Documenti di Didattica
Documenti di Professioni
Documenti di Cultura
Ererging Trend Pharma Times
Caricato da
Tarun KumarDescrizione originale:
Copyright
Formati disponibili
Condividi questo documento
Condividi o incorpora il documento
Hai trovato utile questo documento?
Questo contenuto è inappropriato?
Segnala questo documentoCopyright:
Formati disponibili
Ererging Trend Pharma Times
Caricato da
Tarun KumarCopyright:
Formati disponibili
Indian Pharmaceutical Association - Industrial Pharmacy Division
National Workshop
“ EMERGING TRENDS IN PHARMACEUTICAL QUALITY SYSTEMS“
l Pharmaceutical Quality Systems l Good Documentation Practices l Validation of Analytical Methods
Indian Pharmaceutical Association - Industrial Pharmacy
Division ( IPA IPD ) organized a one day National Workshop on Dr. B. Prabha Shankar,
“ Emerging Trends in Pharmaceutical Quality Systems” S e c r e t a r y, I PA - I P D
on Friday, 18th January 2008 at Bangalore as well as at Chandigarh briefed the delegates
on 20th January 2008 in collaboration with SGS & in co- about the aims and
ordination with IPA - Education Division & University objectives of the IPA,
Institute of Pharmaceutical Sciences, Punjab University, Industrial Phar macy
Chandigarh respectively. Division which has been
playing a role of an
ambassador of Pharma Industry in India and working together
in co-ordination with like minded organizations like SGS,
Pharmexcil, Indian Pharmaceutical Alliance, BDMA, IDMA,
OPPI, FICCI and CII. He said the team of IPD has successfully
organized in the last two years five events in various parts of the
country and with this national workshop, they will be
completing seven national events in their two-year term. The
First event, which was organized, was International Workshop
on “Compliance in New and Generic Clinical Drug
Development” in 2006 followed by the Indo-US Interactive
Seminars in three cities - Hyderabad, Mumbai and Ahmedabad
The Technical Experts mainly from Manufacturing, Quality on 24th, 26th and 28th March 2007 respectively on the topic “
Assurance, Quality Control and R&D departments of various Preparing for USFDA and How to face US FDA Inspection and
Pharma Industries from Bangalore, Hosur, Hyderabad, Goa, Recent Trends in Stability Studies” in collaboration with SGS.
Chennai, Mumbai, Ahmedabad, Vadora, Delhi and Baddi Recently, IPA-IPD had organized one more national seminar at
attended the National Workshop besides academicians and Hyderabad on 22nd December 2007 on the burning topic of “
research scholars. Plastic Hazards in Pharma and Consumer Industry and what are
Mr. Kaushik Desai, the Alternatives to Plastics”.
Vice Chairman, IPA IPD
in his welcome address He further added that, this workshop is designed to provide
both at Bangalore and some high level guidelines on designing a harmonized
Chandigarh informed the pharmaceutical quality system applicable across the life cycle of
delegates from Pharma the product. It will refer the integrated approach described on
industry and the academia ICH Q8, Q9 and Q10. The workshop provides the
that it is the second time fundamentals on Good Documentation Practices, practical tips
that IPA-IPD is organizing a workshop in collaboration with on different types of documents in the manufacturing
SGS, Switzerland as a part of continuous education program operations and the recent regulatory observations on poor
and first time at Bangalore & Chandigarh. documentation. Dr. B. Prabha Shankar informed that the expert
on Validation of Analytical Methods and Method Transfers
While briefing about global pharma industrial scenario, Mr. would cover the existing guidelines for the validation of
Desai said that the life of a professional working in Industry is analytical methods, which include practical examples of various
becoming more & more difficult & challenging because of ever successful method transfers and discuss differences regarding
changing requirements. the respective scope of work.
