Documenti di Didattica
Documenti di Professioni
Documenti di Cultura
MCQS'S On Solutions MARKS: 50X4 200M TIME:150MIN: Y Y X X Y Y X X
Caricato da
prathmfed0 valutazioniIl 0% ha trovato utile questo documento (0 voti)
30 visualizzazioni1 paginaThis document contains a multiple choice quiz on solutions with 4 questions. It covers topics like how vapour pressure is affected by different variables, conditions for Raoult's law to be obeyed in binary solutions, relationships between mole fractions of components in liquid and vapour phases of ideal solutions, and using mole fractions to plot the vapor-liquid equilibrium for an ideal binary solution. The quiz is worth a total of 200 marks and must be completed within 150 minutes.
Descrizione originale:
mcq's
Titolo originale
solution
Copyright
© © All Rights Reserved
Formati disponibili
DOCX, PDF, TXT o leggi online da Scribd
Condividi questo documento
Condividi o incorpora il documento
Hai trovato utile questo documento?
Questo contenuto è inappropriato?
Segnala questo documentoThis document contains a multiple choice quiz on solutions with 4 questions. It covers topics like how vapour pressure is affected by different variables, conditions for Raoult's law to be obeyed in binary solutions, relationships between mole fractions of components in liquid and vapour phases of ideal solutions, and using mole fractions to plot the vapor-liquid equilibrium for an ideal binary solution. The quiz is worth a total of 200 marks and must be completed within 150 minutes.
Copyright:
© All Rights Reserved
Formati disponibili
Scarica in formato DOCX, PDF, TXT o leggi online su Scribd
0 valutazioniIl 0% ha trovato utile questo documento (0 voti)
30 visualizzazioni1 paginaMCQS'S On Solutions MARKS: 50X4 200M TIME:150MIN: Y Y X X Y Y X X
Caricato da
prathmfedThis document contains a multiple choice quiz on solutions with 4 questions. It covers topics like how vapour pressure is affected by different variables, conditions for Raoult's law to be obeyed in binary solutions, relationships between mole fractions of components in liquid and vapour phases of ideal solutions, and using mole fractions to plot the vapor-liquid equilibrium for an ideal binary solution. The quiz is worth a total of 200 marks and must be completed within 150 minutes.
Copyright:
© All Rights Reserved
Formati disponibili
Scarica in formato DOCX, PDF, TXT o leggi online su Scribd
Sei sulla pagina 1di 1
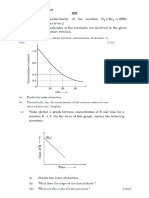
MCQS’S ON SOLUTIONS MARKS: 50X4=200M TIME:150MIN
1. The vapour pressure of a given liquid will
decrease if:
a) surface area of liquid is decreased.
b) The volume of the liquid in the container
is decreased.
c) The volume of vapour phase is increased.
d) The temperature is decreased.
2. Raoult’s law is obeyed by each constituent
of a binary liquid solution when:
a) the forces of attraction between like
molecules are greater than those between
unlike molecules.
b) The forces of attraction between like
molecules are smaller than those between
unlike molecules.
c) The forces of attraction between like
molecules are identical with those between
unlike molecules.
d) The volume occupied by unlike molecules
are different.
3. For an ideal binary liquid solution with
PA0> PB0, which relation between xA and yA is
correct? ( xA and yA are mole fraction of
component A in liquid phase and vapour
phase respectively.)
a) yA > yB b) xA > xB
Y𝐴 X𝐴 Y X𝐴
c) Y𝐵
> X𝐵
d) Y 𝐴 < X𝐵
𝐵
4. An ideal solution is formed by mixing two
volatile liquids A and B. XA and XB are the
mole fractions of A and B respectively in the
solution and YA and YB are the mole
fractions of A and B respectively in the
1
vapour phase. A plot of along y-axis
Y𝐴
1
against X𝐴
along x-axis gives a straight
line. What is the slope of straight line?
