Documenti di Didattica
Documenti di Professioni
Documenti di Cultura
PEP Report 267A: Ihs Chemical
Caricato da
Vinh Do ThanhTitolo originale
Copyright
Formati disponibili
Condividi questo documento
Condividi o incorpora il documento
Hai trovato utile questo documento?
Questo contenuto è inappropriato?
Segnala questo documentoCopyright:
Formati disponibili
PEP Report 267A: Ihs Chemical
Caricato da
Vinh Do ThanhCopyright:
Formati disponibili
`
IHS CHEMICAL
PEP Report 267A
Propane Dehydrogenation Process Technologies
October 2015 ihs.com
PEP Report
Process Economics Program
Victor Wan, Principal Analyst
Marianna Asaro, Senior Principal Analyst
IHS CHEMICAL | Process Economics Program RP267A
PEP Report 267A
Propane Dehydrogenation Process Technologies
Victor Wan, Principal Analyst
Marianna Asaro, Senior Principal Analyst
Abstract
In a propane dehydrogenation (PDH) process, propane is selectively dehydrogenated to propylene. As one
of the “on-purpose” propylene production routes, PDH has recently received much attention, and
propylene production capacity via PDH is slated to grow rapidly over the next several years. Dozens of
new PDH installations have been announced worldwide, and many of them are already under
construction. The single feed/single product feature is one of the most attractive aspects of PDH,
especially for propylene derivative producers looking to back-integrate for a secure and cost-effective
source of propylene.
Despite its simple chemistry, industrial implementation of PDH is very complicated owing to side
reactions such as deep dehydrogenation, hydrogenolysis, cracking, polymerization, and coke formation.
Important aspects in catalytic dehydrogenation of propane entail near-equilibrium conversion while
minimizing side reactions and coke formation. Historically, catalyst design breakthroughs have made
major contributions to the development of commercial PDH process technologies. Significant efforts to
improve process configurations and catalyst formulations are still being undertaken.
In this report, technological progress and the industrialization of propane dehydrogenation process
technologies are analyzed. We also develop and present process designs and preliminary economics of
propylene production by the three commercialized PDH process technologies: the CATOFIN PDH
process licensed by Lummus Technology, the Oleflex PDH process licensed by UOP and the STAR PDH
process with oxydehydrogenation licensed by ThyssenKrupp Uhde.
TM
IHS CHEMICAL
COPYRIGHT NOTICE AND DISCLAIMER © 2015 IHS. For internal use of IHS clients only.
No portion of this report may be reproduced, reused, or otherwise distributed in any form without prior written consent, with the exception of any internal client distribution as may be permitted in
the license agreement between client and IHS. Content reproduced or redistributed with IHS permission must display IHS legal notices and attributions of authorship. The information contained
herein is from sources considered reliable, but its accuracy and completeness are not warranted, nor are the opinions and analyses that are based upon it, and to the extent permitted by law,
IHS shall not be liable for any errors or omissions or any loss, damage, or expense incurred by reliance on information or any statement contained herein. In particular, please note that no
representation or warranty is given as to the achievement or reasonableness of, and no reliance should be placed on, any projections, forecasts, estimates, or assumptions, and, due to various
risks and uncertainties, actual events and results may differ materially from forecasts and statements of belief noted herein. This report is not to be construed as legal or financial advice, and
use of or reliance on any information in this publication is entirely at client’s own risk. IHS and the IHS logo are trademarks of IHS.
