Documenti di Didattica
Documenti di Professioni
Documenti di Cultura
Bifrontal Basal Inter Hemispheric Approach To
Caricato da
Carlos CasalloDescrizione originale:
Copyright
Formati disponibili
Condividi questo documento
Condividi o incorpora il documento
Hai trovato utile questo documento?
Questo contenuto è inappropriato?
Segnala questo documentoCopyright:
Formati disponibili
Bifrontal Basal Inter Hemispheric Approach To
Caricato da
Carlos CasalloCopyright:
Formati disponibili
J Neurosurg 84:951–956, 1996
Bifrontal basal interhemispheric approach to
craniopharyngioma resection with or without division of
the anterior communicating artery
MASATO SHIBUYA, M.D., MASAKAZU TAKAYASU, M.D., YOSHIO SUZUKI, M.D.,
KIYOSHI SAITO, M.D., AND KENICHIRO SUGITA, M.D.
Department of Neurosurgery, Nagoya University School of Medicine, Nagoya, Japan
U The authors describe the use of a bifrontal basal interhemispheric approach with or without division of the anteri-
or communicating artery (ACoA) for removal of large craniopharyngiomas. This approach is a more basal modifica-
tion of the anterior interhemispheric approach, allowing preservation of most bridging veins. Since 1988, 22 patients
underwent operations using this approach to achieve total or near-total excision of large craniopharyngiomas. Division
of the ACoA was performed in 11 of 17 patients with retrochiasmatic tumors with no early or late complications relat-
ed to division of the artery. There were no operative mortalities. Visual improvement (59%) and preservation of the
pituitary stalk (64%) were achieved in a high percentage of patients. Preservation of the pituitary stalk correlated well
with postoperative pituitary function. The bifrontal basal interhemispheric approach allowed a bilateral, wider opera-
tive field with better orientation and views of important neural structures and perforating arteries without requiring
combination with other approaches. When the ACoA limited operative exposure, the artery could be divided safely.
The authors discuss indications for, and advantages of, the bifrontal basal interhemispheric approach with or without
division of the ACoA in the removal of large craniopharyngiomas.
KEY WORDS • anterior communicating artery • craniopharyngioma • pituitary stalk •
interhemispheric approach • surgical approach
LTHOUGH controversy still exists regarding the opti- vides additional space. In the present report we discuss
A mum treatment of craniopharyngiomas, all neuro-
surgeons agree that total removal of the tumor is
desirable if it can be done safely.1,4,7,15,16 However, suc-
indications for, and advantages of, using the bifrontal
basal interhemispheric approach with or without division
of the ACoA in the removal of large craniopharyngiomas.
cessful radical excision of craniopharyngioma remains
one of the most difficult procedures in the era of micro-
surgery. Surgeons use several different approaches, de- Clinical Material and Methods
pending on the location and size of the tumor as well as on Since 1988, 22 patients underwent operations using the
the surgeon’s preference. These include transsphenoidal, bifrontal basal interhemispheric approach to achieve total
pterional, transcortical–transventricular, subfrontal, sub- or near-total excision of large craniopharyngiomas. These
temporal, transtemporal, anterior interhemispheric, and patients ranged in age from 2 to 64 years; there were 12
anterior transcallosal approaches, which are used alone or females and 10 males. The presenting symptoms were
in combination.1,3,6,8,9,11,14–16 In our institution, a bifrontal visual disturbance in 18 patients, signs of hydrocephalus
basal interhemispheric approach has been used since 1988
primarily for operations involving large craniopharyn- in two, amenorrhea in one, and mental disturbance in two
giomas. This approach is a more basal modification of patients. The maximum diameter of the tumors evaluated
the anterior interhemispheric approach17 that has proved by magnetic resonance (MR) imaging or computerized to-
advantageous in the removal of large craniopharyn- mography (CT) scanning was 27 to 69 mm (average 41 6
giomas. It allows a bilateral, wider operative field with 12 mm, mean 6 standard deviation (SD)). Fourteen
better orientation and views of important neural structures tumors (64%) were larger than 40 mm. Five patients
and perforating arteries without requiring combination (23%) had prechiasmatic tumors and 17 patients (77%)
with other approaches. Most of the bridging veins can had retrochiasmatic tumors (Table 1). Bilateral carotid
be preserved using this method. However, occasionally angiography was performed preoperatively and the devel-
the anterior communicating artery (ACoA) may limit the opment of the ACoA and the A1 and A2 segments was
exposure. In such instances, division of the ACoA pro- carefully evaluated to ascertain whether the ACoA could
