Documenti di Didattica
Documenti di Professioni
Documenti di Cultura
Adiabatic Process and Polytropic Process
Caricato da
Avinash GarikapatiDescrizione originale:
Titolo originale
Copyright
Formati disponibili
Condividi questo documento
Condividi o incorpora il documento
Hai trovato utile questo documento?
Questo contenuto è inappropriato?
Segnala questo documentoCopyright:
Formati disponibili
Adiabatic Process and Polytropic Process
Caricato da
Avinash GarikapatiCopyright:
Formati disponibili
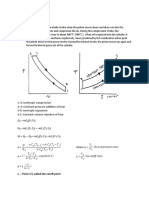
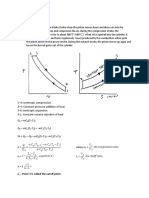
Adiabatic process
An adiabatic process is a process in which no heat transfer takes place. As no heat transfer takes
place into or out of the system it is an isentropic process. This process is of the form:
Pvk = constant --------------------------------------------------------------------------(1)
where,
P = pressure,
v = volume,
k = ratio of specific heats (Cp/Cv)
Polytropic process
A polytropic process is a variable entropy process in which heat transfer takes place during the
process. It is of the form:
Pvn = constant --------------------------------------------------------------------------- (2)
where,
n = k for an adiabatic, isentropic process
= 1 for an isothermal process
= 0 for an isobaric process
= ∞ for an isochoric process
From equation (2) we may write for a polytropic process:
P1v1n = P2v2n = constant
where,
P1 = initial pressure,
P2 = final pressure,
v1 = initial volume,
v2 = final volume,
P2 / P1 = (v1/v2)n ------------------------------------------------------------------------ (3)
Also,
P1v1/T1 = P2v2/T2
v1/v2 = P2T1/P1T2 ----------------------------------------------------------------------- (4)
Using (4) in (3):
P2/P1 = (P2T1/P1T2)n
(P2/P1)(1-n) = (T2/T1)-n
Defining P2/P1 = rP and T2/T1 as rT we can write:
rT = rP(n-1)/n -------------------------------------------------------------------------------- (5)
Taking logarithm on both sides:
ln rT = (n-1)/n ln rP
n/(n-1) = (ln rP)/( ln rT) ----------------------------------------------------------------(6)
Polytropic head and Polytropic efficiency
Polytropic head is defined as the work output per mass of fluid. For a steady state, steady flow
process, the polytropic head is given by the following relationship:
HP = 1∫2 vdP ----------------------------------------------------------------------------- (7)
From equation (2):
Pvn = constant = Cn --------------------------------------------------------------------- (8)
v = C/P1/n
Substituting v in (7) we have:
HP = 1∫2 C/P1/n dP
= C/(1-1/n)*P1-(1/n) 1│ 2
= CnP(n-1)/n/(n-1) 1│ 2
= Cn/(n-1) * [P2(n-1)n – P1(n-1)n]
= Cn P1(n-1)n/(n-1)*[(P2/P1)(n-1)/n - 1]
= Cn P1(n-1)n/(n-1)*[rP(n-1)/n - 1] --------------------------------------------------- (9)
From (8) we have:
Pvn = Cn
C = (Pvn)1/n = P11/n v1
Using the value of C in (9):
HP = nP11/n v1 P1(n-1)n/(n-1)*[rP(n-1)/n - 1]
= n/(n-1) * P1v1[rP(n-1)/n - 1] ------------------------------------------------------ (10)
From the ideal gas law:
P1v1 = RT1
where,
v = specific volume
R = gas constant = function (molecular weight)
= 8314/mol.wt
. Nm/kgK in metric system
Using this in equation (10) we have:
HP = n/(n-1) * RT1[rP(n-1)/n - 1] ----------------------------------------------------- (11)
Polytropic efficiency of a compression process is defined as the ratio of the work output to the
work input.
The work input per mass of fluid is the change of enthalpy of the gas.
w = ∆h
= cp(T2 - T1)
= cp T1(T2/ T1 - 1)
= cp T1(rT - 1)
Using (5) in the above equation:
w = cp T1(rP(n-1)/n - 1) ----------------------------------------------------------------- (12)
But,
cp = Rk/(k - 1)
Using this in (12):
w = Rk/(k - 1) * T1(rP(n-1)/n - 1) ------------------------------------------------------ (13)
Therefore, polytropic efficiency,
ηP = Hp/w
n /(n−1)∗RT 1 [r P(n−1)/n−1]
=
k /( k−1)∗RT 1(r P(n−1)/n−1)
n/( n−1)
ηP = ----------------------------------------------------------------------------(14)
k /( k−1)
Dependence of polytropic efficiency on inlet and outlet conditions.