October 2015 i © 2015 IHS
IHS CHEMICAL | Process Economics Program RP267A
Contents
1 Introduction 1-1
2 Summary 2-1
Commercial aspects 2-1
Global propylene industry 2-1
China 2-2
North America 2-3
Propane dehydrogenation 2-3
Propylene prices 2-5
Technical aspects 2-6
Chemistry 2-7
Catalysts 2-7
Reactor systems 2-8
CB&I Lummus CATOFIN PDH process 2-9
UOP Oleflex PDH process 2-10
Uhde STAR PDH process 2-11
Economic aspects 2-12
Propylene from propane by the Lummus CATOFIN process 2-12
Propylene by the UOP Oleflex process 2-13
Propylene from propane by the Uhde STAR process 2-13
Confidence ratings 2-14
Conclusions 2-14
3 Industry status 3-1
Propylene grades for chemical manufacture 3-1
Global propylene industry 3-2
China 3-3
North America 3-5
Propylene production 3-6
Steam cracking 3-6
Refinery FCC 3-7
Propane dehydrogenation 3-7
Metathesis 3-8
Propylene transportation and storage 3-9
Propylene prices 3-9
Commercial-scale PDH plants 3-10
North America 3-11
China 3-12
Thailand 3-13
Malaysia 3-13
South Korea 3-13
Middle East 3-13
Europe 3-14
Africa 3-14
CIS and Baltic States 3-14
4 Technology review 4-1
Propylene by propane dehydrogenation (PDH) 4-1
Chemistry 4-2
Commercial processes 4-3
The UOP Oleflex process 4-4
October 2015 ii © 2015 IHS
IHS CHEMICAL | Process Economics Program RP267A
Chemistry of the Oleflex process 4-4
Development of dehydrogenation catalysis by UOP 4-7
Pt–based catalysts 4-7
Other PDH catalysts from UOP 4-9
Oleflex reactor/regenerator system 4-9
Oleflex process 4-13
Alternative UOP reactor and process schemes for PDH 4-15
Fluidized bed reactors 4-15
Integration of Oleflex with FCC 4-17
Oxidative dehydrogenation 4-17
The CB&I/LUMMUS CATOFIN process 4-18
Chemistry of the CATOFIN process 4-18
CATOFIN reactor, regeneration, and heat management system 4-19
Development of CATOFIN dehydrogenation catalysis and process 4-20
CATOFIN process configuration 4-22
The Uhde STAR process 4-24
Chemistry of the STAR process 4-24
Development of dehydrogenation catalysis by Phillips Petroleum 4-29
Reactor system for oxydehydrogenation 4-34
Feedstock for the STAR process 4-36
Configuration and operating conditions of the STAR process 4-37
Linde/BASF/Statoil/Borealis PDH process 4-38
FBD–3 process 4-39
5 Propylene from propane by the CATOFIN process 5-1
Process description 5-1
Propane dehydrogenation 5-1
Product separation 5-2
Compression and low temperature gas separation 5-2
Recovery and purification of propylene 5-3
Refrigeration 5-4
Process discussion 5-4
Catalyst and chemicals 5-5
Low temperature gas separation 5-6
Steam system configuration 5-6
Materials of construction 5-6
Integration with a polypropylene plant 5-7
Process economics 5-11
Capital cost 5-11
Production cost 5-11
Profitability 5-12
6 Propylene from propane by the Oleflex process 6-1
Process description 6-1
Propane dehydrogenation (Section 100) 6-2
Feed treatment and heavies removal 6-2
PDH reaction and continuous catalyst regeneration 6-2
Transfer of catalyst between PDH reactors 6-3
Continuous catalyst regeneration 6-3
Product recovery (Section 200) 6-5
Gas separation and hydrogen purification 6-5
SHP and fractionation 6-5
Process discussion 6-6
October 2015 iii © 2015 IHS
IHS CHEMICAL | Process Economics Program RP267A
Feedstocks and products 6-7
Catalyst 6-7
Reactor-regenerator 6-8
SHP reactor 6-9
C3 splitter heat pump design 6-9
Materials of construction 6-9
Environmental and safety aspects 6-9
PDH reactor regeneration vent gas 6-9
PDH catalyst 6-10
Dryer regeneration off-gas 6-10
Process economics 6-18
Capital cost 6-18
Production cost 6-18
Profitability 6-19
7 Propylene from propane by the Uhde STAR process with oxydehydrogenation 7-1
Process description 7-1
Feed pretreatment and reaction section 7-1
Feed treatment and heavies removal 7-2
Reactors (STAR reformer and oxyreactor) 7-2
Recovery and purification of propylene 7-4
Compression 7-4
CO2 removal 7-4
Cold box 7-5
Fractionation 7-5
Refrigerant systems 7-6
Process discussion 7-6
Catalytic dehydrogenation 7-6
Catalyst regeneration 7-7
Recovery and purification of propylene 7-9