J. Neurosurg. / Volume 84 / June, 1996 951
M. Shibuya, et al.
TABLE 1 The olfactory nerves are dissected from the basal sur-
Characteristics and outcomes of 22 patients who underwent a face of the frontal lobes bilaterally all the way to the ol-
bifrontal basal interhemispheric approach with or factory trigone. This maneuver minimizes the risk of in-
without division of the ACoA* advertent damage to the nerves during retraction of the
frontal lobes. Both frontal lobes are retracted together
Characteristics Preserved ACoA Divided ACoA Total
& Outcomes (11 patients) (11 patients) (22 patients) with the superior sagittal sinus, and dissection of the inter-
hemispheric fissure is performed only from the basal side,
characteristics not from the convexity surface (Fig. 1 right). Therefore,
age (yrs) the extent of dissection in the area of the fissure is less
,18 4 5 9 (41%)
$18 7 6 13 (59%) than that caused by the conventional anterior approach
follow-up period (mos)† 35 6 23 31 6 15 33 6 19 and most of the bridging veins that drain into the superior
tumor size (mm)† 42 6 14 40 6 11 41 6 12 sagittal sinus are preserved. The arachnoid dissection is
tumor location directed toward the pre- and suprachiasmal region, expos-
prechiasmatic 5 0 5 (23%) ing the A2 segment bilaterally, the genu of the corpus cal-
retrochiasmatic 6 11 17 (77%)
losum, the ACoA, and the chiasm. Usually, the brain be-
outcomes comes sufficiently slack after removal of cerebrospinal
tumor recurrence 2 1 3 (14%)
visual acuity
fluid from the basal cisterns. In cases of prechiasmatic
improved 7 6 13 (59%) tumors, the ACoA is compressed and displaced supero-
unchanged 4 3 7 (32%) posteriorly and the artery usually does not affect the oper-
deteriorated 0 2 2 (9%) ative approach. On the other hand, in cases of retrochias-
mental problems matic tumors, the ACoA is located anterior to the tumor
preop 1 2 3 (14%)
postop 2 3 5 (23%)
and often limits operative exposure. Division of the
pituitary stalk ACoAs may be necessary for the removal of the latter type
preserved 8 6 14 (64%) of tumor.
severed 1 3 4 (18%) The lamina terminalis is opened in the midline. Internal
undetected 2 2 4 (18%) decompression is the next step toward removal of the tu-
* ACoA = anterior communicating artery. mor. The cyst is punctured and intracapsular removal of
† Values expressed are the mean 6 standard deviation. the tumor is done using a smooth or serrated suction tube
to extract the solid part of the tumor.13 The two-suction
method is useful for this purpose. Chemical meningitis is
prevented by avoiding spillage of cyst fluid into the sub-
be divided safely. The indication for division of the ACoA arachnoid space; this is accomplished using cottonoid
was determined in patients with retrochiasmatic tumors by coverage of the surrounding operative field as well as con-
weighing the importance of a wider exposure for total tinuous suction drainage through a small silastic tube
removal of the tumor with the disadvantage of sacrificing placed in the bottom of the operative field. Sufficient
the ACoA. internal decompression facilitates the following step of
Endocrine studies included preoperative and postopera- capsular dissection from adjacent vital structures such as
tive measurement of basal serum levels of adrenocorti- the hypothalamus.
cotropic hormone (ACTH), growth hormone (GH), and At this stage the surgeon must decide whether the
thyroid-stimulating hormone (TSH). Provocation testing ACoA should be divided. The artery is one of the main
was only performed in limited cases. Insulin (0.1 U/kg) structures limiting operative exposure of retrochiasmatic
and thyroid-releasing hormone (TRH; 500 U) were craniopharyngiomas in the anterior interhemispherical
administered intravenously for provocation testing of approach (Fig. 2 left). Although we always try to preserve
ACTH, GH, and TSH. The provocation test results were the artery, dividing it often provides a much wider operat-
judged as normal when the ACTH level increased to more ing space, which subsequently makes the operation easier
than twice the basal value, the GH level increased to and safer (Fig. 2 right). When the artery is left intact, it
more than 10 ng/ml, and the TSH level increased to more may inadvertently be damaged during retraction of the
than 10 mU/ml. hemispheres, especially in pediatric cases in which one
encounters thin-walled arteries. The ACoA is occluded
Operative Technique with small vascular clips and incised between the clips.
