Documenti di Didattica
Documenti di Professioni
Documenti di Cultura
RNTCP Launches DOTS Plus Treatment
Caricato da
Dinesh KumarDescrizione originale:
Copyright
Formati disponibili
Condividi questo documento
Condividi o incorpora il documento
Hai trovato utile questo documento?
Questo contenuto è inappropriato?
Segnala questo documentoCopyright:
Formati disponibili
RNTCP Launches DOTS Plus Treatment
Caricato da
Dinesh KumarCopyright:
Formati disponibili
RNTCP Launches Cat IV (DOTS Plus) treatment for Multi-Drug Resistant TB
Multi-drug resistant Tuberculosis (MDR-TB) is defined as resistance of Mycobacterium tuberculosis to Rifampiocin and Isoniazid, two of the most effective anti-tubercular drugs for treatment of Tuberculosis. Drug resistant tuberculosis has frequently been encountered in India and its presence has been known virtually from the time anti-tubercular drugs were introduced for the treatment of TB. There have been a number of reports on drug resistance in India including state level Drug resistance surveillance (DRS) surveys conducted in Gujarat and Maharashtra. Data from these studies have found MDR-TB levels of about 3% in new cases and 12%-17% in re-treatment cases. Although the level of MDR-TB in the community is low in relation to percentages and proportions it translates into large absolute numbers. To address this issue the Revised National Tuberculosis Control Programme (RNTCP) has initiated the DOTS Plus strategy for appropriate management of MDR TB patients and to prevent the propagation and dissemination of MDR-TB. The RNTCP views the treatment of MDR-TB patients as a standard of care issue. Recognizing that the diagnosis and treatment of MDR-TB cases is complex, RNTCP has developed national guidelines based on the WHO recommended international DOTS-Plus guidelines. These guidelines promote full integration of DOTS and DOTS-Plus activities under the RNTCP, so that patients with MDR-TB are both correctly identified and properly managed under the recommendations set out in this document. As per the DOTS Plus strategy the diagnosis of MDR-TB will be made at the Intermediate Reference Laboratories (IRLs) accredited to perform culture and Drug Sensitivity Testing (DST). RNTCP has initiated the establishment of the laboratory network in a phased manner in all the states across the country with support from the four National reference Laboratories (TRC, Chennai; NTI, Bangalore; LRS Institute, New Delhi and JALMA, Agra). After diagnosis the treatment of MDR-TB patients is initiated at designated DOTS Plus sites which are established in tertiary care centres (like Medical Colleges, Large speciality hospitals) at least one in each state. The DOTS Plus sites have qualified staff available to manage patients, using standardized Cat IV regimen, using the second-line drugs, given under daily Directly Observed Treatment (DOT) and standardized follow-up protocols, have systems in place to deliver ambulatory DOT after an initial short period of in-patient care to stabilise the patient on the second-line drug regimen, and with a logistics system and standardized information system in place. At the end of 2008 the DOTS Plus services are available in 7 states. Gujarat has been the first state to initiate DOTS Plus services in August 2007 followed by Maharashtra, Andhra Pradesh, Haryana Delhi,Kerala and West Bengal. It is planned to make available DOTS Plus services in all states by 2010 with complete geographical coverage by 2012.