Combining equation (6) and (14) and assuming ‘k’ to remain constant under all circumstances,
we can say:
ηP α (ln rP)/( ln rT)
Therefore, the polytropic efficiency of a stage of a multi-stage centrifugal compressor is directly
proportional to the pressure ratio and inversely proportional to the temperature ratio of the given
stage. Thus, for a process with constant inlet pressure and temperature condition, best
polytropic efficiency will be achieved if maximum pressure rise can be achieved for a
minimum possible temperature rise.
Potrebbero piacerti anche
- Dual CycleDocumento3 pagineDual CycleMohammad Faraz AkhterNessuna valutazione finora
- PNEUMATICS AND AIR CIRCUITS UNDERSTANDING THE CASCADE VALVE AND PLC UNDERSTANDINGDa EverandPNEUMATICS AND AIR CIRCUITS UNDERSTANDING THE CASCADE VALVE AND PLC UNDERSTANDINGNessuna valutazione finora
- Thermodynamics 2 E7Documento41 pagineThermodynamics 2 E7taya699Nessuna valutazione finora
- Reference Guide To Useful Electronic Circuits And Circuit Design Techniques - Part 2Da EverandReference Guide To Useful Electronic Circuits And Circuit Design Techniques - Part 2Nessuna valutazione finora
- Reciprocal CompressorsDocumento40 pagineReciprocal CompressorsSathish KasilingamNessuna valutazione finora
- Physics FormulasDocumento6 paginePhysics FormulasRam PrasadNessuna valutazione finora
- Exercises in Electronics: Operational Amplifier CircuitsDa EverandExercises in Electronics: Operational Amplifier CircuitsValutazione: 3 su 5 stelle3/5 (1)
- Non Flow Processes Gases Closed SystemDocumento9 pagineNon Flow Processes Gases Closed SystemNjabulo NgobeseNessuna valutazione finora
- Unit: 4Documento25 pagineUnit: 4Vikas MishraNessuna valutazione finora
- Air Compressor1Documento24 pagineAir Compressor1Sayem kaifNessuna valutazione finora
- Unit: 4Documento27 pagineUnit: 4Vikas MishraNessuna valutazione finora
- Peng-Robinson Fortran ProgramDocumento12 paginePeng-Robinson Fortran ProgramSamuel Pangeran SilalahiNessuna valutazione finora
- Axial CompressorDocumento18 pagineAxial CompressorSumiran ManghaniNessuna valutazione finora
- Mover, Otherwise Known As The Turbine. For: R P S T K XDocumento16 pagineMover, Otherwise Known As The Turbine. For: R P S T K XMohamad SannanNessuna valutazione finora
- TD Unit 2Documento6 pagineTD Unit 2Shine SonNessuna valutazione finora
- DatasheetDocumento16 pagineDatasheetSandu VisanescuNessuna valutazione finora
- Formula Overview (Aeronautics)Documento8 pagineFormula Overview (Aeronautics)Aishwarya RaviNessuna valutazione finora
- Isochronal Test3Documento36 pagineIsochronal Test3Veviet pomataNessuna valutazione finora
- POWER FACTOR CORRECTION For DUMMIESDocumento1 paginaPOWER FACTOR CORRECTION For DUMMIESJin WuNessuna valutazione finora
- CRE SUBJECT DoubtClearnaceDocumento2 pagineCRE SUBJECT DoubtClearnaceDhanu DhanushNessuna valutazione finora
- Gas Power Cycles Study Guide in Powerpoint: To AccompanyDocumento68 pagineGas Power Cycles Study Guide in Powerpoint: To AccompanyManjunatha TnNessuna valutazione finora
- Gas Reservoirs6Documento35 pagineGas Reservoirs6Veviet pomataNessuna valutazione finora
- Ic Engine Cycles 1Documento85 pagineIc Engine Cycles 1jhpandiNessuna valutazione finora
- ThermodynamicsNotes PDFDocumento41 pagineThermodynamicsNotes PDFAsia CtNessuna valutazione finora
- Lte KomutlarDocumento6 pagineLte Komutlaremre kayaNessuna valutazione finora
- Prelim Ito Na PreDocumento8 paginePrelim Ito Na PreJethro Briza GaneloNessuna valutazione finora
- Command 2G 3G 4G 18052022Documento66 pagineCommand 2G 3G 4G 18052022yacine bouazniNessuna valutazione finora