Process economics 7-18
Capital cost 7-18
Production cost 7-18
Profitability 7-19
Design conditions B-1
Cost bases B-1
Capital investment B-1
Production costs B-2
Effect of operating level on production costs B-2
Feedstock and Energy Pricing B-3
Appendix A: Patent summary table A-1
Appendix B: Design and cost bases B-1
Appendix C: Cited references C-1
Appendix D: Patent references by company D-1
Appendix E: Confidence ratings E-1
Appendix F: Process flow diagrams F-1
October 2015 iv © 2015 IHS
IHS CHEMICAL | Process Economics Program RP267A
Tables
Table 2.1 World prices for propylene 2-5
Table 2.2: Summary economics of propane dehydrogenation process technologies 2-14
Table 3.1 Propylene grades for chemical manufacture 3-2
Table 3.2 World prices for propylene 3-10
Table 4.1 Process parameters for commercial PDH technologies 4-4
Table 4.2 UOP platinum dehydrogenation catalysts over time 4-8
®
Table 4.3 Chronology of representative forerunner patents for the STAR catalyst technology 4-30
Table 5.1 Propylene from propane by the CATOFIN processDesign bases and assumption 5-7
Table 5.2 Propylene from propane by the CATOFIN processStream flows 5-8
Table 5.3 Propylene from propane by the CATOFIN processMajor equipment 5-9
Table 5.4 Propylene from propane by the CATOFIN processUtilities summary 5-10
Table 5.5 Propylene from propane by the CATOFIN processTotal capital investment 5-13
Table 5.6 Propylene from propane by the CATOFIN processCapital investment by section 5-14
Table 5.7 Propylene from propane by the CATOFIN processProduction costs 5-15
Table 6.1 Propylene from propane by the Oleflex processDesign bases and assumptions 6-6
Table 6.2 Performances of Oleflex dehydrogenation process 6-7
Table 6.3 UOP Oleflex catalyst development 6-8
Table 6.4 Propylene from propane by the Oleflex processStream flows 6-10
Table 6.5 Propylene from propane by the Oleflex processMajor equipment 6-11
Table 6.6 Propylene from propane by the Oleflex processUtilities summary 6-13
Table 6.7 Propylene from propane by the Oleflex processTotal capital investment 6-13
Table 6.8 Propylene from propane by the Oleflex processCapital investment by section 6-15
Table 6.9 Propylene from propane by the Oleflex processProduction costs 6-16
Table 7.1 Propylene from propane by the Uhde STAR processDesign bases and assumptions 7-10
Table 7.2 Propylene from propane by the Uhde STAR processStream flows 7-11
Table 7.3 Propylene from propane by the Uhde STAR processMajor equipment 7-12
Table 7.4 Propylene from propane by the Uhde STAR processUtilities summary 7-13
Table 7.5 Propylene from propane by the Uhde STAR processTotal capital investment 7-14
Table 7.6 Propylene from propane by the Uhde STAR processCapital investment by section 7-15
Table 7.7 Propylene from propane by the Uhde STAR processProduction costs 7-16
Table A.1 Patent summaries A-1
Figures
Figure 3.1 China’s propylene self-sufficiency 3-4
Figure 4.1 Yields of various propylene production processes 4-2
Figure 4.2 Equilibrium conversion of light alkanes at 100 kPa pressure 4-3
Figure 4.3 Reactions catalyzed by platinum and acid sites during light paraffin
dehydrogenation with unmodified catalyst 4-6
Figure 4.4 Temperatures required to achieve 10% and 40% conversion of
C2–C15 paraffins at 1 atm 4-7
Figure 4.5 Conventional reactor configuration for propylene production in the Oleflex unit 4-10
Figure 4.6 Moving bed reactor with countercurrent and radial flow 4-11
Figure 4.7 Block flow diagram of conventional Oleflex process 4-14
Figure 4.8 Configuration diagram of the CATOFIN process for propylene production 4-23
Figure 4.9 Chemistry of the STAR process for propylene production 4-25
October 2015 v © 2015 IHS
IHS CHEMICAL | Process Economics Program RP267A
Figure 4.10 Effects of oxygen on conversion in the STAR process 4-26
Figure 4.11 Proposed mechanism of oxidative dehydrogenation on platinum 4-27
Figure 4.12 Catalyst preparation steps for Pt–Sn–Zn/Ca spinel PDH catalysts 4-32
Figure 4.13 Gas premixing assembly in DE 102004024957 4-35