It is IPA-IPD's significant & relentless effort towards this Dr. Mr s. Meena
initiative to educate the Pharma Professionals on the current Kumari, Key Account
trends in the Quality Systems, Good Documentation Practices, Manager- India Africa &
Validation of Analytical Methods and updating them with the Middle East, SGS Life
global regulatory developments. IPA-IPD has been always in Science, Services
forefront in imparting knowledge by organizing such introduced SGS & the
workshops, symposia in various parts of the country. Mr. global functioning of
Kaushik conveyed the greetings to all delegates from Mr. J.A.S. SGS Life Sciences. SGS is
Giri, Chairman, IPA-IPD & Prof. K. Chinna Swamy, Chairman, the world's leading inspection, verification, testing and
IPA -Education Divison. certification company. With 49000 employees, SGS operates a
network of about 840 offices and 320 laboratories around the
world. Originally founded in 1878 in Rouen as French grain Dr. Angela Moy, Global
shipment inspection house, the Company was registered in QA Manager of SGS,
Geneva as Société Générale de Surveillance in 1919. The Canada, in her
facilities are available at US, Canada, Belgium, Germany, France presentation on design of
and recently expanded into many Asian countries with main pharmaceutical quality
facilities at India & Singapore. SGS in India started during 1950, system expressed that
having the headquarters at Mumbai operates a wide network of U.S. FDA and many other
laboratories and offices to cater the needs of all industries and regulators from other
sectors like agriculture, food, phar maceutical and countries have initiated a
environmental, textiles, leather, garments, etc SGS INDIA has risk based inspection approach on quality systems. There is no
entered into exclusive Life Science Services business in Feb. one-size-fits-all approach on quality systems. We need to know
2005, it is fully operational from a specialized bio park at TICEL, our products and our processes well in order to design the most
Chennai and well equipped with experienced personnel and with effective and most efficient quality systems. ICH Q10
the high-quality sophisticated instruments like LC-MS-MS, ICP- demonstrates industry and regulatory authorities' support of an
MS, GC-MS-MS, Real Time PCR, Cell culture, and other effective pharmaceutical quality system to enhance the quality
commonly required pharmaceutical testing instruments. and availability of medicines around the world. Implementation
of an effective, efficient and compliant quality system
Prof. T.V.Narayana, throughout the product lifecycle should facilitate innovation and
Secretary, IPA Education continual improvement and strengthen the link between
Division briefed the pharmaceutical development and manufacturing activities.
delegates at Bangalore
about aims and objectives
about IPA Education
Division and also
informed the delegates
about activities carried in the last two years. He expressed his
happiness for associating with IPA - IPD in organizing this
event, which is equally attended by the Pharma Professionals
from Industry and Academia. He gave a brief introduction of According to Dr. Moy, every Pharma manufacturing processes
the expert speakers Dr. Angela Moy, Director of Global Quality should be effective and efficient to ensure product quality and
Assurance, SGS Life Science Services Division, Canada and Mr. performance through continuous improvement of processes,
Ulrich Markens, Site Manager, SGS, Life Sciences, Singapore. which are monitored by having “real time” assurance of product
quality. Management-based regulation, coupled with regulatory
Prof. A Raghuram Rao, guidance and inspectional oversight provides the foundation for
gave brief idea to the the 'Desired State” GMP + GBP (good business practices) +
delegates at Chandigarh Robust Quality System. A harmonized pharmaceutical quality
about the achievements system applicable across the life cycle of the product
of University Institute of emphasizing an integrated approach to quality risk management
Pharmaceutical Sciences, and science should follow the new ICH tripartite guideline
P u n j a b U n i v e r s i t y, (Q8.Q9.Q10) which simplifies regulatory implementation of
Chandigarh, and the FDA's Initiative and thus serves as a bridge between different
facilities available in their Institute for carrying out Research and regional regulations and thereby helps to achieve global
Development activities for Pharma Industries. He stated that harmonization of quality systems.
his Institution has successfully developed and transferred
technology of three products, which are today commercialized Good Documentation Practices:
by Pharma Industries in India. The department earlier Dr.Angela Moy in her presentation on good documentation
developed an NCE, Chandonium Iodide, whose rights are practices said in the highly regulated pharmaceutical industry, we
currently held by Cipla Ltd. He invited the Pharma Industry to need to produce high quality drug products as well as high
utilize their facilities and expertise for collaborative research. quality documents. All documents associated with drug
He further informed that the University Institute of products from development, registration, validation,
Pharmaceutical Sciences would like to coordinate with IPA manufacturing, quality control, distribution and post approval
IPD in future workshops conducted at Chandigarh. quality assurance are required to meet the GMP standards.
