasers and users can also be reassured that devices manufactured anywhere in the Union should meet common standards of performance and safety. The Directives benefit patients and users, by setting out essential requirements that products must meet. These make it clear that devices must not compromise the health or safety of the patient, user or any other person, and that any risks associated with the device are compatible with patient health and protection. Any side effects must be acceptable when weighed against the intended performance of a device. Devices meeting these requirements generally carry the "CE" mark to show that they comply.
THE DIRECTIVES
THE ACTIVE IMPLANTABLE MEDICAL DEVICES DIRECTIVE
This first Directive covers all powered implants or partial implants that are left in the human body. Heart pacemakers are the most common example of powered implants.
Guide on IVD Directive
EuropeAid /114385/D/SV/CY
THE MEDICAL DEVICES DIRECTIVE
The second Directive covers most other medical devices, ranging from, for example, first aid bandages, tongue depressors, hip prostheses, X-Ray equipment, ECG and heart valves.
THE IN VITRO DIAGNOSTIC MEDICAL DEVICES DIRECTIVE
The third Directive covers any medical device which is a reagent, reagent product, calibrator, control material, kit, instrument, apparatus, equipment or system intended for use in-vitro for the examination of specimens, including blood and tissue donations, derived from the human body. Examples of in-vitro diagnostic devices are blood grouping reagents, pregnancy test kits and Hepatitis B test kits.
KEY POINTS IN THE DIRECTIVES
CE MARKING
The CE mark that appears on a medical device or on its packaging means that the device satisfies the relevant essential requirements and is fit for its intended purpose as specified by the manufacturer. Eventually all devices, (except custom-made devices, those intended for clinical investigations and devices for performance evaluation) whether used in private or public hospitals and nursing homes or sold in retail outlets, will have to carry the CE marking.
CLASSIFICATION
The Medical Devices Directive and the In Vitro Diagnostic Medical Devices Directive include a classification system whereby the level of regulatory control applied to devices is proportionate to the degree of risk associated with the device. The strictest controls therefore apply only to high-risk products.
THE COMPETENT AUTHORITY
The Competent Authority is the body responsible for implementing the requirements of the Directives in each Member State. In the UK, the Competent Authority is the Secretary of State for Health acting through the MHRA. The Competent Authority's main role is to ensure that manufacturers comply with the Regulations, to evaluate adverse incident reports received from manufacturers, and carry out a pre-clinical assessment of devices intended for clinical investigation.
NOTIFIED BODIES
The Competent Authority is also responsible for designating the independent certification organisations (Notified Bodies) that check that manufacturers of medium and high risk medical devices have followed the requirements. Once they are satisfied, manufacturers may apply the CE mark to their products and place them on the market.
CLINICAL INVESTIGATIONS
The Directives require all devices intended for clinical investigation in the EU to be formally assessed on the risks such investigations might pose to the health and safety of patients. For most medical devices (except in-vitro diagnostic devices) manufacturers have to inform the relevant Competent Authority if they
Guide on IVD Directive
EuropeAid /114385/D/SV/CY
intend to start a clinical investigation at least 60 days before it is due to begin. The investigation may start after the 60-day period unless the Competent Authority notifies the manufacturer of a decision to the contrary on grounds of public health.
ADVERSE INCIDENT REPORTING (VIGILANCE)
Manufacturers are required to report serious incidents to the Competent Authority. Information about these is collected and evaluated centrally and, where necessary, made available to other Member States. The overall aim of the system is to improve the safety of patients, users and others by trying to prevent incidents similar to those reported occurring elsewhere in the Union.
IN VITRO DIAGNOSTIC MEDICAL DEVICES DIRECTIVE
The In Vitro Diagnostic Medical Devices Directive (98/79/EC) (the Directive) was formally adopted in October 1998 and published in the Official Journal of European Communities on 7 December 1998 (ref OJ No. L331 7.12.98 p.1). The Directive introduced for the first time common regulatory requirements dealing specifically with the safety, quality and performance of in vitro diagnostic medical devices (IVDs), thereby bringing them into line with other medical devices. In outline, the Directive is intended to ensure that IVDs do not compromise the health and safety of patients, users and third parties and attain the performance levels attributed to them by their manufacturer. The relevant provisions of the Directive came into force on 7 June 2000. However, there was a transitional period until December 2003. From this date IVDs which are placed on the market must comply with the Directive. Non-compliant IVDs which have been placed on the market by 7 December 2003 must be put into service (i.e. first made available to a final user) by 7 December 2005. IVDs which are put into service but not placed on the market have until 7 December 2005 to comply with the legislation.
SCOPE OF THE DIRECTIVE
What is an in vitro diagnostic medical device?
The Directive defines an IVD as: any medical device which is a reagent, reagent product, calibrator, control material, kit, instrument, apparatus, equipment, or system, whether used alone or in combination, intended by the manufacturer to be used in vitro for the examination of specimens, including blood and tissue donations, derived from the human body, solely or principally for the purpose of providing information: concerning a physiological or pathological state, or concerning a congenital abnormality, or to determine the safety and compatibility with potential recipients, or to monitor therapeutic measures." This definition needs to be read in conjunction with the definition of a medical device in the Directive: Medical device means any instrument, apparatus, appliance, material or other article, whether used alone or in combination, including the software necessary for its proper application, intended by the manufacturer to be used for human beings for the purpose of:
Guide on IVD Directive
EuropeAid /114385/D/SV/CY
diagnosis, prevention, monitoring, treatment or alleviation of disease, diagnosis, monitoring, treatment, alleviation or compensation for an injury or handicap, investigation, replacement or modification of the anatomy or of a physiological process, control of conception, and which does not achieve its principal intended action in or on the human body by pharmacological, immunological or metabolic means, but which may be assisted in its function by such means. In order for a product to come within the scope of the Medical Device Directives (including the IVDD) it must be intended by the manufacturer to be used for a medical purpose. If no medical purpose is intended by the manufacturer then the product is not a medical device. Thus the intended purpose for a product will be key in determining whether or not the product is a (IVD) medical device. It is to be noted that Article 1.2 (h) of the IVDD provides that the intended purpose is the use for which a device is intended according to the data (presumably, the information) supplied by the manufacturer on the labelling, in the instructions for use and / or in promotional material. Examples: Devices for detection of agents of biological or chemical warfare in the environment are not IVDs because such products have no medical purpose. On the other hand a device intended to be used on human specimens in the detection of biological or chemical warfare agents with medical purpose would fall within the scope of the IVDD. Devices intended to be used only in the course of law enforcement or other non-medical purposes, for example for detecting drugs of abuse/alcohol, are outside the scope of the IVDD. If however, the in- vitro examination of human specimens with a medical purpose is one of the intended uses of a specific product, the IVD Directive will apply. A product for research use only which has no medical purpose, cannot be a medical device and, therefore, cannot be an IVD medical device. By definition, these products fall outside the scope of the IVDD and the other medical devices directives.