- CH 02Documento20 pagineCH 02Pauline Nguyen100% (1)
- Timing Diagram Timers CycleDocumento1 paginaTiming Diagram Timers Cycledon121don121Nessuna valutazione finora
- Nota PemampatDocumento49 pagineNota PemampatweafareezNessuna valutazione finora
- Chap3 4Documento10 pagineChap3 4rpb109100% (1)
- Diesel Cycle: R - Point 3 Is Called The Cutoff PointDocumento7 pagineDiesel Cycle: R - Point 3 Is Called The Cutoff PointJethro Briza GaneloNessuna valutazione finora
- Lecture Notes OnDocumento200 pagineLecture Notes Onananth k r100% (3)
- Math Formula ListDocumento3 pagineMath Formula ListKumaravel PadmaroopaNessuna valutazione finora
- For An Isentropic Process S SDocumento11 pagineFor An Isentropic Process S SRoshan ShanmughanNessuna valutazione finora
- Thermodynamics FormulasDocumento16 pagineThermodynamics FormulasMohammad SaleemNessuna valutazione finora
- Otto CycleDocumento28 pagineOtto CycleNazeeh Abdulrhman AlbokaryNessuna valutazione finora
- Thermodynamic CyclesDocumento30 pagineThermodynamic CyclesRudra PratapNessuna valutazione finora
- MmcinfinityDocumento17 pagineMmcinfinityKartik GuptaNessuna valutazione finora
- Diesel Cycle: R - Point 3 Is Called The Cutoff PointDocumento8 pagineDiesel Cycle: R - Point 3 Is Called The Cutoff PointJethro Briza GaneloNessuna valutazione finora
- Ambiga JDocumento120 pagineAmbiga Jela666666Nessuna valutazione finora
- ⎟⎟ ⎠ ⎞ ⎜⎜ ⎝ ⎛ − − = T T 1 T T 1 1 η: www.freestudy.co.ukDocumento1 pagina⎟⎟ ⎠ ⎞ ⎜⎜ ⎝ ⎛ − − = T T 1 T T 1 1 η: www.freestudy.co.ukcataiceNessuna valutazione finora
- Power System Simulation Laboratory: A RecordDocumento120 paginePower System Simulation Laboratory: A Recordela666666Nessuna valutazione finora
- Proracun Gasne TurbineDocumento15 pagineProracun Gasne TurbineToni TalevskiNessuna valutazione finora
- Patterns While Loop Part-1Documento13 paginePatterns While Loop Part-1vsnpradeepNessuna valutazione finora
- P P A U M T A M P P U U: AS301, IITM, Lecture 2 (06/08/2007)Documento3 pagineP P A U M T A M P P U U: AS301, IITM, Lecture 2 (06/08/2007)Sunil VermaNessuna valutazione finora
- Module 33D - Closed Loop Proportional-Integral Pressure ControlDocumento4 pagineModule 33D - Closed Loop Proportional-Integral Pressure ControlrhedmishNessuna valutazione finora
- Formulario para Ser Utilizado Durante El Examen TransformadoresDocumento1 paginaFormulario para Ser Utilizado Durante El Examen TransformadoresFernando Manuel Fernandez GhisoNessuna valutazione finora
- System Design Assignment - 1: SUBMITTED BY:-P.D.ANEESH (113079017) VISMAY DESAI (11307R007)Documento10 pagineSystem Design Assignment - 1: SUBMITTED BY:-P.D.ANEESH (113079017) VISMAY DESAI (11307R007)PD AneeshNessuna valutazione finora
- Assignment Exp 5ADocumento6 pagineAssignment Exp 5AMohammed TAOUSSINessuna valutazione finora
- Propeller Design Detail StageDocumento4 paginePropeller Design Detail Stageleokyawlay87Nessuna valutazione finora
- Fa17-Eee-008 - Asssignment 4Documento6 pagineFa17-Eee-008 - Asssignment 4Hammad SattiNessuna valutazione finora
- Thus, This Term Actually Means A in A Constant-Volume System The Measure of Reaction Rate of Component I BecomesDocumento23 pagineThus, This Term Actually Means A in A Constant-Volume System The Measure of Reaction Rate of Component I Becomesalice Annabelle100% (1)
- Engineering Software: Otto Cycle AnalysisDocumento14 pagineEngineering Software: Otto Cycle AnalysisAnonymous J6blfqNessuna valutazione finora