Especially true in the manufacturing operations, if it's not
documented, it's a rumour. We are trained to document almost
everything. She asked the audience whether they are confident
of documenting effectively and efficiently? What and who needs
to be documented? How to ensure traceability in our
documentation? She spoke about the fundamentals of good
Design of Pharmaceutical Quality System : documentation practices, giving several examples and how they
can be linked and traceable, and poor documentation practices
which are cited by regulators. Dr. Moy summarized her
presentation by saying that, a good manufacturing process
coupled with good documentation practice in pharmaceutical
manufacturing can reduce number of manufacturing
supplements and submissions, reward application of prior
knowledge and risk-based approaches, establish the foundation
of Quality by Design, demonstrate the long-term reward of
investing in manufacturing science of operations.
clarified the queries and gave suggestions and advices to the
Validation of Analytical Methods / delegates on the various issues related to Design of
Method Transfers: Pharmaceutical Quality System, Good Documentation
Practices and Validation of Analytical Methods / Method
Mr.Ulrich Markens, Transfers.
Head of Asian
O p e r a t i o n s, S G S, Dr. D. N. Dixit, Vice
Singapore in his President, Quality
presentation said there Assurance, Nicolas
isa worldwide trend in Piramal Ltd., Baddi
the phar maceutical addressed on behalf of
industr y towards the delegates at
reorganization and Chandigarh and
relocation of facilities, transfer of products or product lines congratulated IPA-IPD
from one manufacturing site to another, and increasing for organizing the
outsourcing activities. In this context, the transfer of analytical National Workshop for the first time at Chandigarh, specially for
methods is gaining more and more importance. Unlike the the Pharma Industry Professionals working in and around
validation of analytical methods, there are no guidelines Baddi. He requested IPA-IPD to organize many such
available for the transfer of methods. Here it is up to the Continuing Educational Programs and promised to provide
pharmaceutical company to define a framework in a so-called support.
'Method Transfer Protocol" and record the successful
conclusion of a transfer in a "Method Transfer Report”. Since a Mr. M. Vasudev Rao,
method transfer mostly includes revalidation work, the issues of Executive Council
analytical method transfer and analytical method validation are M e m b e r o f I PA -
inseparably linked. Industrial Phar macy
Division played a key role
at Chandigarh in getting
maximum participants
from Baddi to attend the
workshop and proposed a vote of thanks.
Mr. Nagarjuna Reddy, Executive
Council Member IPA Education
Division Co-ordinated in Making
He sighted some examples of successful analytical method local arrangements and motivated
transfers like, transfer without revalidation, transfer with minor M. Pharm students to attend the
revalidation effort & transfer with complex revalidation effort. workshop at Bangalore. and
Before starting the transfer, following things are necessary to be thanked Dr. V. Satyanarayana,
followed: Ensure that the method you plan to transfer is robust, Treasurer IPA IPD for attending the Workshop at Bangalore.
define all critical points in procedure, bring all involved persons
together (face-to-face meeting), ensure that reference materials
are available, ensure that all necessary information is transferred
in advance and write a comprehensive Transfer Protocol. After
finishing the transfer, review all documents, write Transfer
Report and then Receiving Lab is qualified to test samples and
the Method is transferred successfully.