Figure 4.14 STAR process oxyreactor assembly 4-36
Figure 4.15 Configuration diagram of the STAR process for propylene production 4-37
Figure 5.2 Reactor system 5-5
Figure 6.2 Catalyst regeneration 6-4
Figure 7.2 Both reaction trains in operation 7-8
Figure 7.3 One reaction train in regeneration 7-8
Figure 7.4 Carbon dioxide removal 7-9
Figure 5.1 Propylene from propane by the Lummus CATOFIN process 7-1
Figure 6.1 Propylene from propane by the UOP Oleflex process 7-2
Figure 7.1 Propylene from propane by the Uhde STAR process 7-3
October 2015 vi © 2015 IHS
IHS Customer Care:
Americas: +1 800 IHS CARE (+1 800 447 2273); CustomerCare@ihs.com
Europe, Middle East, and Africa: +44 (0) 1344 328 300; Customer.Support@ihs.com
Asia and the Pacific Rim: +604 291 3600; SupportAPAC@ihs.com
Potrebbero piacerti anche
- Recent Advances in Thermochemical Conversion of BiomassDa EverandRecent Advances in Thermochemical Conversion of BiomassNessuna valutazione finora
- 4244-Article Text-12672-1-10-20180730Documento15 pagine4244-Article Text-12672-1-10-20180730bilal canitezNessuna valutazione finora
- Natural Gas Processing from Midstream to DownstreamDa EverandNatural Gas Processing from Midstream to DownstreamNimir O. ElbashirNessuna valutazione finora
- PolypropyleneDocumento9 paginePolypropyleneprocesspipingdesignNessuna valutazione finora
- Hydrogen and Syngas Production and Purification TechnologiesDa EverandHydrogen and Syngas Production and Purification TechnologiesNessuna valutazione finora
- Filling The Propylene Gap On Purpose TechnologiesDocumento12 pagineFilling The Propylene Gap On Purpose Technologiesvajidqc100% (1)
- Waste and Biodiesel: Feedstocks and Precursors for CatalystsDa EverandWaste and Biodiesel: Feedstocks and Precursors for CatalystsNessuna valutazione finora
- Towards A Zero Gasoline Refinery PTQ Parts 1 2Documento15 pagineTowards A Zero Gasoline Refinery PTQ Parts 1 2Anil UpadhyaNessuna valutazione finora
- 12 PDH - Process Vora UOP PDFDocumento12 pagine12 PDH - Process Vora UOP PDFstxordNessuna valutazione finora
- Process Systems and Materials for CO2 Capture: Modelling, Design, Control and IntegrationDa EverandProcess Systems and Materials for CO2 Capture: Modelling, Design, Control and IntegrationAthanasios I. PapadopoulosNessuna valutazione finora
- Crude-to-Chemicals (CTC) : A Straight-Forward Route For A Strategic TurnDocumento29 pagineCrude-to-Chemicals (CTC) : A Straight-Forward Route For A Strategic TurnrahulNessuna valutazione finora
- Internet of Things in Oil and Gas Second EditionDa EverandInternet of Things in Oil and Gas Second EditionNessuna valutazione finora
- Simulation and Performance Analysis of Propane Propylene SplitterDocumento4 pagineSimulation and Performance Analysis of Propane Propylene SplitterAnonymous UY9QffNessuna valutazione finora
- Reactivity of Naphtha Fractions For Light Olefins ProductionDocumento14 pagineReactivity of Naphtha Fractions For Light Olefins ProductionAMANessuna valutazione finora
- Modeling and Simulation of Steam CrackersDocumento6 pagineModeling and Simulation of Steam CrackersFathan FathullahNessuna valutazione finora
- catalyticDehydrogenationLightAlkanesonMetals PDFDocumento41 paginecatalyticDehydrogenationLightAlkanesonMetals PDFjulianamochizuki100% (2)
- 2 - TDVS GopalkrishnaDocumento61 pagine2 - TDVS GopalkrishnaManav GaneshNessuna valutazione finora
- Bringing It Together: Sophie Babusiaux Leandro Labanca Marie-Amélie Lambert Remi MoniotDocumento41 pagineBringing It Together: Sophie Babusiaux Leandro Labanca Marie-Amélie Lambert Remi MoniotvietnampetrochemicalNessuna valutazione finora
- Background And: Introducti OnDocumento173 pagineBackground And: Introducti Onengr_aqeelNessuna valutazione finora
- Making Premium Alky LateDocumento25 pagineMaking Premium Alky LateChristianGuerreroNessuna valutazione finora
- RP153E Toc PDFDocumento12 pagineRP153E Toc PDFAshwani KumarNessuna valutazione finora
- FCC Propylene Production Tech PaperDocumento8 pagineFCC Propylene Production Tech PaperMaría Belén JaureguiNessuna valutazione finora