SPECIMEN RECEPTACLES
Article 1.2(b) of the IVD Directive clearly states that specimen receptacles are considered as IVDs. It defines specimen receptacles for this purpose as those devices that are specifically intended by the manufacturer to be for the primary containment and preservation of specimens derived from the human body for the purposes of in vitro diagnostic examination. This applies whether the product is vacuum type or not. This specific intended use should be included on the labelling and any associated promotional literature for the product. The manufacturer must also have evidence and technical documentation to support this use for the product. A manufacturer cannot place the CE mark on a piece of general laboratory equipment as a marketing claim, without ensuring that it complies with all the relevant essential and other requirements of the directive and have evidence to substantiate this. It is possible for more that one specimen receptacle to be involved in the collection, transport and storage of an individual specimen. In such cases the manufacturer of each receptacle must have evidence of compliance with the directive as above.
Guide on IVD Directive
EuropeAid /114385/D/SV/CY
During the actual analytical process the receptacles into which the specimen is placed (by aliquoting or otherwise), may be glass or plastic tubes, cups, cuvetttes or other receptacles. These are unlikely to be specimen receptacles as defined in the directive. They are usually considered to be general laboratory equipment, however in some cases they could be considered accessories to an IVD. In the context of the definition of specimen receptacle in Art. 1.2 (b), of the IVD Directive it should be noted that: a. the word primary does not necessarily refer to the initial or first container of the specimen in point of time, but rather to a container that is intended by its manufacturer to mainly come into direct contact with the specimen and which could therefore affect the specimen, and b. the word preservation does not imply that the receptacle has to contain a specimen preservative, but that the receptacle is one intended to protect the specimen, for example, from temperature fluctuations, from light, from physical breakage, etc. Example: Sample tubes, microbial transportation devices (tubes). Article 1 (2) b applies.
PRODUCTS FOR GENERAL LABORATORY USE
Products for general laboratory use are not IVDs unless, in view of their characteristics, they are intended specifically by their manufacturer to be used for in vitro diagnostic examination of samples derived from the human body for the purposes outlined in the definition of an IVD. Thus such a product must possess specific characteristics to make it suitable for in vitro diagnostic procedures in order to be classified as an IVD. Products used in vitro in the preparation of samples that have been obtained for examination, but not used directly in the actual test can be considered to be IVDs if the manufacturer specifically states that the product can be used for in vitro diagnostic purposes, otherwise they fall outside the scope of the directive. The one qualification provided for in the Directive is where, on the basis of its characteristics (emphasis added), a manufacturer specifically intends that the product should be used for in vitro diagnostic examination. In this case, the product becomes an IVD and must comply with the applicable essential and other requirements of the IVDD and must be CE marked. If, however, the product does not in fact possess specific characteristics that make it suitable for one or more identified in vitro diagnostic examination procedures, then the manufacturer is not free to bring it within the scope of the IVDD merely by affixing the CE marking to it. In other words, a manufacturer is not able to bring within the scope of the IVDD a product that, in reality, is a piece of general laboratory equipment simply by affixing the CE mark to it. Example: Laboratory products that are not usually considered to fall within the scope of the IVD directive include: sterilizers, laboratory centrifuges, general purpose automatic pipette, weighing machines, microtomes, multi purpose tubes, pipettes and flasks etc. where such items have no specifically intended in vitro diagnostic use. Examples also include items for general purpose use such as foetal calf serum, culture media and stains, unless the manufacturers intended purpose falls within the definition of an in vitro diagnostic medical device.
Guide on IVD Directive
EuropeAid /114385/D/SV/CY
ACCESSORIES TO IVDS
Accessory means an article which, whilst not being an in vitro diagnostic medical device, is intended specifically by its manufacturer to be used together with a device to enable that device to be used in accordance with its intended purpose. (Article 1.2(c)). For the purposes of the Directive accessories are treated as IVDs in their own right. However "invasive sampling devices" or those which are directly applied to the human body for the purpose of obtaining a specimen, are not considered to be accessories to IVDs. Generally, such devices will be regulated by Directive 93/42/EEC (Medical Devices). However, where a diagnostic device incorporates an invasive element and a diagnostic element and is sold as a single integrated unit (rather than two separate products within the same pack) the general view is that such a device will generally be treated as an IVD, rather than a general device regulated by Directive 93/42/EEC.
DEVICES FOR PERFORMANCE EVALUATION
A device for performance evaluation" means a device intended by the manufacturer to be subject to one or more performance evaluation studies in laboratories for medical analyses, or in other appropriate environments outside his own premises. Instruments, apparatus, appliances, materials or other articles which are intended to be used for research purposes without any medical objective are not regarded as devices for performance evaluation. Devices for performance evaluation are not subject to the normal conformity assessment/CE marking procedures (which are detailed below), but manufacturers must draw up the statement and follow the procedure set out in Annex VIII of the Directive and must also register with the Competent Authority (see later under Registration for further information).
PRODUCTS FOR RESEARCH USE ONLY
A product for research use only which has no medical purpose, cannot be a medical device and, therefore, cannot be an IVD medical device. By definition, these products fall outside the scope of the IVDD and the other medical devices directives. Recital 8 of Directive 98/79/EC states: Whereas instruments, apparatus, appliances, materials or other articles, including software, which are intended to be used for research purposes, without any medical objective, are not regarded as devices for performance evaluation. This means that products for research purposes, which have no medical indication and are not yet established in medical diagnosis, are excluded from the scope of the directive.