The Delegates at Bangalore and Chandigarh actively
participated in the interactive session where the guest speakers
Potrebbero piacerti anche
- Quality Assurance and Quality Management in Pharmaceutical IndustryDa EverandQuality Assurance and Quality Management in Pharmaceutical IndustryValutazione: 4 su 5 stelle4/5 (4)
- How to Integrate Quality by Efficient Design (QbED) in Product DevelopmentDa EverandHow to Integrate Quality by Efficient Design (QbED) in Product DevelopmentNessuna valutazione finora
- 2nd Annual Clinical Trials Asia Summit 2011: Key Speakers IncludeDocumento6 pagine2nd Annual Clinical Trials Asia Summit 2011: Key Speakers IncludeFen CastroNessuna valutazione finora
- QAQC Medical DeviceDocumento32 pagineQAQC Medical DeviceMOHNISH MANCHALWARNessuna valutazione finora
- Food Tea Technology ProspectusDocumento17 pagineFood Tea Technology ProspectusSandra sandyNessuna valutazione finora
- SynopsisDocumento11 pagineSynopsisPramod ShawNessuna valutazione finora
- Quality Assurance6 PDFDocumento6 pagineQuality Assurance6 PDFMaruti SapataleNessuna valutazione finora
- 27EASJune 5192 1Documento19 pagine27EASJune 5192 1Olalekan OjoNessuna valutazione finora
- QBD MaterialDocumento5 pagineQBD MaterialRodrigo BressanNessuna valutazione finora
- Pharmacy Resume TemplateDocumento7 paginePharmacy Resume Templatesathish.sure20Nessuna valutazione finora
- ISPE Kneat ND BG5 CQ Oct Intro EventsDocumento7 pagineISPE Kneat ND BG5 CQ Oct Intro EventscampurriaNessuna valutazione finora
- Sumit Saxena (Speaker Profile)Documento1 paginaSumit Saxena (Speaker Profile)Maven Quality Assurance CounselNessuna valutazione finora
- Implementation of Effective Quality Control To Improve Product Quality (Case Study PT Metiska Pharmaceutical, Jakarta)Documento5 pagineImplementation of Effective Quality Control To Improve Product Quality (Case Study PT Metiska Pharmaceutical, Jakarta)International Journal of Innovative Science and Research TechnologyNessuna valutazione finora
- Fyp BiotechDocumento86 pagineFyp BiotechdharamNessuna valutazione finora
- GMP in Pharmaceutical Industry: Global cGMP & Regulatory ExpectationsDa EverandGMP in Pharmaceutical Industry: Global cGMP & Regulatory ExpectationsValutazione: 5 su 5 stelle5/5 (2)
- Quality by Design: Review ArticleDocumento13 pagineQuality by Design: Review ArticleIjupbs IjupbsNessuna valutazione finora
- IPC BrochureDocumento16 pagineIPC BrochurevihatvipulNessuna valutazione finora
- Important Terminology in Pharmaceutical Quality Assurance: Literature ReviewDocumento21 pagineImportant Terminology in Pharmaceutical Quality Assurance: Literature ReviewKulfi BarfiNessuna valutazione finora
- Research Paper On Clinical Trials in IndiaDocumento4 pagineResearch Paper On Clinical Trials in Indiaegwv92v7100% (1)
- Good Manufacturing Practices For Food Processing Industries: Purposes, Principles and Practical ApplicationsDocumento23 pagineGood Manufacturing Practices For Food Processing Industries: Purposes, Principles and Practical ApplicationsMohamed SamiNessuna valutazione finora
- DR - Yuvaraj Chromosoft CRC ChennaiDocumento15 pagineDR - Yuvaraj Chromosoft CRC ChennaisunilNessuna valutazione finora
- Sea Immun 42Documento70 pagineSea Immun 42american_guy10Nessuna valutazione finora
- INDUSTRIAL TRANING - Garima SinghDocumento17 pagineINDUSTRIAL TRANING - Garima SinghARVIND KUMARNessuna valutazione finora