- Iocl Section 1 - Process SpecficationDocumento73 pagineIocl Section 1 - Process SpecficationYogi173Nessuna valutazione finora
- Integration of Petrochemical and Refinery Plants As An Approach To Compete in Hydrocarbon MarketDocumento31 pagineIntegration of Petrochemical and Refinery Plants As An Approach To Compete in Hydrocarbon MarketHuiJooHweeNessuna valutazione finora
- Understanding Gas Treatment FundamentalsDocumento8 pagineUnderstanding Gas Treatment Fundamentalsazotik1985Nessuna valutazione finora
- Nicholas Oligomerization PDFDocumento16 pagineNicholas Oligomerization PDFTanase DianaNessuna valutazione finora
- UOP Olefin Production Solutions BrochureDocumento2 pagineUOP Olefin Production Solutions BrochureJonas FloresNessuna valutazione finora
- Propylene Via Propane Dehydrogenation Chemical EngineeringDocumento7 paginePropylene Via Propane Dehydrogenation Chemical EngineeringWade Coleman0% (1)
- Uop Benfield DatasheetDocumento2 pagineUop Benfield DatasheetSusan Monteza GrandezNessuna valutazione finora
- Technology Economics Propylene Via Propane Dehydrogenation Part 2 PDFDocumento80 pagineTechnology Economics Propylene Via Propane Dehydrogenation Part 2 PDFHohnWu33% (3)
- 020 Gerwin Wijsman GraceDocumento20 pagine020 Gerwin Wijsman GraceNduong NguyenNessuna valutazione finora
- GTL TechnologyDocumento24 pagineGTL Technologychaitanya_scribd100% (1)
- Predictive Modeling and Optimization For An Industrial Penex Isomerization Unit A Case StudyDocumento57 paginePredictive Modeling and Optimization For An Industrial Penex Isomerization Unit A Case StudyGhazanfer AliNessuna valutazione finora
- Advances in The OCC Process For Propylene ProductionDocumento6 pagineAdvances in The OCC Process For Propylene ProductionAngel Richard MamaniNessuna valutazione finora
- Propylene E61a BDocumento107 paginePropylene E61a BMaría Belén JaureguiNessuna valutazione finora
- Dehydrogenation of Paraffins Synergies Between CatalystDocumento7 pagineDehydrogenation of Paraffins Synergies Between Catalystdiego_daveNessuna valutazione finora
- INDMAXDocumento6 pagineINDMAXswaggeroni yololo100% (1)
- Heurtey Petrochem: Coker Furnace Run LengthDocumento24 pagineHeurtey Petrochem: Coker Furnace Run LengthFayaz MohammedNessuna valutazione finora
- Techno-Economic Assessment About PropyleneDocumento6 pagineTechno-Economic Assessment About PropyleneIntratec SolutionsNessuna valutazione finora
- Co-Cracking of Mixed C4 S and LPG With N PDFDocumento14 pagineCo-Cracking of Mixed C4 S and LPG With N PDFCindy GallosNessuna valutazione finora
- Integration Naphtha DearomatizationDocumento4 pagineIntegration Naphtha Dearomatizationfanoos10000Nessuna valutazione finora
- Pygas HydrogenationDocumento38 paginePygas HydrogenationJohn CyrilNessuna valutazione finora
- Debutaniser OptimisationDocumento5 pagineDebutaniser OptimisationAntonNessuna valutazione finora
- Cargill - Corn To Polylactic AcidDocumento6 pagineCargill - Corn To Polylactic AcidI. Murali KrishnaNessuna valutazione finora
- 003what Can Be Done With Pyrolysis Gasoline Axens Presentation Ertc Pet 2003 ParisDocumento41 pagine003what Can Be Done With Pyrolysis Gasoline Axens Presentation Ertc Pet 2003 ParisJunaid FaruquiNessuna valutazione finora
- New Hydroprocessing Approaches To Increase Petrochemicals Production Rev 4Documento11 pagineNew Hydroprocessing Approaches To Increase Petrochemicals Production Rev 4nishilgeorgeNessuna valutazione finora
- Naphtha Catalytic Cracking For Propylene ProudctionDocumento5 pagineNaphtha Catalytic Cracking For Propylene ProudctionWong Yee SunNessuna valutazione finora
- Metal Oxide CatalystDocumento55 pagineMetal Oxide Catalystbhavesh moorjaniNessuna valutazione finora
- Unit-Iii Processing of Olefinic C4 and C5 Cut From Steam Cracking and Fluid Catalytic CrackingDocumento38 pagineUnit-Iii Processing of Olefinic C4 and C5 Cut From Steam Cracking and Fluid Catalytic Crackinganil kumarNessuna valutazione finora
- Economic Bottom of The Barrel Processing To Minimize Fuel Oil ProductionDocumento5 pagineEconomic Bottom of The Barrel Processing To Minimize Fuel Oil Productionfisco4rilNessuna valutazione finora
