Documenti di Didattica
Documenti di Professioni
Documenti di Cultura
XRD Usfull
Caricato da
pavanmech42Titolo originale
Copyright
Formati disponibili
Condividi questo documento
Condividi o incorpora il documento
Hai trovato utile questo documento?
Questo contenuto è inappropriato?
Segnala questo documentoCopyright:
Formati disponibili
XRD Usfull
Caricato da
pavanmech42Copyright:
Formati disponibili
All Answers (5)
10th Oct, 2017
Grzegorz Cios
AGH University of Science and Technology in Kraków
This is a common question, look at:
https://www.researchgate.net/post/Peak_splitting_at_high_2_theta_angle-can_anyone_help
and
https://www.researchgate.net/post/What_is_the_reason_for_peak_splitting_in_XRD_Can_reitveld_refinement_be_done_for_the_same
How about higher angle peaks, the spitting also occurs? Your case looks like Kalpha1 and Kalpha2 because the intensity of the second peak
is about a half of the first one but more detailed answers you can find in mentioned links.
2 Recommendations
10th Oct, 2017
George Glenn Peterson
University of Nebraska at Lincoln
Gevorg,
A 2nd material (element B) that alloys with the 1st material (element A) in the same crystal structure (i.e. BCC to BCC) will simply shift the
peak positions. So if you look at the XRD pattern of only element A, and the (211) peak is at 74 degrees, and you look at the XRD pattern of
only element B, and the (211) peak is at 76 degrees, then the alloy of the two materials will be somewhere between the two peak positions
depending on how much of element B is present. As for the data you have shown, I think that Grzegorz is correct in his assessment that the
peak splitting of the alloyed data of such a narrow scanning range is showing K_alpha 1 and K_alpha 2. Also, the pure Mo card looks like
calculated values to me instead of actual data. Real data will have some peak broadening due to -- well lots of variables both in the material
and the measurement system, but that is a different discussion -- as well as the peak splitting as shown in the alloyed data.
So to help you identify what the alloy is, limit yourself to BCC materials that would have a (211) peak (If I identified the Mo peak
accurately, I am going off of my memory for the rules of BCC allowed Bragg conditions) slightly higher than Mo.
Hope this helps,
George
1 Recommendation
10th Oct, 2017
P. Grima Gallardo
Centro de Investigaciones de Astronomia (CIDA)
Looking to the diffraction patterns, the first one is calculated and the second one is experimental. The first peak of experimental seems to
coincide with the third one of theoretical. The second peak seems to come from the unknown element, since, if the unknown element alloyed
with Mo is soluble (only one phase is present), you don't have any way to know what´s the element. It is very possible that the second
element (which also crystalize in a bcc structure) has the lattice parameter very close to Mo, maybe is Nb which also crystallize in bcc.
Best regards
Pedro
10th Oct, 2017
Dattatray Gadkari
Mithibai College Mumbai
Gevorg Ter-Isahakyan,
Grzegorz Cios, George Glenn Peterson, P. Grima Gallardo,
I agree with the explanation of these expertise. The XRD pattern after adding an alloy, it seems FWHM decreased due to the adding an alloy
and appeared in XRD pattern as the splitting the original peak, known as the satellite peak of unknown material. This peaks should be very
much similar lattice constant of that Mo. However, the crystallinity of the original material has been decreased after adding the unknown
material, which to be noted. So that identify the elements having closer lattice constant of Mo and that should be answer to your question
(may be similar to the suggested by P. Grima Gallardo). You are welcome to more queries !
10th Oct, 2017
Rolf Berger
Uppsala University
Obviously there is some information missing concerning the homework. I agree that the top pattern belonging to Mo is a calculated one,
seemingly from using copper radiation, but the angular resolution is too low to deduce whether it is well monochromatized or is a weighted
average of Kalpha-1 and Kalpha-2. The second pattern only shows a blowup from the (211) contribution, and it seems (as has been
commented on) that it is due to the two wavelengths. (When showing data, one should include essential information as to radiation used and
on what angular scale.) If that is so, it is easy to confirm that the two peaks belong to the same d-value from calculating the ratio between the
values of the sine-function and compare with the ratio of the wavelengths.
The second pattern cannot contain only one reflection, so it is rather strange to present a problem like that to be solved by a student without
proper information. Again, if we assume that the 2nd pattern is only part of a corresponding full pattern based on a molybdenum alloy, every
one of the reflections would show a splitting (as commented upon earlier by others) but increasingly so by increasing angle, and the only
conclusion to be drawn is that the element addition results in a cell edge decrease (seemingly an accompanying angular increase). However,
you cannot know which elements caused this, since it is then the effect of solid solubility conserving the parent phase. The same effect
would be obtained from any element that is soluble in molybdenum that has a radius less than on the Mo (the structure of the pure solute is
irrelevant). You cannot even say whether that element is a metal or a non-metal! For instance, silicon (not at all bcc) would give that effect
as well as a whole bunch of metals. The solution to your problem to find (from XRD) what the element can be is impossible if the two
peaks are indeed a resolved alpha-1/alpha-2 contribution of the (211) reflection.