- QBD Putting Theory Into Practice - Chp2 THE REGULATORY FRAMEWORK PDFDocumento26 pagineQBD Putting Theory Into Practice - Chp2 THE REGULATORY FRAMEWORK PDFhdmnauNessuna valutazione finora
- A Guide To GXP ComplianceDocumento6 pagineA Guide To GXP ComplianceManel VazquezNessuna valutazione finora
- Research Paper On Knowledge Management Practices in Pharma CompanyDocumento7 pagineResearch Paper On Knowledge Management Practices in Pharma Companyafnhekkghifrbm100% (1)
- Quality Management System, DR - Reddy'sDocumento66 pagineQuality Management System, DR - Reddy'sParul Thakur67% (3)
- Prospectus PMDocumento47 pagineProspectus PMghfjrcsk8vNessuna valutazione finora
- IICRDocumento17 pagineIICRMukund SharmaNessuna valutazione finora
- Stox AnnouncementDocumento5 pagineStox Announcementapi-294657196Nessuna valutazione finora
- ISPE Informer 201210-ENDocumento4 pagineISPE Informer 201210-ENDaryl Barrios LamedaNessuna valutazione finora
- 94Documento95 pagine94lthyaguNessuna valutazione finora
- Curriculum Vitae - Gowtham - 1802Documento2 pagineCurriculum Vitae - Gowtham - 1802Anonymous b1TFqirANessuna valutazione finora
- GMP (Good Manufacturing Practices) GLP (Good Laboratory Practice) GCP (Good Clinical Practice)Documento2 pagineGMP (Good Manufacturing Practices) GLP (Good Laboratory Practice) GCP (Good Clinical Practice)Ashish BhangriyaNessuna valutazione finora
- Thesis M.sc. MicrobiologyDocumento40 pagineThesis M.sc. MicrobiologyMohit Agarwal100% (1)
- Cosmetic Technology ProspectusDocumento20 pagineCosmetic Technology Prospectusdr NayanBharadwajNessuna valutazione finora
- Paper 8873Documento10 paginePaper 8873IJARSCT JournalNessuna valutazione finora
- Brosur ISPE Conference 2014Documento2 pagineBrosur ISPE Conference 2014ISPE_IndonesiaNessuna valutazione finora
- Quality Metrics and KpiDocumento5 pagineQuality Metrics and Kpidilip das100% (2)
- Quality Systems RoadmapDocumento32 pagineQuality Systems Roadmapwasim akramNessuna valutazione finora
- 19.Md. Sahab Uddin Et Al., IAJPS 2016, 3 (12), 1624-1638Documento15 pagine19.Md. Sahab Uddin Et Al., IAJPS 2016, 3 (12), 1624-1638Abdul RahimNessuna valutazione finora
- Industry Training SampleDocumento23 pagineIndustry Training SampleAnwar Ul HaqNessuna valutazione finora
- Pharmaceutical EngineeringDocumento5 paginePharmaceutical EngineeringIzzat EzmanNessuna valutazione finora
- A Textbook of Pharmaceutical Quality AssuranceDa EverandA Textbook of Pharmaceutical Quality AssuranceValutazione: 3 su 5 stelle3/5 (6)
- Some Quality Control Analysis Parameters For Parenteral FormulationsDocumento5 pagineSome Quality Control Analysis Parameters For Parenteral FormulationsEditor IJTSRDNessuna valutazione finora
- Anoop 1Documento23 pagineAnoop 1Akash YadavNessuna valutazione finora
- New Challenges For Biopharmaceutical Process DevelopmentDocumento4 pagineNew Challenges For Biopharmaceutical Process DevelopmentAmgad_SNessuna valutazione finora
- Biswa Mohan Padhy - 102427673Documento3 pagineBiswa Mohan Padhy - 102427673Mumtaj AnsariNessuna valutazione finora
- Review Article (Cleaning Validation)Documento13 pagineReview Article (Cleaning Validation)vikram shankar veerNessuna valutazione finora
- L Q: Mba, MD (A), Bams: Name: D Sreedhar TirunagariDocumento5 pagineL Q: Mba, MD (A), Bams: Name: D Sreedhar TirunagariSreedhar TirunagariNessuna valutazione finora