Status of DOTS Plus activities at the end of 4Q2008
State Gujarat Maharashtra Andhra Pradesh Haryana Delhi West Bengal Kerala Date of Initiation August 2007 September 2007 October 2008 December 2008 December 2008 December 2008 December 2008 No. of MDR patients on treatment 131 84 25 6 3 1 3
Potrebbero piacerti anche
- The Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeDa EverandThe Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeValutazione: 4 su 5 stelle4/5 (5794)
- Rifampin ResistanceDocumento4 pagineRifampin ResistanceHeldrian dwinanda SuyuthieNessuna valutazione finora
- The Little Book of Hygge: Danish Secrets to Happy LivingDa EverandThe Little Book of Hygge: Danish Secrets to Happy LivingValutazione: 3.5 su 5 stelle3.5/5 (399)
- Identification and Classification According To Bergy's Manual of Systemic Bacteriology 2Documento46 pagineIdentification and Classification According To Bergy's Manual of Systemic Bacteriology 2Cezar Alexander GuevaraNessuna valutazione finora
- A Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryDa EverandA Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryValutazione: 3.5 su 5 stelle3.5/5 (231)
- Guidelines Updates - Plab 1 Keys-1Documento2 pagineGuidelines Updates - Plab 1 Keys-1Cynthia GNessuna valutazione finora
- Hidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceDa EverandHidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceValutazione: 4 su 5 stelle4/5 (894)
- Analisis Pelayanan Terapeutik Pada Pasien Tuberkulosis Resisten Obat Di Kota MedanDocumento14 pagineAnalisis Pelayanan Terapeutik Pada Pasien Tuberkulosis Resisten Obat Di Kota MedanwahyuNessuna valutazione finora
- The Yellow House: A Memoir (2019 National Book Award Winner)Da EverandThe Yellow House: A Memoir (2019 National Book Award Winner)Valutazione: 4 su 5 stelle4/5 (98)
- Strategi Nasional Pengobatan TB RO DR - Endang LukitosariDocumento24 pagineStrategi Nasional Pengobatan TB RO DR - Endang LukitosariGadis BangsawanNessuna valutazione finora
- Shoe Dog: A Memoir by the Creator of NikeDa EverandShoe Dog: A Memoir by the Creator of NikeValutazione: 4.5 su 5 stelle4.5/5 (537)
- Faktor Faktor Yang Mempengaruhi Kejadian TB Paru Dan Upaya Penanggulangannya - Edza Aria Wikurendra, S.KL, M.KL PDFDocumento12 pagineFaktor Faktor Yang Mempengaruhi Kejadian TB Paru Dan Upaya Penanggulangannya - Edza Aria Wikurendra, S.KL, M.KL PDFsalsa nNessuna valutazione finora
- Elon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureDa EverandElon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureValutazione: 4.5 su 5 stelle4.5/5 (474)
- Tuberculosis - Wikipedia, The Free EncyclopediaDocumento15 pagineTuberculosis - Wikipedia, The Free EncyclopediaitsmepranielNessuna valutazione finora
- Never Split the Difference: Negotiating As If Your Life Depended On ItDa EverandNever Split the Difference: Negotiating As If Your Life Depended On ItValutazione: 4.5 su 5 stelle4.5/5 (838)
- The Paleopathology of Specific Infectious Diseases From Southeastern Hungary: A Brief OverviewDocumento6 pagineThe Paleopathology of Specific Infectious Diseases From Southeastern Hungary: A Brief OverviewLucía Chavarría RíosNessuna valutazione finora
- Grit: The Power of Passion and PerseveranceDa EverandGrit: The Power of Passion and PerseveranceValutazione: 4 su 5 stelle4/5 (587)
- List TB Screening ClinicsDocumento2 pagineList TB Screening ClinicsArvind K VinayakNessuna valutazione finora
- Devil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaDa EverandDevil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaValutazione: 4.5 su 5 stelle4.5/5 (265)
- ID Gambaran Pemeriksaan Makroskopis Urin PaDocumento7 pagineID Gambaran Pemeriksaan Makroskopis Urin PahukamaNessuna valutazione finora
- Microbiology - 18Documento7 pagineMicrobiology - 18karmylle andradeNessuna valutazione finora
- Pertussis: Dr. Turki Bukhari Pediatric R2 at PSMMCDocumento23 paginePertussis: Dr. Turki Bukhari Pediatric R2 at PSMMCnarNessuna valutazione finora