- Hydrotreater Optimization With WpheDocumento29 pagineHydrotreater Optimization With Wpheandrei12320003181Nessuna valutazione finora
- T Basak AromaticsDocumento41 pagineT Basak AromaticsDP PurwadiNessuna valutazione finora
- DMEDocumento7 pagineDMEc_vivi92Nessuna valutazione finora
- Coke Formation in The Thermal CrackinDocumento7 pagineCoke Formation in The Thermal Crackinfaez100% (1)
- New Olefin Production Technologies in SINOPECDocumento10 pagineNew Olefin Production Technologies in SINOPECTrevor J. HutleyNessuna valutazione finora
- Hydrocracking ModelDocumento6 pagineHydrocracking Modelantzlck100% (1)
- Catalytic Reformer SimDocumento10 pagineCatalytic Reformer Simmihaileditoiu2010Nessuna valutazione finora
- DME Synthesis Technology Ready For Market: © Gastech 2005Documento6 pagineDME Synthesis Technology Ready For Market: © Gastech 2005yan energiaNessuna valutazione finora
- Tỷ số air-fuel lý tưởng (14.7)Documento9 pagineTỷ số air-fuel lý tưởng (14.7)Vinh Do ThanhNessuna valutazione finora
- Ansi B16-104Documento1 paginaAnsi B16-104Monica Suarez100% (1)
- Air-Fuel Ratio, Lambda and Engine Performance: AFR M MDocumento12 pagineAir-Fuel Ratio, Lambda and Engine Performance: AFR M MVinh Do ThanhNessuna valutazione finora
- Modeling and Simulation of A Co-Current Rotary Dryer Under Steady ConditionsDocumento8 pagineModeling and Simulation of A Co-Current Rotary Dryer Under Steady ConditionsVinh Do ThanhNessuna valutazione finora
- Studies On Drying Kinetics of Solids in A Rotary DryerDocumento6 pagineStudies On Drying Kinetics of Solids in A Rotary DryerVinh Do ThanhNessuna valutazione finora
- Aoac - Methods.1.1990. MoistureDocumento2 pagineAoac - Methods.1.1990. MoistureVinh Do ThanhNessuna valutazione finora
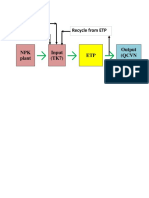
- Recycle From ETP Make Up H2O DAP, UreaDocumento1 paginaRecycle From ETP Make Up H2O DAP, UreaVinh Do ThanhNessuna valutazione finora
- Modelling and Simulation of A Direct Contact Rotary DryerDocumento16 pagineModelling and Simulation of A Direct Contact Rotary DryerVinh Do ThanhNessuna valutazione finora
- Dryer CalculationsDocumento4 pagineDryer CalculationsVinh Do Thanh0% (1)
- E. E.' " (75 Ion Agent of Firm Rope-Mckay & AssociatesDocumento7 pagineE. E.' " (75 Ion Agent of Firm Rope-Mckay & AssociatesVinh Do ThanhNessuna valutazione finora
- DRS 279-2015 Organic Fertilizer - SpecificationDocumento17 pagineDRS 279-2015 Organic Fertilizer - SpecificationVinh Do ThanhNessuna valutazione finora
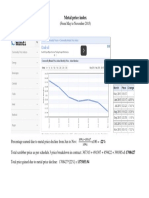
- Metal Price IndexDocumento1 paginaMetal Price IndexVinh Do ThanhNessuna valutazione finora
- 4244 12672 1 PB PDFDocumento15 pagine4244 12672 1 PB PDFVinh Do ThanhNessuna valutazione finora
- 4244 12672 1 PB PDFDocumento15 pagine4244 12672 1 PB PDFVinh Do ThanhNessuna valutazione finora
- CRACKER A PC Based Simulator For Industr PDFDocumento6 pagineCRACKER A PC Based Simulator For Industr PDFVinh Do ThanhNessuna valutazione finora
- Equivalent Grades of Cast IronsDocumento2 pagineEquivalent Grades of Cast IronsVinh Do Thanh100% (1)
- Tinh Luong Nuoc Bay HoiDocumento22 pagineTinh Luong Nuoc Bay HoiVinh Do ThanhNessuna valutazione finora
- Estimating Evaporation From Water SurfacesDocumento27 pagineEstimating Evaporation From Water SurfacesVinh Do ThanhNessuna valutazione finora
- MCCM 69 3 s192-197 KrauseDocumento6 pagineMCCM 69 3 s192-197 KrauseVinh Do ThanhNessuna valutazione finora
- Application of Excel in Psychrometric AnalysisDocumento20 pagineApplication of Excel in Psychrometric AnalysisVinh Do ThanhNessuna valutazione finora
- How Mixing Affects The Rheology of Refractory Castables - Part IIDocumento25 pagineHow Mixing Affects The Rheology of Refractory Castables - Part IIVinh Do ThanhNessuna valutazione finora
- 8D Problem Solving Worksheet: AQDEF.406Documento8 pagine8D Problem Solving Worksheet: AQDEF.406Vinh Do ThanhNessuna valutazione finora