- QCTech BulletinDocumento20 pagineQCTech BulletinOmairNessuna valutazione finora
- Quality ControlDocumento11 pagineQuality Controlsinghdikshaddn123Nessuna valutazione finora
- Synopsis PharmaDocumento15 pagineSynopsis PharmaEkansh NarangNessuna valutazione finora
- CosmeticTechnologyProspectus PDFDocumento24 pagineCosmeticTechnologyProspectus PDFKetan HulajiNessuna valutazione finora
- Everest 23 Chandra - Consultant ProfileDocumento1 paginaEverest 23 Chandra - Consultant ProfileAbdul ZahinNessuna valutazione finora
- Historical Incidents Leading To The Evolution of Good Manufacturing PracticeDocumento3 pagineHistorical Incidents Leading To The Evolution of Good Manufacturing PracticeSantosh Kumar TataNessuna valutazione finora
- GCLPDocumento57 pagineGCLPkapindraNessuna valutazione finora
- BioPharma Asia PremailerDocumento34 pagineBioPharma Asia PremailerMing En100% (1)
- Good Laboratory Practices and Compliance MonitoringDa EverandGood Laboratory Practices and Compliance MonitoringNessuna valutazione finora
- NoticePreSchool 2017Documento1 paginaNoticePreSchool 2017sushantNessuna valutazione finora
- TransBio-filter Sewage Treatment Plant PresentationDocumento21 pagineTransBio-filter Sewage Treatment Plant PresentationsushantNessuna valutazione finora
- TransBio-filter Green Waste Water Technology - Convert Your Waste Into Bio-Nutritional ProductsDocumento52 pagineTransBio-filter Green Waste Water Technology - Convert Your Waste Into Bio-Nutritional ProductssushantNessuna valutazione finora
- TransBio-filter Presentation With Case StudiesDocumento61 pagineTransBio-filter Presentation With Case StudiessushantNessuna valutazione finora
- Technological - Comparision - Chart For Continuous Online Monitoring SystemDocumento45 pagineTechnological - Comparision - Chart For Continuous Online Monitoring SystemsushantNessuna valutazione finora
- Request For Advance or Reimbursement: (See Instructions On Back)Documento1 paginaRequest For Advance or Reimbursement: (See Instructions On Back)sushantNessuna valutazione finora
- Lepro Herbal Borewell Water Quality StandardsDocumento5 pagineLepro Herbal Borewell Water Quality StandardssushantNessuna valutazione finora
- Enzyme CatalysisDocumento65 pagineEnzyme Catalysissushant100% (1)
- Unit One This or That 1Documento9 pagineUnit One This or That 1sushantNessuna valutazione finora
- Flow Meter Slection GuideDocumento8 pagineFlow Meter Slection GuidesushantNessuna valutazione finora
- Topic 2: Molecular Biology (Student)Documento5 pagineTopic 2: Molecular Biology (Student)sushantNessuna valutazione finora
- Unit One EOC Review: Molecule That Contains Carbon and Is Important To LifeDocumento4 pagineUnit One EOC Review: Molecule That Contains Carbon and Is Important To LifesushantNessuna valutazione finora
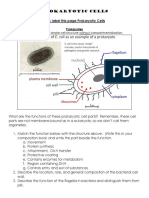
- In Your Composition Book Label This Page Prokaryotic CellsDocumento5 pagineIn Your Composition Book Label This Page Prokaryotic CellssushantNessuna valutazione finora
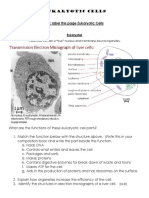
- In Your Composition Book Label This Page Eukaryotic CellsDocumento4 pagineIn Your Composition Book Label This Page Eukaryotic CellssushantNessuna valutazione finora
- Lecture 1 PDFDocumento33 pagineLecture 1 PDFsushantNessuna valutazione finora