DEVICES FOR IVD PURPOSES WITH AN INVASIVE BODY CONTACT
Some devices may incorporate both specimen collection and analytical functions. These devices may be borderline between the Medical Device Directive 93/42/EC and the IVD Directive 98/79/EC. Borderline cases such like this should be approached with regard to the principal intended purpose of the product. Thus if the principal intended purpose is for the product is to be used in vitro for the examination of
Guide on IVD Directive
EuropeAid /114385/D/SV/CY
specimens derived from the human body for the purposes of providing information the IVD directive would apply. (Art 2b). Devices which during their measuring function involve contact with the human system (as defined in 93/42/EEC Annex IX) in order to obtain a continuous sample are not considered to be IVD devices. Example: A device involving the vacuum suction of saliva into the integrated handle of a device which contains reagent material (e.g. for the detection of HIV). The use of such a device involves the penetration of the device into a body orifice for the collection of the specimen and this may appear to make it a medical device within the scope of the MDD. However, its principal intended purpose is the provision of relevant information by the in vitro examination of the specimen derived from the patient. The devices brief contact with the patient or penetration into the patients body to collect the specimen is subsidiary and incidental to its principal intended purpose. Mouth and other swabs having integrated reagents or reagent areas are IVDs because their principal intended purpose is to provide information relevant to the medical purposes specified in Article 1.2 (b). The intended purpose, invasiveness and continuity of sampling are important criteria in deciding the correct regulatory route for this kind of product. Example: A Holter blood glucose monitoring system that includes a subcutaneous catheter to provide a continuous supply of the patients specimen to an in vitro analysing instrument is a medical device not an IVD because, during the in vitro measuring function, surgically invasive contact with the patient is necessary in order to obtain a continuous specimen flow. In this case, the analytical function is carried out at the same time as the continuous specimen collection process is still going on. There is no dissociation of the specimen from the patient and therefore the analytical function cannot properly be regarded as being in vitro. Such a device would therefore be a medical device within the scope of the MDD. Other examples are continuous pH measurements during haemodialysis and oxygen saturation devices.
OTHER SAMPLING DEVICES
Sampling devices which are either invasive (needles, lancets etc) or that come into contact with patients, that are made available together with IVD devices (in kits, procedure packs etc) are not considered to be accessories to IVD devices, They come within the scope of the MDD and have to be CE marked as medical devices. Note: For devices that have key features, which could be considered as being covered by the MDD as well, the relevant requirements (e.g. biocompatibility, sterility, etc.) of that directive need to be considered during the conformity assessment procedure.
KITS CONTAINING IVDS AND MEDICINAL PRODUCTS
Medicinal products made available together with IVD devices are to be authorised for marketing by the normal process for medicinal products and the primary, and if applicable the secondary packages of the medicinal products are to be labelled according to the rules for medicinal products. The IVD components of the kits themselves fall within the scope of the IVD Directive and must therefore comply with its requirements and carry the CE mark, but will not need not be authorised for marketing as medicinal. This
Guide on IVD Directive
EuropeAid /114385/D/SV/CY
also applies to any MDD components. I.e. each set of regulatory requirements needs to be met for each of the individual medicinal products, medical device and/or IVD medical device components. Example: A Helicobacter pylori breath test kit containing labelled urea (a licensed pharmaceutical product) to be ingested prior to sampling for analysis, a straw (a medical device) and a sample container (an IVD). Note: So far as the medicinal product is concerned, it must have been granted a marketing authorisation covering the actual use for which it is being included in the IVD kit and it must be labelled in accordance with the regulations relating to medicinal products. The IVD component of the kit must comply with all the applicable essential and other requirements of the IVDD (including the labelling requirements) and it must bear the CE marking. It is important to note that, while the CE marking of the IVD content of the kit entitles it to free movement within the European Economic Area (subject to language requirements), that is not so as regards the medicinal product. That aspect must be considered separately under the relevant medicinal product legislation as must the extent to which the label of the kit must provide information about the medicinal product over and above a statement that the kit contains the medicinal product.
CERTIFIED REFERENCE MATERIAL
Although internationally certified reference material and those materials used for external quality assurance schemes are not covered by the legislation, calibrators and control materials needed to establish or verify performance of devices are IVDs.
DEVICES WHERE NO SPECIMEN IS INVOLVED
Some medical diagnostic devices function without a specimen is being taken from the patient. Since it is an essential part of the definition of an in vitro diagnostic medical device that the manufacturers intention is that the product should be used .. for the examination of specimens., it follows that a medical device that functions without the need for a patient specimen cannot be an IVD within the scope of the IVDD. Such products will almost always be a medical device within the scope of the MDD. Example A non-invasive medical device for the detection of blood glucose by energy emission (e.g. near infrared energy) is not an IVD because no specimen derived from the human body is involved, but it would be a medical device for the purposes of the MDD.
DEVICES USED IN LAW ENFORCEMENT
Devices intended to be used only in the course of law enforcement or other non-medical purposes, for example paternity tests or tests for detecting drugs of abuse/alcohol, are not IVDs. If however, the in vitro examination of human specimens with a medical purpose is one of the intended uses of a specific product, the IVD Directive will apply.
Guide on IVD Directive
EuropeAid /114385/D/SV/CY
IVDS NOT PLACED ON THE MARKET
The conformity assessment procedures apply not only to IVDs which are placed on the market, but also to any person who manufactures IVDs and, without placing them on the market, puts them into service and uses them within the context of his professional activity. (Article 9.13 of the Directive). Thus, for example, a person who manufactures an IVD and then uses it to provide diagnostic services would generally need to comply with the appropriate conformity assessment procedure in respect of that device.
EXEMPTION FOR HEALTH INSTITUTIONS
Article 1.5 of the Directive (see Regulation 33) excludes from its scope devices manufactured and used only within the same health institution and on the premises of their manufacture or used on premises in the immediate vicinity without having being transferred to another legal entity. As to the ambit of this exemption, it is clear that where a health institution manufactures an IVD and transfers it to another legal entity (whether free or as part of a commercial transaction), the exemption does not apply and therefore the Directive does apply.
TRADE FAIRS ETC
IVDs which are not in compliance with the regulatory requirements may be shown at trade fairs, exhibitions, demonstrations, scientific or technical gatherings etc. provided that such devices are not used on specimens taken from the participants and a visible sign clearly indicates that the device cannot be marketed or put into service until it complies with the requirements of the Directive.
THE CONFORMITY ASSESSMENT PROCESS
OVERVIEW
In general terms, a manufacturer wishing to place his products on the market under this Directive must: assign his devices to one of the relevant risk categories defined in the Directive; ensure that the device meets the "essential requirements" specified in Annex I of the Directive; follow the appropriate conformity assessment procedure; if appropriate (depending on the risk category of the device), ensure that an independent certification body (called a Notified Body) is involved in the conformity assessment procedure. As stated earlier, manufacturers of IVDs which are not placed on the market but which are put into service and used in the context of the manufacturers professional activity must also follow the appropriate conformity assessment procedure. MANUFACTURER The manufacturer is defined in the Directive as: a. the person with responsibility for the design, manufacture, packaging and labelling of a device before it is placed on the market under his own name, regardless of whether these operations are carried out by that person himself or on his behalf by a third party; or b. a person who assembles, packages, processes, fully refurbishes and/or labels one or more ready made products and/or assigns to them their intended purpose as devices with a view to their being
Guide on IVD Directive
EuropeAid /114385/D/SV/CY
placed on the market under his own name. This does not apply to a person who assembles or adapts devices already on the market to their intended purpose for an individual patient. AUTHORISED REPRESENTATIVE Manufacturers who do not have a registered place of business in the EC must designate an authorised representative to perform certain obligations (e.g. to make certain documentation available on request) and may designate an authorised representative to perform other substantive obligations. Additionally, such a manufacturer must also designate an authorised representative as the person responsible for marketing the IVD in the EC and for registering that device with the appropriate Competent Authority.