- NRC Report On FitzPatrick Condenser LeaksDocumento15 pagineNRC Report On FitzPatrick Condenser LeaksTim KnaussNessuna valutazione finora
- DerbyDB and EclipseDocumento15 pagineDerbyDB and EclipseHessa Al-AwwadNessuna valutazione finora
- Synonym and AntonymDocumento4 pagineSynonym and Antonymjean wongNessuna valutazione finora
- 5 Methods: Mark Bevir Jason BlakelyDocumento21 pagine5 Methods: Mark Bevir Jason BlakelyGiulio PalmaNessuna valutazione finora
- Itm University, Raipur (Chhattisgarh) IndiaDocumento5 pagineItm University, Raipur (Chhattisgarh) Indiajassi7nishadNessuna valutazione finora
- Review Paper On Three Phase Fault AnalysisDocumento6 pagineReview Paper On Three Phase Fault AnalysisPritesh Singh50% (2)
- Manual WP 34s 3 1Documento211 pagineManual WP 34s 3 1richrichri100% (1)
- Job AnalysisDocumento17 pagineJob AnalysisMd. Mezba Uddin ShaonNessuna valutazione finora
- Abra State Institute of Sciences and TechnologyDocumento9 pagineAbra State Institute of Sciences and TechnologyTintin Bejarin100% (3)
- Sampling TechDocumento5 pagineSampling TechJAMZ VIBESNessuna valutazione finora
- Curriculum Implementation & EvaluationDocumento121 pagineCurriculum Implementation & Evaluationwaseem555100% (2)
- Bob Trish Duggan FoundationDocumento19 pagineBob Trish Duggan FoundationWilfried HandlNessuna valutazione finora
- TC1 Response To A Live Employer Brief: Module Code: BSOM084Documento16 pagineTC1 Response To A Live Employer Brief: Module Code: BSOM084syeda maryemNessuna valutazione finora
- Picaresque Novel B. A. Part 1 EnglishDocumento3 paginePicaresque Novel B. A. Part 1 EnglishIshan KashyapNessuna valutazione finora
- NarcissusDocumento84 pagineNarcissusAlexandra Maria NeaguNessuna valutazione finora
- Perceived Impact of Community Policing On Crime Prevention and Public Safety in Ozamiz CityDocumento7 paginePerceived Impact of Community Policing On Crime Prevention and Public Safety in Ozamiz Cityjabezgaming02Nessuna valutazione finora
- Unit - 3 Consignment: Learning OutcomesDocumento36 pagineUnit - 3 Consignment: Learning OutcomesPrathamesh KambleNessuna valutazione finora
- E. Zobel, Inc. v. CADocumento2 pagineE. Zobel, Inc. v. CAYllisa ZambranoNessuna valutazione finora
- Ptfe ManufacturingDocumento16 paginePtfe ManufacturingKokki Kumar Dpce100% (1)
- A Comprehensive Study of Types of Conditionals in LinguisticsDocumento4 pagineA Comprehensive Study of Types of Conditionals in LinguisticsRyan CortezNessuna valutazione finora
- Immunology: 'S CompanionsDocumento74 pagineImmunology: 'S CompanionsRoahit RajanNessuna valutazione finora
- LESSON 1 Overview of Toeic Speaking WritingDocumento29 pagineLESSON 1 Overview of Toeic Speaking WritingPhạm Thị HuyềnNessuna valutazione finora
- ESMOE-EOST: Birth at HomeDocumento4 pagineESMOE-EOST: Birth at HomeSlindy Noty MtetwaNessuna valutazione finora
- DD McqsDocumento21 pagineDD McqsSyeda MunazzaNessuna valutazione finora
- What Is The Importance of Research in Language Development?Documento2 pagineWhat Is The Importance of Research in Language Development?Kyle Joshua VerdidaNessuna valutazione finora
- Predictor Corrector MethodsDocumento5 paginePredictor Corrector MethodsM2C7r6Nessuna valutazione finora
- N4 Fortran Codes For Computing The Space-Time Correlations of Turbulent Flow in A ChannelDocumento47 pagineN4 Fortran Codes For Computing The Space-Time Correlations of Turbulent Flow in A Channelabhi71127112Nessuna valutazione finora
- IBbusiness Revision NotesDocumento58 pagineIBbusiness Revision NotesMayankJhaNessuna valutazione finora
- TRAFFIC JUNCTION SIMULATION-projectDocumento45 pagineTRAFFIC JUNCTION SIMULATION-projectmacklyn tyan100% (2)
- Lars Part Ix - Safety Managment System Requirements-SmsDocumento24 pagineLars Part Ix - Safety Managment System Requirements-SmssebastienNessuna valutazione finora
- Periodic Tales: A Cultural History of the Elements, from Arsenic to ZincDa EverandPeriodic Tales: A Cultural History of the Elements, from Arsenic to ZincValutazione: 3.5 su 5 stelle3.5/5 (137)