If, on the other hand, the "satellite" peaks belong to another bcc-element, the alloy is a mechanical mixture of two bcc-type phases and an
identification might succeed as Pedro Gallardo indicated.
1 Recommendation
Potrebbero piacerti anche
- Shoe Dog: A Memoir by the Creator of NikeDa EverandShoe Dog: A Memoir by the Creator of NikeValutazione: 4.5 su 5 stelle4.5/5 (537)
- The Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeDa EverandThe Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeValutazione: 4 su 5 stelle4/5 (5795)
- ENGINEERING DRAWING (CSE) - Aug - 2014Documento4 pagineENGINEERING DRAWING (CSE) - Aug - 2014pavanmech42Nessuna valutazione finora
- ENGINEERING DRAWING (EEE) - Jan - 2015Documento8 pagineENGINEERING DRAWING (EEE) - Jan - 2015pavanmech42Nessuna valutazione finora
- ENGINEERING DRAWING (ME) - Aug - 2014Documento8 pagineENGINEERING DRAWING (ME) - Aug - 2014pavanmech42Nessuna valutazione finora
- Refference For Residual StressesDocumento18 pagineRefference For Residual Stressespavanmech42Nessuna valutazione finora
- "3D Printing and Design": Registration FormDocumento2 pagine"3D Printing and Design": Registration Formpavanmech42Nessuna valutazione finora
- Hidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceDa EverandHidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceValutazione: 4 su 5 stelle4/5 (895)
- The Yellow House: A Memoir (2019 National Book Award Winner)Da EverandThe Yellow House: A Memoir (2019 National Book Award Winner)Valutazione: 4 su 5 stelle4/5 (98)
- Grit: The Power of Passion and PerseveranceDa EverandGrit: The Power of Passion and PerseveranceValutazione: 4 su 5 stelle4/5 (588)
- The Little Book of Hygge: Danish Secrets to Happy LivingDa EverandThe Little Book of Hygge: Danish Secrets to Happy LivingValutazione: 3.5 su 5 stelle3.5/5 (400)
- The Emperor of All Maladies: A Biography of CancerDa EverandThe Emperor of All Maladies: A Biography of CancerValutazione: 4.5 su 5 stelle4.5/5 (271)
- Never Split the Difference: Negotiating As If Your Life Depended On ItDa EverandNever Split the Difference: Negotiating As If Your Life Depended On ItValutazione: 4.5 su 5 stelle4.5/5 (838)
- The World Is Flat 3.0: A Brief History of the Twenty-first CenturyDa EverandThe World Is Flat 3.0: A Brief History of the Twenty-first CenturyValutazione: 3.5 su 5 stelle3.5/5 (2259)
- On Fire: The (Burning) Case for a Green New DealDa EverandOn Fire: The (Burning) Case for a Green New DealValutazione: 4 su 5 stelle4/5 (74)
- Elon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureDa EverandElon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureValutazione: 4.5 su 5 stelle4.5/5 (474)
- A Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryDa EverandA Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryValutazione: 3.5 su 5 stelle3.5/5 (231)
- Team of Rivals: The Political Genius of Abraham LincolnDa EverandTeam of Rivals: The Political Genius of Abraham LincolnValutazione: 4.5 su 5 stelle4.5/5 (234)
- Devil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaDa EverandDevil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaValutazione: 4.5 su 5 stelle4.5/5 (266)
- The Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersDa EverandThe Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersValutazione: 4.5 su 5 stelle4.5/5 (345)
- The Unwinding: An Inner History of the New AmericaDa EverandThe Unwinding: An Inner History of the New AmericaValutazione: 4 su 5 stelle4/5 (45)
- The Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreDa EverandThe Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreValutazione: 4 su 5 stelle4/5 (1090)
- The Sympathizer: A Novel (Pulitzer Prize for Fiction)Da EverandThe Sympathizer: A Novel (Pulitzer Prize for Fiction)Valutazione: 4.5 su 5 stelle4.5/5 (121)
- Her Body and Other Parties: StoriesDa EverandHer Body and Other Parties: StoriesValutazione: 4 su 5 stelle4/5 (821)
- Viva QuestionsDocumento3 pagineViva QuestionssanjayshekarncNessuna valutazione finora