The patient is placed supine with the neck slightly The hypothalamic arteries must always be preserved, even
extended. A bicoronal skin incision is made behind the if they look small. The cut edges of the artery and the vas-
hairline and subperiosteal dissection of the skin flap is cular clips are covered with Biobond (glue)-soaked oxycel
extended to the glabella and the orbital ridges. The supra- to prevent slippage of the clips. Clipping and dividing of
orbital nerves are preserved. A bifrontal craniotomy is the ACoA may be difficult when it is too short and many
performed, which includes removal of the nasal part of the perforating arteries branch off from the artery. When one
frontal bone in a single continuous bone flap or separate side of the A1 segment is hypoplastic, it may be divided
flaps (Fig. 1 left). The frontal sinus is almost always instead of the short ACoA, although we have not had such
opened, the mucosa is pushed downward, and the inner a case.
table of the sinus and the crista galli are removed with The tumor usually adheres most tightly to the chiasm,
rongeurs or air drills. The falx and the superior sagittal pituitary stalk, or hypothalamus, whereas it usually sepa-
sinus are divided after double ligation. rates easily from the upper wall of the third ventricle. The
952 J. Neurosurg. / Volume 84 / June, 1996
Bifrontal basal interhemispheric approach to craniopharyngiomas
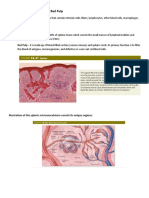
FIG. 1. Drawings displaying the bifrontal basal interhemispheric approach. Left: Skin incision and craniotomy. A
bifrontal craniotomy is performed, which includes removal of the nasal part of the frontal bone in separate bone flaps.
Right: The extent of dissection of the interhemispheric fissure. The dissection area of the fissure in this approach (A) is
much less than that created in the conventional anterior interhemispheric approach (B). Most of the bridging veins drain-
ing into the superior sagittal sinus are preserved in the bifrontal basal interhemispheric approach.
bilateral, wider exposure obtained by this approach en- obtained when the ACoA was cut between two Weck
ables the surgeon to view the lesion from many different clips. Additional parts of the tumor were removed by
angles and to locate the dissection plane between the means of scissors, bipolar cutting, and suction. The por-
tumor capsule and the important neural structures or per- tion of tumor that tightly adhered behind the optic chiasm
forating arteries on both sides. Consequently, there is a was dissected using a silver dissector under direct vision,
good chance of preserving the hypothalamus, pituitary which was obtained by changing the angle of the micro-
stalk, and perforating arteries using this approach. More- scope. Fortunately, there was a membrane separating the
over, the wider operative field reduces blind operative tumor from the perforating arteries, and the tumor could
procedures and facilitates complete tumor resection. be totally removed without disturbing these vessels (Fig.
3 right). The pituitary stalk was left intact.
Postoperative Course. Postoperatively, the patient ex-
Illustrative Case hibited diabetes insipidus and required hormone replace-
Examination. This 51-year-old man was referred to ment for a few months. By 6 months after the operation
our hospital for visual disturbance. His visual acuity was he was able to do well without hormone replacement.
20/100 on both sides. Goldmann’s perimetry showed a His vision improved markedly postoperatively (right eye,
binasal lower quadrantanopsia with central scotoma in the 20/40, left eye, 20/20) and his visual fields widened. He
lower half of both eyes. Gadolinium-enhanced MR imag- still has central scotoma in the lower halves of both eyes.
ing showed a well-enhanced multilobular suprasellar solid Although his olfaction decreased slightly postoperatively,
mass, 40 mm in diameter, in the third ventricle (Fig. 3 he experiences no difficulties in daily life.
left). The pituitary stalk was clearly shown in the midline
on the coronal image (Fig. 3 center).
Operation. The operation was performed using the bi-
frontal basal interhemispheric approach. When the inter-
hemispheric fissure was dissected, as described previous-
ly, the pituitary stalk could be clearly seen below the
chiasm. The lamina terminalis was bulging and was
opened in the midline, avoiding damage to the hypotha-
lamic arteries. A yellowish, granulomatous, solid tumor
appeared in the third ventricle. The tumor was tough and
could not be removed by suction using the usual smooth
tip. However, a serrated suction tip was effective in cut-
ting and removing the tumor. Fortunately, the tumor did
not adhere tightly to the wall of the third ventricle except
behind the optic chiasm and thus could be retracted infe-
riorly as it was removed little by little. Initially, the ACoA FIG. 2. Drawings depicting the operative view in a patient with
a retrochiasmatic craniopharyngioma before (left) and after (right)
was preserved; however, total removal of the tumor ap- division of the anterior communicating artery. The broken line
peared to be impossible without sacrificing the artery. The shows the extent of the tumor. Division of the artery provides a
ACoA was fenestrated with a median artery of the corpus much wider operating space, subsequently making the operation
callosum arising from it. A wide operative field was easier and safer.