- Bulk Material Handling: Practical Guidance for Mechanical EngineersDa EverandBulk Material Handling: Practical Guidance for Mechanical EngineersValutazione: 5 su 5 stelle5/5 (1)
- Pilot's Handbook of Aeronautical Knowledge (2024): FAA-H-8083-25CDa EverandPilot's Handbook of Aeronautical Knowledge (2024): FAA-H-8083-25CNessuna valutazione finora
- The Nature of Drugs Vol. 1: History, Pharmacology, and Social ImpactDa EverandThe Nature of Drugs Vol. 1: History, Pharmacology, and Social ImpactValutazione: 5 su 5 stelle5/5 (5)
- The Regenerative Grower's Guide to Garden Amendments: Using Locally Sourced Materials to Make Mineral and Biological Extracts and FermentsDa EverandThe Regenerative Grower's Guide to Garden Amendments: Using Locally Sourced Materials to Make Mineral and Biological Extracts and FermentsValutazione: 5 su 5 stelle5/5 (3)
- Chemistry for Breakfast: The Amazing Science of Everyday LifeDa EverandChemistry for Breakfast: The Amazing Science of Everyday LifeValutazione: 4.5 su 5 stelle4.5/5 (14)
- ICH Quality Guidelines: An Implementation GuideDa EverandICH Quality Guidelines: An Implementation GuideAndrew TeasdaleNessuna valutazione finora
- Well Integrity for Workovers and RecompletionsDa EverandWell Integrity for Workovers and RecompletionsValutazione: 5 su 5 stelle5/5 (3)
- Handbook of Formulating Dermal Applications: A Definitive Practical GuideDa EverandHandbook of Formulating Dermal Applications: A Definitive Practical GuideNessuna valutazione finora
- Hyperspace: A Scientific Odyssey Through Parallel Universes, Time Warps, and the 10th DimensionDa EverandHyperspace: A Scientific Odyssey Through Parallel Universes, Time Warps, and the 10th DimensionValutazione: 4.5 su 5 stelle4.5/5 (3)
- Pressure Vessels: Design, Formulas, Codes, and Interview Questions & Answers ExplainedDa EverandPressure Vessels: Design, Formulas, Codes, and Interview Questions & Answers ExplainedValutazione: 5 su 5 stelle5/5 (1)
- It's Elemental: The Hidden Chemistry in EverythingDa EverandIt's Elemental: The Hidden Chemistry in EverythingValutazione: 4 su 5 stelle4/5 (10)
- Chemistry: a QuickStudy Laminated Reference GuideDa EverandChemistry: a QuickStudy Laminated Reference GuideValutazione: 5 su 5 stelle5/5 (1)
- 1,001 Questions & Answers for the CWI Exam: Welding Metallurgy and Visual Inspection Study GuideDa Everand1,001 Questions & Answers for the CWI Exam: Welding Metallurgy and Visual Inspection Study GuideValutazione: 3.5 su 5 stelle3.5/5 (7)
- Corrosion and Materials in Hydrocarbon Production: A Compendium of Operational and Engineering AspectsDa EverandCorrosion and Materials in Hydrocarbon Production: A Compendium of Operational and Engineering AspectsNessuna valutazione finora
- Basic Digital Signal Processing: Butterworths Basic SeriesDa EverandBasic Digital Signal Processing: Butterworths Basic SeriesValutazione: 5 su 5 stelle5/5 (1)
- Post Weld Heat Treatment PWHT: Standards, Procedures, Applications, and Interview Q&ADa EverandPost Weld Heat Treatment PWHT: Standards, Procedures, Applications, and Interview Q&ANessuna valutazione finora
- Machinery's Handbook Pocket Companion: Quick Access to Basic Data & More from the 31st EditionDa EverandMachinery's Handbook Pocket Companion: Quick Access to Basic Data & More from the 31st EditionNessuna valutazione finora
- Cyber-Physical Systems: Foundations, Principles and ApplicationsDa EverandCyber-Physical Systems: Foundations, Principles and ApplicationsHoubing H. SongNessuna valutazione finora
- Monkeys, Myths, and Molecules: Separating Fact from Fiction, and the Science of Everyday LifeDa EverandMonkeys, Myths, and Molecules: Separating Fact from Fiction, and the Science of Everyday LifeValutazione: 4 su 5 stelle4/5 (1)
- The Nature of Drugs Vol. 1: History, Pharmacology, and Social ImpactDa EverandThe Nature of Drugs Vol. 1: History, Pharmacology, and Social ImpactValutazione: 5 su 5 stelle5/5 (1)
- Einstein's Fridge: How the Difference Between Hot and Cold Explains the UniverseDa EverandEinstein's Fridge: How the Difference Between Hot and Cold Explains the UniverseValutazione: 4.5 su 5 stelle4.5/5 (51)