NOTIFIED BODY
A "Notified Body" is a certification organisation which the Competent Authority designates to carry out certain tasks in respect of the conformity assessment procedures described in the Annexes to the Directive. A notified body must be qualified to perform all the functions set out in any Annex for which it is designated. The tasks which a notified body can carry out may be restricted by the Competent Authority. The activities of notified bodies are regularly monitored. Manufacturers are free to apply to any notified body in the EU designated to carry out the desired conformity assessment procedure, regardless of which Member State that notified body is designated in. Manufacturers are required to inform their notified body of changes to their product ranges and quality system. In cases where design or type examination has been carried out by the notified body the manufacturer is required to notify them of changes to the design, as well as any information they have on changes to the pathogen and markers of infection to be tested. All such changes need to be approved by the notified body prior to implementation.
ESSENTIAL REQUIREMENTS
Annex I of the Directive lists various "essential requirements" with which IVDs must comply before being placed on the market/put into service. These aim to ensure that IVDs do not compromise the health and safety of patients, users, and others, and are designed and manufactured so that they are suitable for the relevant purpose specified by the manufacturer and achieve the performances stated by the manufacturer. Not all the essential requirements will apply to all devices and it is for the manufacturer of the device to assess which are appropriate for his particular product. In determining this, account must betaken of the intended purpose of the device.
LEVEL OF REGULATORY CONTROL
The majority of IVDs do not require the intervention of a notified body in the conformity assessment process. However, for some IVDs (the correct performance of which is perceived to be essential to health), involvement of a notified body will be required. For the purposes of the conformity assessment procedures, the Directive groups IVDs into four categories.
Guide on IVD Directive
EuropeAid /114385/D/SV/CY
THE FOUR CATEGORIES
These categories are, in order of increasing perceived risk: general IVDs, i.e. all IVDs other than those covered by Annex II and IVDs for self-testing; IVDs for self-testing (a device intended by the manufacturer to be able to be used by lay persons in a home environment) excluding self-test devices covered in Annex II; IVDs in Annex II List B of the Directive: Which, amongst others, includes reagents products for rubella, toxoplasmosis and phenylketonuria as well as devices for self testing for blood sugar; IVDs in Annex II List A of the Directive: Which includes reagents and products for HIV I and II, Hepatitis B, C and D, and reagent products for determining ABO systems and anti-Kell.
THE CONFORMITY ASSESSMENT ROUTES
In order to demonstrate compliance with the essential requirements, the manufacturer must follow the conformity assessment procedure appropriate for the category of IVD concerned. Conformity assessment routes are detailed in Article 9 of the Directive, which cross-refers to the relevant Annexes. The conformity assessment routes are outlined below.
GENERAL IVDS
The manufacturer must fulfil the applicable obligations imposed by sections 1 to 5 of Annex III of the Directive and must declare and ensure that the device meets the provisions of the Directive which apply (EC declaration of conformity). No notified body involvement is required.
SELF-TEST IVDS NOT COVERED IN ANNEX II
The manufacturer, in addition to complying with the requirements for general IVDs, must, before a declaration of conformity can be made, lodge an application with a notified body for the examination of the design of the device (section 6 of Annex III of the Directive). This will include aspects affecting its suitability for non-professional users. Alternatively the manufacturer may follow the conformity assessment routes for higher risk products as below.
ANNEX II IVDS
For Annex II List B devices, the manufacturer must follow the applicable obligations imposed either by Annex IV of the Directive (EC declaration of conformity, full quality assurance system), or by Annexes V (EC type-examination) and VI (EC verification), or alternatively by Annexes V (EC type-examination) and VII (EC declaration of conformity, production quality assurance) and must declare and ensure that the device meets the provisions of the Directive which apply. For List A devices, the manufacturer must follow either Annex IV, or alternatively Annexes V and VII (i.e. it cannot follow Annexes V and VI). All Annex II IVDs require the intervention of a notified body before a declaration of conformity with the Directive can be made.
ANNEX II LIST B IVDS
The notified body will
Guide on IVD Directive
EuropeAid /114385/D/SV/CY
either carry out an audit of the full quality assurance system or carry out type examination plus verification of each batch or product or carry out type examination plus audit of the production quality assurance system.
ANNEX II LIST A IVDS
A notified body will either carry out an audit of the full quality assurance system and review the product design dossier or carry out type examination plus audit of the production quality assurance system. In addition, for Annex II list A IVDs, the notified body must verify each product or batch of product before the manufacturer may place them on the market.
Guide on IVD Directive
EuropeAid /114385/D/SV/CY
CONFORMITY ASSESSMENT PROCEDURE
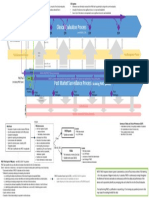
The conformity assessment routes are summarised in the following diagrams. Diagram 1: Conformity Assessment Routes for IVDs (1)
Guide on IVD Directive
EuropeAid /114385/D/SV/CY
Diagram 2:
Conformity Assessment Routes for IVDs (2).
Guide on IVD Directive
EuropeAid /114385/D/SV/CY
DOCUMENTATION
The declaration of conformity, the technical documentation and the decision, reports and certificates of notified bodies must be kept available for inspection for a period of five years after manufacture of the last device.
HARMONISED STANDARDS
European Standards that have been harmonised under the IVD Directive may be used to show conformity with the relevant essential requirements. Compliance with an appropriate harmonised European Standard gives a presumption of conformity with the essential requirements to which the standard relates. The use of harmonised standards is not mandatory, and other standards exist that are not harmonised which may be used to assist in showing conformity. However, unlike harmonised standards they offer no presumption of conformity.
COMMON TECHNICAL SPECIFICATIONS
For products currently in List A of Annex II conformity assessment may involve the use of Common Technical Specifications (CTS) (Commission Decision of 7 May 2002 on common technical specifications for in-vitro diagnostic medical devices (notified under document C (2002) 1344) to establish performance evaluation and re-evaluation criteria, batch release criteria, reference methods and reference materials. CTS are drawn up by an Expert Group convened by the Commission rather than by the Standards Bodies. Manufacturers are expected to comply with the CTS. If they have justified reasons for not doing so they must adopt solutions that they can prove are at least equivalent to the CTS in terms of the above criteria. As with harmonised standards, Member States must presume compliance with the essential requirements in respect of devices designed and manufactured in conformity with common technical specifications.