J. Neurosurg. / Volume 84 / June, 1996 953
M. Shibuya, et al.
FIG. 3. Gadolinium-enhanced magnetic resonance images showing a retrochiasmatic craniopharyngioma in our illus-
trative case (left). The pituitary stalk is clearly shown in the midline on the coronal image (center). Postoperatively, total
removal of the tumor is shown (right).
Results tion between preservation of the pituitary stalk and basal
Patient follow-up periods in this study ranged from 4 to serum levels of pituitary hormones was investigated (Ta-
78 months (33 6 19 months, mean 6 SD) (Table 1). Total ble 2). Basal serum levels of ACTH and TSH were with-
or near-total removal of the tumors was confirmed in in a normal range postoperatively in approximately one-
all cases by postoperative CT or MR imaging studies. half of the patients whose pituitary stalks were preserved.
Division of the ACoAs was performed in 11 (65%) of 17 On the other hand, only one of the patients whose pituitary
patients with retrochiasmatic tumors and in none of the stalk was undetected or severed showed normal serum
five patients with prechiasmatic tumors. In a 2-year-old levels of ACTH and TSH postoperatively. Only six
boy, the ACoA was initially left intact but was lacerat- patients in this series underwent both preoperative and
ed during retraction of the frontal lobes and eventually postoperative provocation tests of ACTH, GH, and TSH.
divided. In this group the pituitary stalks were preserved in five
No operative mortality occurred. However, three late patients and severed in one. Provocation tests of GH se-
deaths occurred during the follow-up period. One patient cretion were negative both preoperatively and postopera-
died of acute subdural hematoma caused by a fall 5 years tively in all six patients, suggesting friability of the pi-
after the operation. Another man, who had preoperative tuitary–GH axis. On the other hand, ACTH and TSH
mental disturbance and postoperative deterioration, ran showed a normal or subnormal response to the provoca-
away from the hospital 8 months after operation and was tion tests preoperatively. Postoperative ACTH and TSH
found dead 1 month later in an abandoned house where he provocation tests did not elicit any response in the patient
had lived before hospitalization. The third patient, an 18- whose pituitary stalk was severed, although they showed
year-old woman who had the entire tumor removed with a normal and subnormal responses in two and three patients,
section of the pituitary stalk, appeared to be well until 3 respectively, of the five patients whose pituitary stalks
years postsurgery, when she suddenly became ill and died were preserved. Correlation between preservation of the
within a day, probably due to acute pituitary insufficiency. pituitary stalk and postoperative hormone replacement is
Three patients experienced recurrence of their tumors 16 shown in Table 3. The degree of hormone replacement
months, 20 months, and 5 years after the initial operation. was classified into three types: none, needing no hormone
In two of these patients the ACoA was not divided, which replacement; mild, requiring no daily hormone replace-
might have prevented complete removal of the tumor. The ment except DDAVP; and strict, requiring strict daily
pituitary stalks were severed in two of these patients and replacement of corticosteroid and thyroid hormone. When
preserved in the remaining one. the pituitary stalk was anatomically preserved during the
Postoperative visual function tests, including visual operation, postoperative hormone replacement was none
acuity and visual fields, showed improvement in 13 pa- or mild in more than half of cases. On the other hand, in
tients, no change in seven, and deterioration in two pa- cases in which the pituitary stalk was severed or undetect-
tients (Table 1). Mental disturbance was noted in five ed, hormone replacement had to be strict in all but one
patients, three of whom had experienced such disturbance case. Results in postoperative pituitary function were sim-
preoperatively (Table 1). Postoperative visual and mental ilar in patients with and without division of the ACoA.
functions were similar between patients with and without No other major morbidity, such as hemiparesis or cra-
division of the ACoA. nial nerve palsy, was noted in these patients.