A)
Potrebbero piacerti anche
- Chemistry Class 12 Revision MaterialDocumento52 pagineChemistry Class 12 Revision Materialkrish dabhiNessuna valutazione finora
- Class XII - Study Material - ChemistryDocumento53 pagineClass XII - Study Material - ChemistryUnwantedNessuna valutazione finora
- Mass Transfer 1 CLB 20804Documento54 pagineMass Transfer 1 CLB 20804muhammad fazmiNessuna valutazione finora
- Lect.12 Ion Exchange FinalDocumento39 pagineLect.12 Ion Exchange FinalDr.D.SELVI100% (5)
- Solution Appendix-Ii: Vapour Pressure of Pure Liquid and SolutionDocumento4 pagineSolution Appendix-Ii: Vapour Pressure of Pure Liquid and SolutionSrijan JaiswalNessuna valutazione finora
- Assignment 2 (7.4.2022) - 24921131Documento5 pagineAssignment 2 (7.4.2022) - 24921131Harshwardhan Singh Thakur 7Nessuna valutazione finora
- 3.4.THERMODYNAMICS OF SOLUTIONS - SOME BASIC CONCEPTS - BusmeonDocumento5 pagine3.4.THERMODYNAMICS OF SOLUTIONS - SOME BASIC CONCEPTS - BusmeonAJ Kent Ray BusmeonNessuna valutazione finora
- CBSE Class 12 Chem Notes Question Bank Solutions PDFDocumento16 pagineCBSE Class 12 Chem Notes Question Bank Solutions PDFMonika AdhikariNessuna valutazione finora
- SolutionsDocumento5 pagineSolutionsPranav ShinojNessuna valutazione finora
- Solutions - Practice Sheet - VIJETA SERIES CLASS-12THDocumento6 pagineSolutions - Practice Sheet - VIJETA SERIES CLASS-12THpragy SinhaNessuna valutazione finora
- Soal Titrasi Asam Basa1 (Annisa Rahmah 17176001)Documento3 pagineSoal Titrasi Asam Basa1 (Annisa Rahmah 17176001)annisa rahmahNessuna valutazione finora
- 12 Chemistry English 2020 21Documento360 pagine12 Chemistry English 2020 21Aanchal SaranNessuna valutazione finora
- Solutions CLASS 12 PDF QUESTIONSDocumento35 pagineSolutions CLASS 12 PDF QUESTIONSJampa SaicharanNessuna valutazione finora
- Properties of MixtureDocumento26 pagineProperties of MixtureDuy Anh ĐàoNessuna valutazione finora
- Differential EquationsDocumento25 pagineDifferential EquationsNilo Daniel MielesNessuna valutazione finora
- Chemistry (PT-2) : Topic: SolutionDocumento2 pagineChemistry (PT-2) : Topic: SolutionchitranshNessuna valutazione finora
- Solutions NCERTDocumento16 pagineSolutions NCERTPrecisive OneNessuna valutazione finora
- Topic:-: SolutionsDocumento3 pagineTopic:-: SolutionsGnaneshwarNessuna valutazione finora
- Learning Materials of Chemistry For Board ExamDocumento80 pagineLearning Materials of Chemistry For Board Examnwork0274Nessuna valutazione finora
- Solution Tsd3Documento3 pagineSolution Tsd3Mahendra RajpootNessuna valutazione finora
- Solutions+ +Top+Practice+Problems++ (19!04!2021)Documento57 pagineSolutions+ +Top+Practice+Problems++ (19!04!2021)Ritheesh NagarajanNessuna valutazione finora
- SOLUTIONSDocumento15 pagineSOLUTIONSbijjaljosephNessuna valutazione finora
- Solution Learning PointsDocumento19 pagineSolution Learning Pointstiwari_anunay1689Nessuna valutazione finora
- 12 Chemistry Keypoints Revision Questions Chapter 2Documento15 pagine12 Chemistry Keypoints Revision Questions Chapter 2aesthetic rushNessuna valutazione finora