- Ee 705-Vlsi Design Lab: Describing A Finite State Machine (FSM)Documento10 pagineEe 705-Vlsi Design Lab: Describing A Finite State Machine (FSM)Indranil ChakrabortyNessuna valutazione finora
- Op Amp ApplicationDocumento15 pagineOp Amp ApplicationChris Real PabiaNessuna valutazione finora
- TutUM UM MarcH-2017Documento6 pagineTutUM UM MarcH-2017Dennis LingNessuna valutazione finora
- Plant Engineering 2 - Compressors 1 The Perfect Gas ModelDocumento12 paginePlant Engineering 2 - Compressors 1 The Perfect Gas ModelDee RajanNessuna valutazione finora
- Pump Classifications:: Transportation of FluidsDocumento2 paginePump Classifications:: Transportation of FluidsAvinash GarikapatiNessuna valutazione finora
- ExtractionDocumento13 pagineExtractionAvinash GarikapatiNessuna valutazione finora
- Compressible and Incompressible FluidDocumento3 pagineCompressible and Incompressible FluidAvinash GarikapatiNessuna valutazione finora
- Fluid Mechanics IntroductionDocumento4 pagineFluid Mechanics IntroductionAvinash GarikapatiNessuna valutazione finora
- Extraction of Essential OilsDocumento9 pagineExtraction of Essential OilsAvinash GarikapatiNessuna valutazione finora
- FiltarationDocumento22 pagineFiltarationAvinash GarikapatiNessuna valutazione finora
- Unsteady State Batch ReactorDocumento11 pagineUnsteady State Batch ReactorAvinash GarikapatiNessuna valutazione finora
- Tin ExtractionDocumento10 pagineTin ExtractionAvinash GarikapatiNessuna valutazione finora
- Process Calculations 2nd Ed. - V. Venkataramani, N. Anantharaman & K.M. Meera Sheriffa Begum 2011 PDFDocumento260 pagineProcess Calculations 2nd Ed. - V. Venkataramani, N. Anantharaman & K.M. Meera Sheriffa Begum 2011 PDFantonino69100% (6)
- Binomial DistributionDocumento10 pagineBinomial DistributionAvinash GarikapatiNessuna valutazione finora
- Photoelectric PyrometerDocumento13 paginePhotoelectric PyrometerAvinash Garikapati100% (1)
- Cannabis ManufacturingDocumento6 pagineCannabis ManufacturingSebastián RamirezNessuna valutazione finora
- Presentation Slides AgilentDocumento36 paginePresentation Slides AgilentAngela MoraNessuna valutazione finora
- Endotoxin Removal: The Solution With Membrane Separation TechnologyDocumento4 pagineEndotoxin Removal: The Solution With Membrane Separation TechnologykapilNessuna valutazione finora
- Stress Strain CurveDocumento4 pagineStress Strain CurveTherese Kyle LauritoNessuna valutazione finora
- Pt. Graha Dwi Mitra Mandiri at Arumdaeun KimiaDocumento20 paginePt. Graha Dwi Mitra Mandiri at Arumdaeun Kimianovi cmidNessuna valutazione finora
- Standard Operating Procedure For Calibration and Maintenance of PH MetersDocumento10 pagineStandard Operating Procedure For Calibration and Maintenance of PH MetersAnonymous w7gZFE8Nessuna valutazione finora
- Material P1Documento24 pagineMaterial P1Salik SalikNessuna valutazione finora
- Installation Instruction For Conventional Castables PDFDocumento2 pagineInstallation Instruction For Conventional Castables PDFakv_bhelNessuna valutazione finora
- Paschotta2010 - Derivation of The Schawlow - Townes Linewidth of LasersDocumento6 paginePaschotta2010 - Derivation of The Schawlow - Townes Linewidth of LasersHiso JokeyNessuna valutazione finora
- Gen Chem ReviewerDocumento4 pagineGen Chem Revieweraldrin josephNessuna valutazione finora
- Armand Barbault - Gold of A Thousand Mornings (1969)Documento88 pagineArmand Barbault - Gold of A Thousand Mornings (1969)ghostplane93% (14)
- Chapter-6 Life Processes MCQsDocumento13 pagineChapter-6 Life Processes MCQsfagstdjdNessuna valutazione finora
- 4ch1 1c Pef 20220825Documento37 pagine4ch1 1c Pef 20220825sherNessuna valutazione finora