The pituitary stalk was anatomically preserved in 14
patients (64%), severed in four (18%), and unidentified in
the remaining four patients (18%) (Table 1). All patients Discussion
had diabetes insipidus postoperatively except one, but We have shown the usefulness of the bifrontal basal
their illness was well controlled with a course of 1- interhemispheric approach with or without division of the
desamine-8-D-arginine vasopressin (DDAVP). Correla- ACoA for large craniopharyngiomas in a series of 22
954 J. Neurosurg. / Volume 84 / June, 1996
Bifrontal basal interhemispheric approach to craniopharyngiomas
TABLE 2 TABLE 3
Correlation between preservation of the pituitary stalk and Correlation between preservation of the pituitary stalk and
normal levels of basal serum ACTH and TSH* postoperative hormonal replacement
Pituitary Stalk (no. of patients) Pituitary Stalk (no. of patients)
Normal Levels Preserved Undetected Severed Preserved Undetected Severed
of Basal Serum (12 patients) (3 patients) (2 patients) Hormone Replacement* (14 patients) (4 patients) (4 patients)
ACTH (>10 pg/ml) none 1 0 0
preop 12 1 2 mild 7 1 0
postop 6 1 0 strict 6 3 4
TSH (>0.5 mU/ml)
preop 8 3 1 * None = no hormone replacement needed; mild = no daily hormone
replacement other than pitressin needed; strict = strict daily replacement
postop 5 0 0
was indispensable.
* Based on total number of cases with measurement of basal serum
ACTH and TSH. ACTH = adrenocorticotropic hormone; TSH = thyroid-
stimulating hormone.
used in the basal interhemispheric approach. The hypo-
thalamus is one of the most important structures requiring
careful handling during surgery. When the hypothalamus
patients. Total or near-total removal of the tumors was is involved in the tumor, one side of the gland is usually
performed in all patients with reasonably low morbidity more severely infiltrated than the other. The preoperative
and no operative mortality. coronal MR image helps to identify the extent of tumor
A bifrontal anterior interhemispheric approach has been invasion into the hypothalamus. Because unilateral hypo-
advocated for use in removing third ventricular tumors by thalamic damage does not seem to cause severe problems
Suzuki, et al.14 This bilateral approach provides a wider postoperatively,16 one can identify the less infiltrated side
operative view of the anterior circle of Willis, the optic of the hypothalamus and preserve that side more easily.
chiasm, and the third ventricular tumor itself, minimizing The ACoA often limits lateral exposure of retrochias-
damage to the brain and vessels. One disadvantage of this matic tumors in the anterior interhemispheric approach.
approach, however, is that it often requires sacrifice of the We have divided this artery without significant early or
bridging veins that connect the medial superior aspects of late complications in 11 of 17 cases of retrochiasmatic
the frontal lobe and the superior sagittal sinus. Division of tumors. This procedure, also described by others,3,8,14 pro-
the bridging veins, when combined with prolonged retrac- vided a much wider operating space, which made the op-
tion of the frontal lobes, increases the risk of postoperative eration easier and safer in these cases. Although preserva-
cerebral ischemia and contusional hemorrhage.10 tion of the artery should be attempted, an undivided
A basal interhemispheric approach has been described ACoA may be damaged inadvertently during retraction of
by Yasui and colleagues17 as a modification of the anteri- the hemispheres. A proper and early decision regarding
or interhemispheric approach for surgery of anterior com- whether to divide the ACoA increases the ease and safety
municating aneurysms, although these surgeons have used of the operation. Since we started to use this approach,
unilateral dissection. Advantages of this approach are: 1) the ACoA has been subjected to more careful scrutiny.
most bridging veins are preserved because few veins of Interestingly, approximately 60% of patients had some
significant size are found near the frontal base; 2) the type of anomaly around the artery, such as a duplication of
extent of dissection in the area of the interhemispheric fis- or window formation in the ACoAs, the median artery of
sure is much less (limited to only the basal part of the fis- the corpus callosum, the accessory middle cerebral artery,
sure) and, consequently, the operative time is shorter and and the azygos anterior cerebral artery, as indicated by
brain damage may be less; and 3) the upper part of the Serizawa and coworkers.12 These variations in arteries
third ventricular tumor can be visualized better from the should be carefully examined when the ACoA is divided.
lower angle. Samii and Samii11 recommended a subfrontal Recurrence rates of craniopharyngiomas after what was
approach with a similar craniotomy to ours for removing believed to be a total excision are surprisingly high in
craniopharyngiomas. However, their approach was made spite of the benign histological nature of this tumor. The
via a subfrontal space without dissecting the interhemi- recurrence rate has been reported as 5.5% by Symon and
spheric fissure, which might require more retraction of the Sprich,15 7% by Yasargil, et al.,16 and 29% by Hoffman, et
frontal lobe. al.9 In our relatively small series, three patients experi-
We believe that bilateral median approaches are superi- enced recurrence during the follow-up period. To reduce
or to unilateral ones, such as the pterional approach, in the recurrence of the tumor, it may be important to minimize
removal of large midline tumors because of the parasagit- blind spots during the operation, using an approach that
tal alignment of important neural structures and perfora- allows a wider exposure. The bifrontal basal interhemi-
tors. In the unilateral approaches, important structures on spheric approach is useful for this purpose.