- Test Bank SolutionsDocumento47 pagineTest Bank SolutionsMohammed AhmedNessuna valutazione finora
- PDF PDFDocumento3 paginePDF PDFAshwin NairNessuna valutazione finora
- Gcesoln 7Documento7 pagineGcesoln 7api-3734333Nessuna valutazione finora
- Distillation ChEN DLSUDocumento2 pagineDistillation ChEN DLSUJelain HumarangNessuna valutazione finora
- MostlikelyquestionsDocumento19 pagineMostlikelyquestionsendtimes066xNessuna valutazione finora
- Study Material Chemistry 2022-23Documento166 pagineStudy Material Chemistry 2022-23Akash Kumar UpadhyayNessuna valutazione finora
- (Viii) Formality (F) - : Mass of The Ionic Solute in Gram Per Litre Formula Mass of The SolutionDocumento1 pagina(Viii) Formality (F) - : Mass of The Ionic Solute in Gram Per Litre Formula Mass of The SolutionAakash MalviyaNessuna valutazione finora
- CH 2 1Documento15 pagineCH 2 1Mohammed AmmaarNessuna valutazione finora
- Wa0006.Documento3 pagineWa0006.YOGA SAI GUHANNessuna valutazione finora
- 14A-Chapter 14, Secs 14.1 - 14.7 BlackDocumento18 pagine14A-Chapter 14, Secs 14.1 - 14.7 BlackArunNessuna valutazione finora
- lbc2 PDFDocumento9 paginelbc2 PDFKwin CaragNessuna valutazione finora
- Ion Exchange - Treatment-2Documento86 pagineIon Exchange - Treatment-2Shabbir OsmaniNessuna valutazione finora
- Xicbse-Chemistry Asst 2Documento3 pagineXicbse-Chemistry Asst 2tanishkakannan3253Nessuna valutazione finora
- Reverse Osmosis.: EnglishDocumento27 pagineReverse Osmosis.: EnglishGøbindNessuna valutazione finora
- 6.4 Solutions Edited 2020Documento62 pagine6.4 Solutions Edited 2020Anisha Syazwana Binti RoslyNessuna valutazione finora
- Unit 2 Solutions: Points To RememberDocumento15 pagineUnit 2 Solutions: Points To RememberHarpreet kaurNessuna valutazione finora
- Target 1 - Level 2 - Chapter 2 PDFDocumento26 pagineTarget 1 - Level 2 - Chapter 2 PDFRohan ArekatlaNessuna valutazione finora
- 6645646Documento2 pagine6645646honeylet tayactacNessuna valutazione finora
- 03ADifusion Mass TransferDocumento22 pagine03ADifusion Mass TransferIhya UlumudinNessuna valutazione finora
- Exemplar SolutionsDocumento20 pagineExemplar Solutionsdrishtidalakoti24001Nessuna valutazione finora
- Units of SolutionsDocumento17 pagineUnits of SolutionsamoNessuna valutazione finora
- CH1 Soution HHW Worksheet1Documento6 pagineCH1 Soution HHW Worksheet1Aaditya SharmaNessuna valutazione finora
- ChemDocumento9 pagineChemSagar SharmaNessuna valutazione finora
- MRT MDocumento8 pagineMRT MSrijan JaiswalNessuna valutazione finora
- Alizar, PH.D: Fakultas Matematika Dan Ilmu Pengetahuan Alam Universitas Negeri PadangDocumento27 pagineAlizar, PH.D: Fakultas Matematika Dan Ilmu Pengetahuan Alam Universitas Negeri PadangNathanNessuna valutazione finora
- Concentration Units (Theory For Lesson N 3)Documento7 pagineConcentration Units (Theory For Lesson N 3)Shahebaz KhanNessuna valutazione finora
- Warmup 20222Documento2 pagineWarmup 20222rania khasawnehNessuna valutazione finora