THE CE MARKING
WHAT THE CE MARK MEANS
IVDs must bear the CE marking when they are placed on the market. Devices for performance evaluation do not need to be CE marked. A manufacturer must not apply the CE mark unless he has fulfilled the applicable obligations of the Directive. The CE mark is therefore seen as a declaration by the manufacturer that the product meets all of the appropriate provisions of the relevant legislation, including those relating to safety. A device bearing a CE mark can be freely marketed anywhere in the EU without further control, except that competent authorities can take action to prevent the supply of a device in certain circumstances. The CE marking is as illustrated in diagram 3 below. It may not be smaller than 5 mm in its vertical height, and the proportions must be maintained whatever its size. The grid does not form part of the marking and is for information only. Diagram 3
Guide on IVD Directive
EuropeAid /114385/D/SV/CY
It should be noted that the C and E are not formed by perfect semi-circles, i.e. the top and bottom arms extend one square beyond the semi-circles, and the middle arm of the E stops one square short.
AFFIXING THE CE MARKING
The CE marking must be affixed in a visible, legible and indelible form on the device (where practicable and appropriate) and on the instructions for use. It must also appear on the sales packaging. The relevant notified body number (where one has been used) should accompany the CE marking.
DEVICES NOT PLACED ON THE MARKET
Where a device is not placed on the market, but Article 9.13 applies (i.e. the device is put into service and used in the context of a professional activity), it is not practical or appropriate to apply a CE mark. Therefore the CE marking requirement does not apply to such a situation.
OTHER REGULATORY REQUIREMENTS
LANGUAGE USED IN LABELLING AND INSTRUCTIONS FOR USE
The Directive allows Member States to stipulate in their implementing legislation that the information needed to use an IVD (labelling and instructions) is in their official language. If the device is a device for self-testing, the instructions for use and label must include a translation into the official language of any member state of the community in which the device reaches a final user.
REGISTRATION
A manufacturer who places a relevant device on the market (remember that for these purposes, market means Community market) or who makes available a device for performance evaluation under his own name must register with the Competent Authority in the Member State where he, or his authorised representative is established.
EUROPEAN DATABANK
A European Databank is being set up, accessible only by Member States and the European Commission. It will contain data relating to all devices available on the territory of the Community. Member States will load the information they receive from manufacturers and Notified Bodies onto the Databank. The Databank will include; Data relating to registration of manufacturers and devices; Data relating to certificates issued, modified; complemented, suspended, withdrawn or refused by notified bodies; Data obtained in accordance with the vigilance system (see below).
Guide on IVD Directive
EuropeAid /114385/D/SV/CY
POST MARKET SURVEILLANCE AND VIGILANCE PROCEDURES
The conformity assessment procedures include obligations with regard to experience gained in the postproduction phase, including implementation of any necessary corrective actions. Manufacturers must maintain a vigilance system to notify the regulatory authorities of incidents that might lead to or might have led to death or serious health consequences, or to a systematic recall of a device.
MEMBER STATES REPORTING OBLIGATIONS
In outline, Member States have obligations under the Directive, to inform the European Commission and other Member States where an IVD has been withdrawn, prohibited or restricted because it may compromise the health and safety of patients, users or others and/or where an incident has been reported as part of the vigilance procedure.
ANNEX: LIST OF HARMONISED STANDARDS
Standard reference EN 375: 2001 Titles Information supplied by the manufacturer with in vitro diagnostic reagents for professional use Information supplied by the manufacturer with in vitro diagnostic reagents for self-testing Medical gloves for single use Part 1: Requirements and testing for freedom from holes Medical gloves for single use Part 2: Requirements and testing for physical properties (including Corrigendum 1996) Sterilisation of medical devices Validation and routine control of sterilisation by irradiation Sterilisation of medical devices Requirements for medical devices EN 556-1: 2001 to be designated 'Sterile' - Part 1: Requirements for terminally sterilised medical devices EN 591: 2001 EN 592: 2002 EN 794-1: 1997/ A1:2000 Instructions for use in vitro diagnostic instruments for professional use Instructions for use for in vitro diagnostic instruments for self-testing Lung ventilators Part 1: Particular requirements for critical care ventilators In vitro diagnostic systems Guidance on the application of EN 928: 1995 EN 29001 and EN 46001 and of EN 29002 and EN 46002 for in vitro diagnostic medical devices EN 980: 1996/ A1:1999 Graphical symbols for use in the labelling of medical devices C 319 of 2001-11-14 C 227 of 1999-08-10 C 182 of 2002-07-31 C 182 of 2002-07-31 C 182 of 2002-07-31 C 182 of 2002-07-31 Publication OJ
C 182 of 2002-07-31
EN 376: 2002
C 182 of 2002-07-31
EN 455-1: 2000
C 319 of 2001-11-14
EN 455-2: 2000 EN 552: 1994/ A2:2000
C 182 of 2002-07-31
C 182 of 2002-07-31
Guide on IVD Directive
EuropeAid /114385/D/SV/CY
EN 980: 1996/ A2:2001 EN 1280-1: 1997/ A1:2000 EN 1658: 1996 EN ISO 4074: 2002 EN ISO 4135: 2001
Graphical symbols for use in the labelling of medical devices Agent specific filling systems for anaesthetic vaporisers - Part 1: Rectangular keyed filling systems Requirements for marking of in vitro diagnostic instruments Natural latex rubber condoms Requirements and test methods (ISO 4074:2002) Anaesthetic and respiratory equipment - Vocabulary (ISO 4135:2001) Anaesthesic and respiratory equipment Heat and moisture
C 182 of 2002-07-31
C 182 of 2002-07-31 C 227 of 1999-08-10 C 182 of 2002-07-31
C 182 of 2002-07-31
EN ISO 9360-1: 2000