the contralateral side are located in a blind spot behind the Another important issue relevant to recurrence is the
tumor, and the ipsilateral perforators located on the side of use of GH in children. Growth hormone was used in both
the approach interfere with tumor dissection. On the other of our cases of recurrent tumor in children. In one of these
hand, bilateral median approaches allow direct visualiza- patients, the tumor was believed to have been totally re-
tion of both sides of these structures. Perforating arteries moved together with the pituitary stalk. In the other
do not disturb the operative view in the more median route patient, a minimum portion of tumor capsule was left on
J. Neurosurg. / Volume 84 / June, 1996 955
M. Shibuya, et al.
the stalk. Administration of GH had to be stopped in both for craniopharyngioma.] No Shinkei Geka 17:799–812, 1989
patients after recurrence of the tumors was confirmed. (Jpn)
Whether use of GH induces tumor recurrence is an unset- 4. Baskin DS, Wilson CB: Surgical management of craniopharyn-
tled question;2,5 however, it is not recommended for pa- giomas. A review of 74 cases. J Neurosurg 65:22–27, 1986
tients with residual tumors. 5. Clayton PE, Shalet SM, Gattamaneni HR, et al: Does growth
hormone cause relapse of brain tumours? Lancet 1:711–713,
Postoperative improvement in visual function was ob- 1987
tained in more than half of the patients in our series (13 6. Day JD, Giannotta SL, Fukushima T: Extradural temporopolar
cases) and deterioration was observed in only two pa- approach to lesions of the upper basilar artery and infrachias-
tients. These results seem to be better than those reported matic region. J Neurosurg 81:230–235, 1994
in recent series, in which unilateral approaches were pri- 7. Fischer EG, Welch K, Shillito J Jr, et al: Craniopharyngiomas
marily used.9,16 The bifrontal basal interhemispheric ap- in children. Long-term effects of conservative surgical pro-
proach enables the surgeon to identify both sides of the cedures combined with radiation therapy. J Neurosurg 73:
optic nerves, chiasm, and the optic tracts, possibly allow- 534–540, 1990
ing more chance for postoperative visual improvement. 8. Fujitsu K, Sekino T, Sakata K, et al: Basal interfalcine approach
Anatomical preservation of the pituitary stalk was ob- through a frontal sinusotomy with vein and nerve preservation.
tained in 64% of our cases. This value is much higher than Technical note. J Neurosurg 80:575–579, 1994
that reported by Yasargil, et al.16 (36.5%). When the pitu- 9. Hoffman HJ, Silva MD, Humphreys RP, et al: Aggressive sur-
gical management of craniopharyngiomas in children. J Neu-
itary stalk was anatomically preserved during the op- rosurg 76:47–52, 1992
eration, postoperative hormone replacement was none or 10. Kanno T, Kasama A, Shoda M, et al: A pitfall in the interhemi-
mild in more than half of the cases, whereas it had to spheric translamina terminalis approach for the removal of a
be strict in all but one case in which the pituitary stalk craniopharyngioma. Significance of preserving draining veins.
was severed or undetected. Although anatomical preser- Part I. Clinical study. Surg Neurol 32:111–115, 1989
vation does not necessarily mean preservation of function, 11. Samii M, Samii A: Surgical management of craniopharyn-
patients have a greater chance of remaining free from giomas, in Schmidek HH, Sweet WH (eds): Operative Neuro-
strict postoperative hormone replacement therapy. More- surgical Techniques, ed 3. Philadelphia: WB Saunders, 1995,
over, severe endocrine dysfunction might be related to one pp 357–370
late death in our series, in which the patient’s pituitary 12. Serizawa T, Saeki N, Fukuda K, et al: [Microsurgical anatomy
stalk was severed during the operation. Preservation of the of the anterior communicating artery and its perforating arteries
stalk does not seem to increase the recurrence rate. important for interhemispheric trans-lamina terminalis ap-
proach: analysis based on cadaver brains.] No Shinkei Geka
22:447–454, 1994 (Jpn)
Conclusions 13. Shibuya M, Suzuki Y, Nakane T: A serrated suction tip for tu-
mor removal. Technical note. J Neurosurg 69:140–141, 1988
The present results suggest that the bifrontal basal in- 14. Suzuki J, Katakura R, Mori T: Interhemispheric approach
terhemispheric approach with or without division of the through the lamina terminalis to tumors of the anterior parts of
ACoA is a useful and safe procedure for removal of large the third ventricle. Surg Neurol 22:157–163, 1984
craniopharyngiomas. When the ACoA limits operative 15. Symon L, Sprich W: Radical excision of craniopharyngioma.