- 1.a. Fundamentals of Mass TransferDocumento17 pagine1.a. Fundamentals of Mass TransferThaer MarwanNessuna valutazione finora
- Solution AssignmentDocumento4 pagineSolution AssignmentAnkitha shajiNessuna valutazione finora
- Sheet - 01 (Solution) Liquid Theory NJ - 247Documento31 pagineSheet - 01 (Solution) Liquid Theory NJ - 247Bhola SolankiNessuna valutazione finora
- Chemistry: SolutionDocumento68 pagineChemistry: SolutionSatyajit RoutNessuna valutazione finora
- Mcq-Sol, Elctro, Chemical Kine PDFDocumento24 pagineMcq-Sol, Elctro, Chemical Kine PDFTaranjot SinghNessuna valutazione finora
- NEET UG Chemistry Solutions PDFDocumento31 pagineNEET UG Chemistry Solutions PDFAlexa Siddhi0% (1)
- Solution NotesDocumento110 pagineSolution NotesMomosNessuna valutazione finora
- Holiday Homework Class Xii-ADocumento42 pagineHoliday Homework Class Xii-Ageetanjaliawasthi375Nessuna valutazione finora
- DPP-01 Chemical KineticsDocumento1 paginaDPP-01 Chemical Kineticsprathmfed100% (1)
- Assignment On Solid State, Solutions and KineticsDocumento12 pagineAssignment On Solid State, Solutions and KineticsprathmfedNessuna valutazione finora
- QSN Bank On Aldehydes 2012-2022Documento26 pagineQSN Bank On Aldehydes 2012-2022prathmfedNessuna valutazione finora
- Chemical Kinetics 2020-2022Documento6 pagineChemical Kinetics 2020-2022prathmfedNessuna valutazione finora
- QSN Bank On Coordination Compounds 2012-2022Documento10 pagineQSN Bank On Coordination Compounds 2012-2022prathmfedNessuna valutazione finora
- Cblechpl 06125Documento9 pagineCblechpl 06125prathmfedNessuna valutazione finora
- Assignment On Aldehydes-Ketones-Carboxylic AcidsDocumento2 pagineAssignment On Aldehydes-Ketones-Carboxylic AcidsprathmfedNessuna valutazione finora
- Assignment On Coordination CompoundsDocumento8 pagineAssignment On Coordination CompoundsprathmfedNessuna valutazione finora
- Assignment On Nomenclature of Aldehyde-KetonesDocumento2 pagineAssignment On Nomenclature of Aldehyde-KetonesprathmfedNessuna valutazione finora
- Chemical Kinetics (Order/First Order/Half - Life) Code: 2019-20/3Documento1 paginaChemical Kinetics (Order/First Order/Half - Life) Code: 2019-20/3prathmfedNessuna valutazione finora
- Classification of PlantsDocumento12 pagineClassification of Plantsprathmfed100% (1)
- Jee Advanced Test On EquilibriaDocumento2 pagineJee Advanced Test On EquilibriaprathmfedNessuna valutazione finora
- DPP 02Documento1 paginaDPP 02prathmfedNessuna valutazione finora
- Qsns On Chemical KineticsDocumento1 paginaQsns On Chemical KineticsprathmfedNessuna valutazione finora
- Lesson 7 EnergyDocumento16 pagineLesson 7 EnergyprathmfedNessuna valutazione finora
- Yet QSNS On Mole ConceptDocumento2 pagineYet QSNS On Mole ConceptprathmfedNessuna valutazione finora
- School Lesson PlanDocumento17 pagineSchool Lesson PlanprathmfedNessuna valutazione finora
- Lesson 2 WorkDocumento15 pagineLesson 2 WorkprathmfedNessuna valutazione finora
- Niit Most Effective Social Media Platform TodayDocumento24 pagineNiit Most Effective Social Media Platform Todayprathmfed50% (4)