exhangers (HMEs) for humidifying respired gases in humans Part 1: HMEs for use with minimum tidal volumes of 250 ml (ISO 93601:2000)
C 319 of 2001-11-14
EN ISO 10993-8: 2001
Biological evaluation of medical devices Part 8: Selection and qualification of reference materials for biological tests (ISO 109938:2000) Biological evaluation of medical devices - Part 14: Identification and quantification of degradation products from ceramics (ISO 1099314:2001) Biological evaluation of medical devices Part 15: Identification and quantification of degradation products from metals and alloys (ISO 10993-15:2000) Non-active surgical implants Body contouring implants Specific C 182 of 2002-07-31 C 319 of 2001-11-14 C 182 of 2002-07-31 C 182 of 2002-07-31
EN ISO 10993-14: 2001
EN ISO 10993-15: 2000
EN 12180: 2000
requirements for mammary implants
In vitro diagnostic medical devices Measurement of quantities in EN 12286: 1998 samples of biological origin Presentation of reference measurement procedures EN 12286: 1998/ A1:2000 In vitro diagnostic medical devices Measurement of quantities in samples of biological origin Presentation of reference measurement procedures In vitro diagnostic medical devices Measurement of quantities in samples of biological origin Description of reference materials In vitro diagnostic medical devices Culture media for microbiology Performance criteria for culture media In vitro diagnostic medical devices Culture media for microbiology Performance criteria for culture media C 293 of 2000-10-14 C 319 of 2001-11-14 C 227 of 1999-08-10
EN 12287: 1999
EN 12322: 1999 EN 12322: 1999/ A1:2001 EN 12718: 2001 corrigendum ENV 12719: 2001 corrigendum EN 13328-1: 2001
C 288 of 1999-10-09
C 182 of 2002-07-31
Breathing system filters for anaesthetic and respiratory use Part 1:
C 182 of 2002-07-31
Guide on IVD Directive
EuropeAid /114385/D/SV/CY
Test method to assess filtration performance Quality systems - Medical devices - Particular requirements for the EN 13485: 2000 application of EN ISO 9001:1994 (revision of EN 46001:1996) C 182 of 2002-07-31
Quality systems Medical devices - Particular requirements for the EN 13488: 2000 application of EN ISO 9002:1994 (revision of EN 46002 :1996)
C 182 of 2002-07-31
EN 13532: 2002
General requirements for in vitro diagnostic medical devices for selftesting Respiratory therapy equipment - Part 1: Nebulising systems and their components Respiratory therapy equipment - Part 3: Air entrainment devices Performance evaluation of in vitro diagnostic medical devices Stability testing of in vitro diagnostic medical devices Elimination or reduction of risk of infection related to in vitro diagnostic reagents Sampling procedures used for acceptance testing of in vitro diagnostic medical devices - Statistical aspects Sterilisation of health care products - General requirements for
C 314 of 2002-12-17
EN 13544-1: 2001 EN 13544-3: 2001 EN 13612: 2002 EN 13640: 2002 EN 13641: 2002
C 182 of 2002-07-31 C 182 of 2002-07-31 C 314 of 2002-12-17 C 314 of 2002-12-17 C 314 of 2002-12-17
EN 13975:2003
C 280 of 2003-11-21
EN ISO 14937: 2000
characterisation of a sterilising agent and the development, validation and routine control of a sterilisation process for medical devices (ISO 14937:2000) Medical devices Application of risk management to medical
C 182 of 2002-07-31
EN ISO 14971: 2000
devices (ISO 14971:2000)
C 182 of 2002-07-31
In vitro diagnostic medical devices - Measurement of quantities in EN ISO 18153:2003 biological samples - Metrological traceability of values for catalytic concentration of enzymes assigned to calibrators and control materials (ISO 18153:2003) EN ISO 15225: 2000 Nomenclature - Specification for a nomenclature system for medical devices for the purpose of regulatory data exchange (ISO 15225:2000) Safety requirements for electrical equipment for measurement, EN 61010-2-101: 2002 control, and laboratory use - Part 2-101: Particular requirements for in vitro diagnostic (IVD) medical equipment Reference document: IEC 61010-2-101:2002 (Modified) C 182 of 2002-07-31 C 280 of 2003-11-21
Guide on IVD Directive
EuropeAid /114385/D/SV/CY
Note 1: Generally the date of cessation of presumption of conformity will be the date of withdrawal ("dow"), set by the European Standardisation Organisation, but attention of users of these standards is drawn to the fact that in certain exceptional cases this can be otherwise.
FURTHER INFORMATION
Information on medical devices directives, harmonised standards, competent authorities notified bodies, etc. may be obtained from the web site of the European Commission at; http://europa.eu.int/comm/enterprise/medical_devices/index.htm
For information on all New Approach Directives, try: http://www.newapproach.org/
Consolidated texts of the Directives may be downloaded from: http://www.europa.eu.int/eur-lex/en/consleg/index1.html
STANDARDS
Further information on standards and guidance information can be obtained from the Standards Body of Cyprus: CYS Cyprus Organisation for the Promotion of Quality At the Ministry of Commerce, Industry and Tourism 13-15, Andreas Araouzou Street 1421 Nicosia http://www.cys.mcit.gov.cy
USEFUL LINKS
Further information and links to all relevant European websites are available at
www.cys.mcit.gov.cy
under the button named:
NEW APPROACH
Guide on IVD Directive
EuropeAid /114385/D/SV/CY
CYPRUS COMPETENT AUTHORITY
In Cyprus the Ministry of Health is responsible for enforcement of the medical devices regulations. Further details on enforcement and penalties are available at Cyprus Contact point at the
Ministry of Heath Contact: Mary Avraamidou mavraamidou@mphs.moh.gov.cy Stelios Christofides mpd.mh@cytanet.com.cy
CYPRUS LEGISLATION
General requirements and stipulation are layed down in the Cyprus Framework Law N. 30(I)/2002 (published 5.4.2002) as amended by Law N. 29(I)/2003 (published 28.3.2003). The Framework Law represents the legal basis for the Cyprus Regulations addressing respective Directives. The Cyprus Regulation addressing the IVDD (In vitro diagnostic medical devices) directive is implemented as Regulation P.I. 597/2003 (published 18.7.2003). Furthermore, the MDD directive is Regulation P.I. 598/2003 (published 18.7.2003) and the AIMD directive is Regulation P.I. 599/2003 (published 18.7.2003).