exposure, division of the artery can be accomplished safe- Results in 20 patients. J Neurosurg 62:174–181, 1985
ly without significant early or late complications. 16. Yasargil MG, Curcic M, Kis M, et al: Total removal of cranio-
pharyngiomas. Approaches and long-term results in 144 pa-
tients. J Neurosurg 73:3–11, 1990
References
17. Yasui N, Nathal E, Fujiwara H, et al: The basal interhemispher-
1. Al-Mefty O, Hassounah M, Weaver P, et al: Microsurgery ic approach for acute anterior communicating aneurysms. Acta
for giant craniopharyngiomas in children. Neurosurgery 17: Neurochir 118:91–97, 1992
585–594, 1985
2. Arslanian SA, Becker DJ, Lee PA, et al: Growth hormone ther- Manuscript received September 21, 1995.
apy and tumor recurrence. Findings in children with brain neo- Accepted in final form January 17, 1996.
plasms and hypopituitarism. Am J Dis Child 139:347–350, Address reprint requests to: Masato Shibuya, M.D., Depart-
1985 ment of Neurosurgery, Nagoya University School of Medicine, 65
3. Asano T: [Interhemispheric, trans-lamina terminalis approach Tsurumai-cho, Showa-ku, Nagoya 466, Japan.
956 J. Neurosurg. / Volume 84 / June, 1996
Potrebbero piacerti anche
- The Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeDa EverandThe Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeValutazione: 4 su 5 stelle4/5 (5794)
- The Little Book of Hygge: Danish Secrets to Happy LivingDa EverandThe Little Book of Hygge: Danish Secrets to Happy LivingValutazione: 3.5 su 5 stelle3.5/5 (400)
- Shoe Dog: A Memoir by the Creator of NikeDa EverandShoe Dog: A Memoir by the Creator of NikeValutazione: 4.5 su 5 stelle4.5/5 (537)
- Hidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceDa EverandHidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceValutazione: 4 su 5 stelle4/5 (895)
- The Yellow House: A Memoir (2019 National Book Award Winner)Da EverandThe Yellow House: A Memoir (2019 National Book Award Winner)Valutazione: 4 su 5 stelle4/5 (98)
- The Emperor of All Maladies: A Biography of CancerDa EverandThe Emperor of All Maladies: A Biography of CancerValutazione: 4.5 su 5 stelle4.5/5 (271)
- A Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryDa EverandA Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryValutazione: 3.5 su 5 stelle3.5/5 (231)
- Never Split the Difference: Negotiating As If Your Life Depended On ItDa EverandNever Split the Difference: Negotiating As If Your Life Depended On ItValutazione: 4.5 su 5 stelle4.5/5 (838)
- Grit: The Power of Passion and PerseveranceDa EverandGrit: The Power of Passion and PerseveranceValutazione: 4 su 5 stelle4/5 (588)
- On Fire: The (Burning) Case for a Green New DealDa EverandOn Fire: The (Burning) Case for a Green New DealValutazione: 4 su 5 stelle4/5 (73)
- Elon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureDa EverandElon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureValutazione: 4.5 su 5 stelle4.5/5 (474)
- Devil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaDa EverandDevil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaValutazione: 4.5 su 5 stelle4.5/5 (266)
- The Unwinding: An Inner History of the New AmericaDa EverandThe Unwinding: An Inner History of the New AmericaValutazione: 4 su 5 stelle4/5 (45)
- Team of Rivals: The Political Genius of Abraham LincolnDa EverandTeam of Rivals: The Political Genius of Abraham LincolnValutazione: 4.5 su 5 stelle4.5/5 (234)
- The World Is Flat 3.0: A Brief History of the Twenty-first CenturyDa EverandThe World Is Flat 3.0: A Brief History of the Twenty-first CenturyValutazione: 3.5 su 5 stelle3.5/5 (2259)
- The Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreDa EverandThe Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreValutazione: 4 su 5 stelle4/5 (1090)
- The Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersDa EverandThe Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersValutazione: 4.5 su 5 stelle4.5/5 (344)