- Scatchell Jr. V Village of Melrose Park Et Al.Documento48 pagineScatchell Jr. V Village of Melrose Park Et Al.Gianna ScatchellNessuna valutazione finora
- Introduction To Instrumented IndentationDocumento7 pagineIntroduction To Instrumented Indentationopvsj42Nessuna valutazione finora
- Apache Hive Essentials 2nd PDFDocumento204 pagineApache Hive Essentials 2nd PDFketanmehta4u0% (1)
- Business Finance and The SMEsDocumento6 pagineBusiness Finance and The SMEstcandelarioNessuna valutazione finora
- Serological and Molecular DiagnosisDocumento9 pagineSerological and Molecular DiagnosisPAIRAT, Ella Joy M.Nessuna valutazione finora
- Why Do Firms Do Basic Research With Their Own Money - 1989 - StudentsDocumento10 pagineWhy Do Firms Do Basic Research With Their Own Money - 1989 - StudentsAlvaro Rodríguez RojasNessuna valutazione finora
- Anindya Anticipatory BailDocumento9 pagineAnindya Anticipatory BailYedlaNessuna valutazione finora
- Barangay Sindalan v. CA G.R. No. 150640, March 22, 2007Documento17 pagineBarangay Sindalan v. CA G.R. No. 150640, March 22, 2007FD BalitaNessuna valutazione finora
- Motion To Strike BasedDocumento16 pagineMotion To Strike BasedForeclosure Fraud100% (1)
- Complete PDFDocumento495 pagineComplete PDFMárcio MoscosoNessuna valutazione finora
- Process of CounsellingDocumento15 pagineProcess of CounsellingSamuel Njenga100% (1)
- La Navassa Property, Sovereignty, and The Law of TerritoriesDocumento52 pagineLa Navassa Property, Sovereignty, and The Law of TerritoriesEve AthanasekouNessuna valutazione finora
- GAJ Mod 18 Ace Your InterviewDocumento12 pagineGAJ Mod 18 Ace Your InterviewAnjela SantiagoNessuna valutazione finora
- Chpater 2 PDFDocumento44 pagineChpater 2 PDFBilalNessuna valutazione finora
- Introduction To ICT EthicsDocumento8 pagineIntroduction To ICT EthicsJohn Niño FilipinoNessuna valutazione finora
- 011 - Descriptive Writing - UpdatedDocumento39 pagine011 - Descriptive Writing - UpdatedLeroy ChengNessuna valutazione finora
- Jyotish - 2003 Sri Jagannath Center - Narayan Iyer - Divisional Charts - Divining Through DivisionsDocumento36 pagineJyotish - 2003 Sri Jagannath Center - Narayan Iyer - Divisional Charts - Divining Through DivisionsDeepa MishraNessuna valutazione finora
- Article 1156 Gives The Civil Code Definition of Obligation, in Its Passive Aspect. Our Law MerelyDocumento11 pagineArticle 1156 Gives The Civil Code Definition of Obligation, in Its Passive Aspect. Our Law MerelyFeir, Alexa Mae C.Nessuna valutazione finora
- HW 2Documento2 pagineHW 2Dubu VayerNessuna valutazione finora
- ''Want To Learn To Speak Latin or Greek This (2018) Summer''Documento10 pagine''Want To Learn To Speak Latin or Greek This (2018) Summer''ThriwNessuna valutazione finora
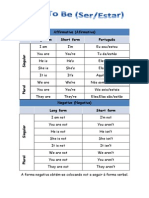
- Affirmative (Afirmativa) Long Form Short Form PortuguêsDocumento3 pagineAffirmative (Afirmativa) Long Form Short Form PortuguêsAnitaYangNessuna valutazione finora
- Strategic ManagementDocumento14 pagineStrategic ManagementvishakhaNessuna valutazione finora
- Work Teams and GroupsDocumento6 pagineWork Teams and GroupsFides AvendanNessuna valutazione finora
- City/ The Countryside: VocabularyDocumento2 pagineCity/ The Countryside: VocabularyHương Phạm QuỳnhNessuna valutazione finora
- Work Immersion Experience at Formaply Inustry in Brgy. de Ocampo, Trece Martires City, CaviteDocumento34 pagineWork Immersion Experience at Formaply Inustry in Brgy. de Ocampo, Trece Martires City, CaviteKen AshleyNessuna valutazione finora
- Large Span Structure: MMBC-VDocumento20 pagineLarge Span Structure: MMBC-VASHFAQNessuna valutazione finora
- Great Is Thy Faithfulness - Gibc Orch - 06 - Horn (F)Documento2 pagineGreat Is Thy Faithfulness - Gibc Orch - 06 - Horn (F)Luth ClariñoNessuna valutazione finora
- DND Homebrew Ideas V1 - The HomebreweryDocumento3 pagineDND Homebrew Ideas V1 - The HomebreweryKalazans CardialNessuna valutazione finora
- Sodium Borate: What Is Boron?Documento2 pagineSodium Borate: What Is Boron?Gary WhiteNessuna valutazione finora