Potrebbero piacerti anche
- Analyzing Sri Lankan Ceramic IndustryDocumento18 pagineAnalyzing Sri Lankan Ceramic Industryrasithapradeep50% (4)
- Employee Conduct Policy StpsoDocumento57 pagineEmployee Conduct Policy StpsoWWLTVWebteamNessuna valutazione finora
- Links To GoodnessDocumento194 pagineLinks To GoodnessKevin Itwaru0% (3)
- BS 7346-6-2005 Specifications For Cable SystemsDocumento26 pagineBS 7346-6-2005 Specifications For Cable SystemsFathyNessuna valutazione finora
- Medical Devices and IVDs: Fit for the new EU-Regulations: Your complete seminar for projekt, study and jobDa EverandMedical Devices and IVDs: Fit for the new EU-Regulations: Your complete seminar for projekt, study and jobNessuna valutazione finora
- The Survival Guide to EU Medical Device RegulationsDa EverandThe Survival Guide to EU Medical Device RegulationsValutazione: 5 su 5 stelle5/5 (1)
- PMS PMCF CER RelationshipDocumento1 paginaPMS PMCF CER RelationshipMohammed HammedNessuna valutazione finora
- MDSAP AU P0002.004 Audit Model PDFDocumento81 pagineMDSAP AU P0002.004 Audit Model PDFCarolina MolfinoNessuna valutazione finora
- The Definitive Guide To ISO 14971 Risk Management For Medical DevicesDocumento31 pagineThe Definitive Guide To ISO 14971 Risk Management For Medical DevicesMichael FadjarNessuna valutazione finora
- FDA Guidance - 510 K ChecklistDocumento3 pagineFDA Guidance - 510 K ChecklistHila Cohen100% (3)
- WP.026 Leveraging Post Market Surveillance and Post Market Clinical Follow Up Data - A01Documento20 pagineWP.026 Leveraging Post Market Surveillance and Post Market Clinical Follow Up Data - A01Neelakandan R100% (1)
- Managing Medical Devices within a Regulatory FrameworkDa EverandManaging Medical Devices within a Regulatory FrameworkBeth Ann FiedlerValutazione: 5 su 5 stelle5/5 (1)
- Eu MDRDocumento34 pagineEu MDRgobu269104100% (1)
- Bsi Smart Support Post Market SurveillanceDocumento10 pagineBsi Smart Support Post Market SurveillanceMauro CostaNessuna valutazione finora
- GreenLight Guru Supply Chain Management Under MDR Brexit Swixit - MedEnvoyDocumento24 pagineGreenLight Guru Supply Chain Management Under MDR Brexit Swixit - MedEnvoyliesbeth albertsNessuna valutazione finora
- Bsi VigilanceDocumento19 pagineBsi VigilanceHazalNessuna valutazione finora
- MDR Namsa SlidesDocumento44 pagineMDR Namsa SlidesswarhiliNessuna valutazione finora
- Improving Technical Dcumentation - Medical DevicesDocumento53 pagineImproving Technical Dcumentation - Medical Devicesrev2000Nessuna valutazione finora
- Europe IVDR Regulatory Process EMERGODocumento1 paginaEurope IVDR Regulatory Process EMERGOHarold Braustein100% (1)
- IVD Medical Device V2Documento44 pagineIVD Medical Device V2Vadi VelanNessuna valutazione finora
- GG Guide To Risk ManagementDocumento43 pagineGG Guide To Risk ManagementSamrat SinghaNessuna valutazione finora
- Medical Device Testing GuideDocumento33 pagineMedical Device Testing GuideRoxanaBurla100% (1)
- ISO 9001 13485 and FDA QSR CompareDocumento71 pagineISO 9001 13485 and FDA QSR CompareNoorm MENessuna valutazione finora
- Technical Documentation and Medical Device RegulationDocumento15 pagineTechnical Documentation and Medical Device Regulationzfo302Nessuna valutazione finora
- Post Market Surveillance: Global Guidance For Adverse Event Reporting For Medical DevicesDocumento37 paginePost Market Surveillance: Global Guidance For Adverse Event Reporting For Medical DevicesSachin KumarNessuna valutazione finora
- Medical Device Validation RadhaDocumento27 pagineMedical Device Validation Radhas1a2d3f40% (1)
- Med-Info: Biological EvaluationDocumento4 pagineMed-Info: Biological EvaluationRand OmNessuna valutazione finora
- Europe IVD Regulatory ProcessDocumento1 paginaEurope IVD Regulatory Processpsp710Nessuna valutazione finora
- Special and Abbreviated 510 (K)Documento18 pagineSpecial and Abbreviated 510 (K)hemkenbpNessuna valutazione finora
- European Medical Device Usability RequirementsDocumento3 pagineEuropean Medical Device Usability RequirementsHong-Nam KimNessuna valutazione finora
- Comparison Chart of ISO 13485 and FDA QSR RequirementsDocumento4 pagineComparison Chart of ISO 13485 and FDA QSR RequirementsjvivoloNessuna valutazione finora
- TUV Sud Process Validation in Medical DevicesDocumento16 pagineTUV Sud Process Validation in Medical DevicesShane Shine100% (1)
- WW ISO10993 Biocompatibility White Paper EMERGODocumento20 pagineWW ISO10993 Biocompatibility White Paper EMERGOJezreel ZaragosaNessuna valutazione finora
- Validating Medical Device Packaging FinalDocumento9 pagineValidating Medical Device Packaging FinalSaoodi AlkatheriNessuna valutazione finora
- Compliance With 21 CFR 820 and Iso 13485 Using MastercontrolDocumento6 pagineCompliance With 21 CFR 820 and Iso 13485 Using MastercontrolAmit Singh100% (1)
- A Step-By-Step Guide To Determine How Your Medical Device Will Be ClassifiedDocumento18 pagineA Step-By-Step Guide To Determine How Your Medical Device Will Be ClassifiedRajeshNessuna valutazione finora
- Medical Device Regulatory Process UsaDocumento1 paginaMedical Device Regulatory Process UsaFlu Plant0% (1)
- Comparing Medical Device Law & Reg of Japan, US, and EUDocumento5 pagineComparing Medical Device Law & Reg of Japan, US, and EUpavan_baggaNessuna valutazione finora
- EU US PostMarket Surveillance Whitepaper PDFDocumento7 pagineEU US PostMarket Surveillance Whitepaper PDFLakshmana Perumal RamarajNessuna valutazione finora
- Leitfaden Fuer App Entwickler enDocumento65 pagineLeitfaden Fuer App Entwickler enloipoilNessuna valutazione finora
- Ivdr Classification Avril AylwardDocumento19 pagineIvdr Classification Avril AylwardTuấn Minh PhùngNessuna valutazione finora
- An Introductory Guide To The Medical Device Regulation (MDR) and The in Vitro Diagnostic Medical Device Regulation (IVDR)Documento27 pagineAn Introductory Guide To The Medical Device Regulation (MDR) and The in Vitro Diagnostic Medical Device Regulation (IVDR)Shivaraj K Yadav100% (1)