- The Emperor of All Maladies: A Biography of CancerDa EverandThe Emperor of All Maladies: A Biography of CancerValutazione: 4.5 su 5 stelle4.5/5 (271)
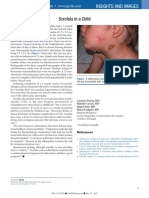
- Scrofula in A Child: Insights and ImagesDocumento1 paginaScrofula in A Child: Insights and Imagesmawarni siahaanNessuna valutazione finora
- On Fire: The (Burning) Case for a Green New DealDa EverandOn Fire: The (Burning) Case for a Green New DealValutazione: 4 su 5 stelle4/5 (73)
- Indonesia's National Guidelines for Tuberculosis Control (2011Documento111 pagineIndonesia's National Guidelines for Tuberculosis Control (2011widia mujtiawatiNessuna valutazione finora
- The Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersDa EverandThe Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersValutazione: 4.5 su 5 stelle4.5/5 (344)
- Republic of The Philippines Province of Batangas Municipality of San JoseDocumento3 pagineRepublic of The Philippines Province of Batangas Municipality of San JoseSample BakeshopNessuna valutazione finora
- Team of Rivals: The Political Genius of Abraham LincolnDa EverandTeam of Rivals: The Political Genius of Abraham LincolnValutazione: 4.5 su 5 stelle4.5/5 (234)
- CHN Ii ... Aparna Roll No. 12Documento17 pagineCHN Ii ... Aparna Roll No. 12Aparna PandeyNessuna valutazione finora
- Cutaneous Manifestations of TuberculosisDocumento35 pagineCutaneous Manifestations of TuberculosisSaid Plazola MercadoNessuna valutazione finora
- Kemoterapi Lepra: Bag. Farmakologi Dan Terapi FK Undip 2017Documento24 pagineKemoterapi Lepra: Bag. Farmakologi Dan Terapi FK Undip 2017Yeni HerawatiNessuna valutazione finora
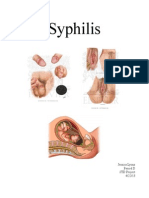
- Syphilis: Jessica Lyons Period D STD Project 4/22/13Documento3 pagineSyphilis: Jessica Lyons Period D STD Project 4/22/13jesssielynnn33Nessuna valutazione finora
- What was the Black DeathDocumento1 paginaWhat was the Black DeathNADA TAZKIA PURBANessuna valutazione finora
- The Unwinding: An Inner History of the New AmericaDa EverandThe Unwinding: An Inner History of the New AmericaValutazione: 4 su 5 stelle4/5 (45)
- Curs 14 Boala Lyme 2Documento63 pagineCurs 14 Boala Lyme 2n1u1s1h1100% (1)
- The World Is Flat 3.0: A Brief History of the Twenty-first CenturyDa EverandThe World Is Flat 3.0: A Brief History of the Twenty-first CenturyValutazione: 3.5 su 5 stelle3.5/5 (2219)
- TUBERCULOSISDocumento25 pagineTUBERCULOSISManuel Fatima Goncalves83% (6)
- Bordetella Pertussis: Dr. M. NoofeliDocumento50 pagineBordetella Pertussis: Dr. M. NoofeliJlizi MalekNessuna valutazione finora
- Tuberculosis of The Central Nervous System-Pathogenesis, Imaging, and ManagementDocumento615 pagineTuberculosis of The Central Nervous System-Pathogenesis, Imaging, and ManagementRodrigo Uribe PachecoNessuna valutazione finora
- The Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreDa EverandThe Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreValutazione: 4 su 5 stelle4/5 (1090)
- Preparasi Radiofarmaka Tc-Etambutol Untuk Deteksi TuberkulosisDocumento6 paginePreparasi Radiofarmaka Tc-Etambutol Untuk Deteksi TuberkulosisNad PtrNessuna valutazione finora
- Hubungan Pengetahuan Pencegahan TB dengan Perilaku Pencegahan pada Kepala KeluargaDocumento9 pagineHubungan Pengetahuan Pencegahan TB dengan Perilaku Pencegahan pada Kepala KeluargaRindy PrawitaNessuna valutazione finora
- TB Overview FactsheetDocumento3 pagineTB Overview FactsheetGavinNessuna valutazione finora
- STD Chapter on Gonorrhea, Chlamydia & TreatmentDocumento1 paginaSTD Chapter on Gonorrhea, Chlamydia & TreatmentJohn OmandacNessuna valutazione finora
- The Sympathizer: A Novel (Pulitzer Prize for Fiction)Da EverandThe Sympathizer: A Novel (Pulitzer Prize for Fiction)Valutazione: 4.5 su 5 stelle4.5/5 (119)
- TuberculosisDocumento16 pagineTuberculosisjojolim18100% (1)
- 120 1 312 1 10 20170109Documento4 pagine120 1 312 1 10 20170109Riza TamaNessuna valutazione finora
- Her Body and Other Parties: StoriesDa EverandHer Body and Other Parties: StoriesValutazione: 4 su 5 stelle4/5 (821)