- Alloy AllDocumento68 pagineAlloy AllMousa HamadNessuna valutazione finora
- Ion Exchange Process Problems SolutionsDocumento73 pagineIon Exchange Process Problems Solutionsanup_nairNessuna valutazione finora
- Refrigeration CyclesDocumento29 pagineRefrigeration CyclesJoulie Jonsi100% (1)
- (Richard C. Alkire, Dieter M. Kolb, Jacek LipkowskDocumento281 pagine(Richard C. Alkire, Dieter M. Kolb, Jacek LipkowskYanpiero BalladoresNessuna valutazione finora
- MAT 2640 MCGB - Data Sheet For Suppliers Old MAT No.: 308Documento3 pagineMAT 2640 MCGB - Data Sheet For Suppliers Old MAT No.: 308pkjenaleoNessuna valutazione finora
- Calibration Curve: Kathleen Faith C. Briones Hope Louis Simbrio BS in Chemistry III Experiment No. 9Documento6 pagineCalibration Curve: Kathleen Faith C. Briones Hope Louis Simbrio BS in Chemistry III Experiment No. 9Kat Chavez BrionesNessuna valutazione finora
- Haba JawapanDocumento10 pagineHaba JawapanScotrraaj GopalNessuna valutazione finora
- Model 10 10fb 11 Carbon HousingsDocumento4 pagineModel 10 10fb 11 Carbon HousingsrezNessuna valutazione finora
- Monarch Epoxy Flooring - Floor Coating - For Industrial & Domestic UseDocumento2 pagineMonarch Epoxy Flooring - Floor Coating - For Industrial & Domestic UsegangadevimgmNessuna valutazione finora
- Base Plate (Template)Documento3 pagineBase Plate (Template)li yongzhongNessuna valutazione finora
- CH 224 Assignment Test-IIDocumento1 paginaCH 224 Assignment Test-IIBathula KasiNessuna valutazione finora
- Flo-Grout EP270 - Filler - MSDSDocumento3 pagineFlo-Grout EP270 - Filler - MSDSahmed draiNessuna valutazione finora
- Detergent Alkylates - World Markets To 2025 ProspectusDocumento27 pagineDetergent Alkylates - World Markets To 2025 ProspectusRafflesia GroupNessuna valutazione finora
- The Medical Biochemistry PageDocumento1 paginaThe Medical Biochemistry PageSamNessuna valutazione finora
- Condenser Tube Failure GeneralDocumento273 pagineCondenser Tube Failure GeneralRagvendra BistNessuna valutazione finora
- Toxicology Laboratory ManualDocumento110 pagineToxicology Laboratory Manualnguyen ba trung100% (1)
- WPS 1-1-GT-SM-6 Rev. 0Documento8 pagineWPS 1-1-GT-SM-6 Rev. 0Melby de la rosaNessuna valutazione finora
- Sully: The Untold Story Behind the Miracle on the HudsonDa EverandSully: The Untold Story Behind the Miracle on the HudsonValutazione: 4 su 5 stelle4/5 (103)
- Fire on the Horizon: The Untold Story of the Gulf Oil DisasterDa EverandFire on the Horizon: The Untold Story of the Gulf Oil DisasterNessuna valutazione finora
- The End of Craving: Recovering the Lost Wisdom of Eating WellDa EverandThe End of Craving: Recovering the Lost Wisdom of Eating WellValutazione: 4.5 su 5 stelle4.5/5 (83)
- The Fabric of Civilization: How Textiles Made the WorldDa EverandThe Fabric of Civilization: How Textiles Made the WorldValutazione: 4.5 su 5 stelle4.5/5 (58)
- Hero Found: The Greatest POW Escape of the Vietnam WarDa EverandHero Found: The Greatest POW Escape of the Vietnam WarValutazione: 4 su 5 stelle4/5 (19)
- Transformed: Moving to the Product Operating ModelDa EverandTransformed: Moving to the Product Operating ModelValutazione: 4 su 5 stelle4/5 (1)
- The Intel Trinity: How Robert Noyce, Gordon Moore, and Andy Grove Built the World's Most Important CompanyDa EverandThe Intel Trinity: How Robert Noyce, Gordon Moore, and Andy Grove Built the World's Most Important CompanyNessuna valutazione finora
- Dirt to Soil: One Family’s Journey into Regenerative AgricultureDa EverandDirt to Soil: One Family’s Journey into Regenerative AgricultureValutazione: 5 su 5 stelle5/5 (125)