- Syringe Tech. File ChecklistDocumento3 pagineSyringe Tech. File ChecklistdearistyaNessuna valutazione finora
- Essential Requirement Checksheet - FC161Documento14 pagineEssential Requirement Checksheet - FC161Jackeline PeredaNessuna valutazione finora
- Pharma Quality WorkDocumento26 paginePharma Quality WorkBhagwan YadavNessuna valutazione finora
- Bsi MD Risk Management For Medical Devices Webinar 131119 Uk enDocumento29 pagineBsi MD Risk Management For Medical Devices Webinar 131119 Uk enrakesh marwahNessuna valutazione finora
- ISO 14971 RISK Management For Medical Devices: The Definitive GuideDocumento41 pagineISO 14971 RISK Management For Medical Devices: The Definitive GuideOrielson Cruz100% (2)
- Schmitt Regulatory Handbook Final Jan 2015Documento9 pagineSchmitt Regulatory Handbook Final Jan 2015param540Nessuna valutazione finora
- RAC EU Exam Content OutlineDocumento3 pagineRAC EU Exam Content OutlineSenthil ThyagarajanNessuna valutazione finora
- BEP Sophie-MichelDocumento37 pagineBEP Sophie-MichelRand OmNessuna valutazione finora
- Biocompatability, FDA, IsO 10993, IVDDocumento31 pagineBiocompatability, FDA, IsO 10993, IVDsam3557Nessuna valutazione finora
- White Paper - IsO 13485Documento14 pagineWhite Paper - IsO 13485Gisela Cristina MendesNessuna valutazione finora
- Medical Device Software DevelopmentDocumento26 pagineMedical Device Software DevelopmentAdalnei GomideNessuna valutazione finora
- 2019 RAC Candidate Guide EUDocumento22 pagine2019 RAC Candidate Guide EURonak choksiNessuna valutazione finora
- Bio Compatibility PerformanceDocumento60 pagineBio Compatibility PerformanceJorgeMechanicNessuna valutazione finora
- BSI MD Consultants Day Usability and Human Factors Presentation UK ENDocumento38 pagineBSI MD Consultants Day Usability and Human Factors Presentation UK ENAngel LaraNessuna valutazione finora
- GDP For Medical DevicesDocumento33 pagineGDP For Medical DevicesВладимир Бургић50% (2)
- Db10g Rac Exam Study Guide 320804Documento5 pagineDb10g Rac Exam Study Guide 320804Anand ShankarNessuna valutazione finora
- Acceptance Sampling Update - AQL LTPDDocumento13 pagineAcceptance Sampling Update - AQL LTPDBrandon YOU100% (1)
- 06 MDCG 2020-6 Guidance On Sufficient Clinical Evidence For Legacy DevicesDocumento22 pagine06 MDCG 2020-6 Guidance On Sufficient Clinical Evidence For Legacy DevicesSantiago IbañezNessuna valutazione finora
- 02 IVD-R Deep-Dive Deck NewDocumento215 pagine02 IVD-R Deep-Dive Deck NewrajiveacharyaNessuna valutazione finora
- RAC Review Chapter 10Documento35 pagineRAC Review Chapter 10Ayele BizunehNessuna valutazione finora
- Plasticizers For CPE ElastomersDocumento8 paginePlasticizers For CPE Elastomersbatur42Nessuna valutazione finora
- Intro To Forensic Final Project CompilationDocumento15 pagineIntro To Forensic Final Project Compilationapi-282483815Nessuna valutazione finora
- GTAG 1 2nd EditionDocumento36 pagineGTAG 1 2nd EditionChristen Castillo100% (2)
- TCRP Wheel Rail Noise ReportDocumento220 pagineTCRP Wheel Rail Noise ReportAnonymous OmwbxkB100% (1)
- Paper - 1: Principles & Practice of Accounting Questions True and FalseDocumento29 paginePaper - 1: Principles & Practice of Accounting Questions True and FalsePiyush GoyalNessuna valutazione finora
- 20779A ENU CompanionDocumento86 pagine20779A ENU Companionmiamikk204Nessuna valutazione finora
- QuartzDocumento5 pagineQuartzKannaTaniyaNessuna valutazione finora
- General Director AdDocumento1 paginaGeneral Director Adapi-690640369Nessuna valutazione finora
- Appraisal: From Theory To Practice: Stefano Stanghellini Pierluigi Morano Marta Bottero Alessandra Oppio EditorsDocumento342 pagineAppraisal: From Theory To Practice: Stefano Stanghellini Pierluigi Morano Marta Bottero Alessandra Oppio EditorsMohamed MoussaNessuna valutazione finora
- In The High Court of Delhi at New DelhiDocumento3 pagineIn The High Court of Delhi at New DelhiSundaram OjhaNessuna valutazione finora
- Artificial Intelligence and Parametric Construction Cost Estimate Modeling State-of-The-Art ReviewDocumento31 pagineArtificial Intelligence and Parametric Construction Cost Estimate Modeling State-of-The-Art ReviewmrvictormrrrNessuna valutazione finora
- Why Is Inventory Turnover Important?: ... It Measures How Hard Your Inventory Investment Is WorkingDocumento6 pagineWhy Is Inventory Turnover Important?: ... It Measures How Hard Your Inventory Investment Is WorkingabhiNessuna valutazione finora
- PDS Air CompressorDocumento1 paginaPDS Air Compressordhavalesh1Nessuna valutazione finora
- Slope Stability Analysis Using FlacDocumento17 pagineSlope Stability Analysis Using FlacSudarshan Barole100% (1)
- Gabuyer Oct13Documento72 pagineGabuyer Oct13William Rios0% (1)
- 1.2 Server Operating SystemDocumento20 pagine1.2 Server Operating SystemAzhar AhmadNessuna valutazione finora
- Expanded Breastfeeding ActDocumento9 pagineExpanded Breastfeeding ActJohn Michael CamposNessuna valutazione finora
- Method Statement: Vetotop XT539Documento4 pagineMethod Statement: Vetotop XT539محمد عزتNessuna valutazione finora
- Taiwan Petroleum Facilities (1945)Documento85 pagineTaiwan Petroleum Facilities (1945)CAP History LibraryNessuna valutazione finora
- Example of Praxis TicketDocumento3 pagineExample of Praxis TicketEmily LescatreNessuna valutazione finora
- Project SummaryDocumento59 pagineProject SummarynaseebNessuna valutazione finora
- Siemens Power Engineering Guide 7E 223Documento1 paginaSiemens Power Engineering Guide 7E 223mydearteacherNessuna valutazione finora
- Maturity AssessmentDocumento228 pagineMaturity AssessmentAli ZafarNessuna valutazione finora
- S No. Store Type Parent ID Partner IDDocumento8 pagineS No. Store Type Parent ID Partner IDabhishek palNessuna valutazione finora
- Exercises Service CostingDocumento2 pagineExercises Service Costingashikin dzulNessuna valutazione finora
- KR 10 Scara: The Compact Robot For Low PayloadsDocumento4 pagineKR 10 Scara: The Compact Robot For Low PayloadsBogdan123Nessuna valutazione finora