- The Sympathizer: A Novel (Pulitzer Prize for Fiction)Da EverandThe Sympathizer: A Novel (Pulitzer Prize for Fiction)Valutazione: 4.5 su 5 stelle4.5/5 (121)
- Her Body and Other Parties: StoriesDa EverandHer Body and Other Parties: StoriesValutazione: 4 su 5 stelle4/5 (821)
- Interpretation of FibroScanDocumento16 pagineInterpretation of FibroScanAAMIRNessuna valutazione finora
- 16 Immune System Review SheetDocumento2 pagine16 Immune System Review Sheetjohn gusmanNessuna valutazione finora
- New NORMAL PUERPERIUMDocumento21 pagineNew NORMAL PUERPERIUMvarshaNessuna valutazione finora
- Histology of The Adrenal GlandsDocumento1 paginaHistology of The Adrenal GlandsSabrina NodariNessuna valutazione finora
- A Holistic Approach To CAKUT (Congenital Anomalies of The Kidney and Urinary Tract)Documento3 pagineA Holistic Approach To CAKUT (Congenital Anomalies of The Kidney and Urinary Tract)Kusuma IntanNessuna valutazione finora
- MsgenitourinaryDocumento7 pagineMsgenitourinarybekbekk cabahugNessuna valutazione finora
- Physiological Changes During PregnancyDocumento44 paginePhysiological Changes During PregnancyvrutipatelNessuna valutazione finora
- Final CholelithiasisDocumento36 pagineFinal CholelithiasisRalph Pelegrino100% (2)
- Liver Function TestDocumento22 pagineLiver Function TestSheldon MillerNessuna valutazione finora
- ImmunochemistryDocumento44 pagineImmunochemistryJowe VarnalNessuna valutazione finora
- Saint Louis University School of Medicine Mmxxii: Dr. Baldovino - October 13, 2020Documento11 pagineSaint Louis University School of Medicine Mmxxii: Dr. Baldovino - October 13, 2020Djan Kurvie ValencerinaNessuna valutazione finora
- Anastomosis Left Anterior Cerebral Artery BlockageDocumento5 pagineAnastomosis Left Anterior Cerebral Artery BlockagemcwnotesNessuna valutazione finora
- Pathophysiology of EndocrinologyDocumento25 paginePathophysiology of EndocrinologyISRAELNessuna valutazione finora
- Guideline ThyroidDocumento21 pagineGuideline ThyroidDesti EryaniNessuna valutazione finora
- Human Vision: Jitendra Malik U.C. BerkeleyDocumento45 pagineHuman Vision: Jitendra Malik U.C. BerkeleyElisée Ndjabu DHNessuna valutazione finora
- Pulmonary Edema Vs Pneumonia OrderDocumento25 paginePulmonary Edema Vs Pneumonia OrderRizqon RohmatussadeliNessuna valutazione finora
- Serum Interleukin - 6 Level Among Sudanese Patients With Chronic Kidney DiseaseDocumento3 pagineSerum Interleukin - 6 Level Among Sudanese Patients With Chronic Kidney DiseaseBIOMEDSCIDIRECT PUBLICATIONSNessuna valutazione finora
- EKGin PEDocumento2 pagineEKGin PEพงศธร ปัตถาNessuna valutazione finora
- New Contruncal DevelopmentDocumento66 pagineNew Contruncal DevelopmentMarian AlexNessuna valutazione finora
- Git GutDocumento4 pagineGit GutAngelica Murillo Ang-AngcoNessuna valutazione finora
- The Digestive SystemDocumento33 pagineThe Digestive SystemGulAwezNessuna valutazione finora
- White and Red Pulp (Spleen)Documento4 pagineWhite and Red Pulp (Spleen)Noreen BañagadoNessuna valutazione finora
- Las-Science 6 Quarter 2 Week 1Documento4 pagineLas-Science 6 Quarter 2 Week 1EvelynMartinezNessuna valutazione finora
- Heart Failure 2011 SVDocumento74 pagineHeart Failure 2011 SVtoobstudyingNessuna valutazione finora
- Crush InjuryDocumento14 pagineCrush InjuryDioNessuna valutazione finora
- Thyroid Eye DiseaseDocumento162 pagineThyroid Eye Diseasereza arlasNessuna valutazione finora
- Pancreas: Anatomy, Diseases and Health Implications: September 2012Documento53 paginePancreas: Anatomy, Diseases and Health Implications: September 2012DrFarah Emad AliNessuna valutazione finora
- Themenstrualcycle 120606211927 Phpapp02Documento16 pagineThemenstrualcycle 120606211927 Phpapp02Ludy LynNessuna valutazione finora
- Nursing Care of Client With Endocrine DisorderDocumento93 pagineNursing Care of Client With Endocrine DisorderApril_Anne_Vel_343Nessuna valutazione finora
- DissolutionDocumento14 pagineDissolutionLePetitCeriseNessuna valutazione finora