- The Future of Geography: How the Competition in Space Will Change Our WorldDa EverandThe Future of Geography: How the Competition in Space Will Change Our WorldValutazione: 4 su 5 stelle4/5 (6)
- Process Plant Equipment: Operation, Control, and ReliabilityDa EverandProcess Plant Equipment: Operation, Control, and ReliabilityValutazione: 5 su 5 stelle5/5 (1)
- The Technology Trap: Capital, Labor, and Power in the Age of AutomationDa EverandThe Technology Trap: Capital, Labor, and Power in the Age of AutomationValutazione: 4.5 su 5 stelle4.5/5 (46)
- When the Heavens Went on Sale: The Misfits and Geniuses Racing to Put Space Within ReachDa EverandWhen the Heavens Went on Sale: The Misfits and Geniuses Racing to Put Space Within ReachValutazione: 3.5 su 5 stelle3.5/5 (6)
- System Error: Where Big Tech Went Wrong and How We Can RebootDa EverandSystem Error: Where Big Tech Went Wrong and How We Can RebootNessuna valutazione finora
- Mini Farming: Self-Sufficiency on 1/4 AcreDa EverandMini Farming: Self-Sufficiency on 1/4 AcreValutazione: 4 su 5 stelle4/5 (76)
- Permaculture for the Rest of Us: Abundant Living on Less than an AcreDa EverandPermaculture for the Rest of Us: Abundant Living on Less than an AcreValutazione: 4.5 su 5 stelle4.5/5 (33)
- The Beekeeper's Lament: How One Man and Half a Billion Honey Bees Help Feed AmericaDa EverandThe Beekeeper's Lament: How One Man and Half a Billion Honey Bees Help Feed AmericaNessuna valutazione finora
- Four Battlegrounds: Power in the Age of Artificial IntelligenceDa EverandFour Battlegrounds: Power in the Age of Artificial IntelligenceValutazione: 5 su 5 stelle5/5 (5)
- Faster: How a Jewish Driver, an American Heiress, and a Legendary Car Beat Hitler's BestDa EverandFaster: How a Jewish Driver, an American Heiress, and a Legendary Car Beat Hitler's BestValutazione: 4 su 5 stelle4/5 (28)
- Pale Blue Dot: A Vision of the Human Future in SpaceDa EverandPale Blue Dot: A Vision of the Human Future in SpaceValutazione: 4.5 su 5 stelle4.5/5 (588)
- Highest Duty: My Search for What Really MattersDa EverandHighest Duty: My Search for What Really MattersNessuna valutazione finora
- How to Build a Car: The Autobiography of the World’s Greatest Formula 1 DesignerDa EverandHow to Build a Car: The Autobiography of the World’s Greatest Formula 1 DesignerValutazione: 4.5 su 5 stelle4.5/5 (54)
- ChatGPT Money Machine 2024 - The Ultimate Chatbot Cheat Sheet to Go From Clueless Noob to Prompt Prodigy Fast! Complete AI Beginner’s Course to Catch the GPT Gold Rush Before It Leaves You BehindDa EverandChatGPT Money Machine 2024 - The Ultimate Chatbot Cheat Sheet to Go From Clueless Noob to Prompt Prodigy Fast! Complete AI Beginner’s Course to Catch the GPT Gold Rush Before It Leaves You BehindNessuna valutazione finora
- The Book of the Moon: A Guide to Our Closest NeighborDa EverandThe Book of the Moon: A Guide to Our Closest NeighborValutazione: 4.5 su 5 stelle4.5/5 (11)
- From Darwin to Derrida: Selfish Genes, Social Selves, and the Meanings of LifeDa EverandFrom Darwin to Derrida: Selfish Genes, Social Selves, and the Meanings of LifeValutazione: 4 su 5 stelle4/5 (2)
- Packing for Mars: The Curious Science of Life in the VoidDa EverandPacking for Mars: The Curious Science of Life in the VoidValutazione: 4 su 5 stelle4/5 (1396)
- This Is What It Sounds Like: What the Music You Love Says About YouDa EverandThis Is What It Sounds Like: What the Music You Love Says About YouValutazione: 4 su 5 stelle4/5 (33)