Documenti di Didattica
Documenti di Professioni
Documenti di Cultura
Mineral o Corticoids
Caricato da
Aayush Adhikari0 valutazioniIl 0% ha trovato utile questo documento (0 voti)
20 visualizzazioni20 pagineThis document discusses mineralocorticoids and aldosterone. It describes how aldosterone receptors occur in target tissues like the kidney and bind aldosterone with high affinity. It also discusses how aldosterone increases sodium reabsorption in the kidney by increasing sodium pumps and mitochondrial enzymes. The renin-angiotensin system and its role in stimulating aldosterone production is also summarized. Primary aldosteronism or Conn's syndrome is caused by aldosterone-secreting adrenal adenomas and results in hypertension, hypokalemia, and metabolic alkalosis. Diagnosis involves low renin levels and failure of aldosterone to suppress with saline infusion.
Descrizione originale:
Rr
Copyright
© © All Rights Reserved
Formati disponibili
PDF, TXT o leggi online da Scribd
Condividi questo documento
Condividi o incorpora il documento
Hai trovato utile questo documento?
Questo contenuto è inappropriato?
Segnala questo documentoThis document discusses mineralocorticoids and aldosterone. It describes how aldosterone receptors occur in target tissues like the kidney and bind aldosterone with high affinity. It also discusses how aldosterone increases sodium reabsorption in the kidney by increasing sodium pumps and mitochondrial enzymes. The renin-angiotensin system and its role in stimulating aldosterone production is also summarized. Primary aldosteronism or Conn's syndrome is caused by aldosterone-secreting adrenal adenomas and results in hypertension, hypokalemia, and metabolic alkalosis. Diagnosis involves low renin levels and failure of aldosterone to suppress with saline infusion.
Copyright:
© All Rights Reserved
Formati disponibili
Scarica in formato PDF, TXT o leggi online su Scribd
0 valutazioniIl 0% ha trovato utile questo documento (0 voti)
20 visualizzazioni20 pagineMineral o Corticoids
Caricato da
Aayush AdhikariThis document discusses mineralocorticoids and aldosterone. It describes how aldosterone receptors occur in target tissues like the kidney and bind aldosterone with high affinity. It also discusses how aldosterone increases sodium reabsorption in the kidney by increasing sodium pumps and mitochondrial enzymes. The renin-angiotensin system and its role in stimulating aldosterone production is also summarized. Primary aldosteronism or Conn's syndrome is caused by aldosterone-secreting adrenal adenomas and results in hypertension, hypokalemia, and metabolic alkalosis. Diagnosis involves low renin levels and failure of aldosterone to suppress with saline infusion.
Copyright:
© All Rights Reserved
Formati disponibili
Scarica in formato PDF, TXT o leggi online su Scribd
Sei sulla pagina 1di 20
Mineralocorticoids
Biochemical and
Physiological Actions…
Dr. Aseem Bhattarai, M.D.
Introduction
Broad features of Mineralocorticoid Hormone (Aldosterone)
resemble those of other steroid hormones.
Receptors that bind aldosterone with high affinity occur in
the cytoplasm and nuclei of target cells. These cells include
those in the kidney, parotid and colon and also other organs
not normally thought to be targets of aldosterone action (like
the hippocampus and the heart).
These receptors have equal affinity for aldosterone, cortisol
and corticosterone and are called type I receptors to
distinguish them from the classic glucocorticoid receptor
(type II).
Introduction
Despite the much lower levels of aldosterone in the plasma,
given the fact that there are no carrier proteins for the
molecule of aldosterone unlike corticosteroids, the effective
concentration of aldosterone is (i.e. free aldosterone) is much
higher, and hence it can bind to the type I receptors in spite
of circulating steroid hormones.
An additional fail safe mechanism is also present for the
action of aldosterone. The mineralocorticoid receptor in target
tissue has absolute selectivity for aldosterone because of the
presence of the enzyme 11β hydrosteroid dehydrogenase
which converts cortisol and corticosterone to its inactive 11β
metabolites giving aldosterone unimpeded access.
Actions
Na+ from the luminal fluid bathing the apical surface of renal
cells enters passively through Na+ channels. Na+ is then
transported to the interstitial fluids through the serosal side
by the ATP dependent Na+ / K+ ATPase pump.
Aldosterone increases the number of apical membrane
pumps as well as increases the activity of several
mitochondrial enzymes leading to generation of more ATPs.
Other mechanisms, involving different aldosterone regulated
proteins are thought responsible for handling of K+ and H+.
Renin / Angiotensin
Renin is a proteolytic enzyme that is produced and stored in
the granules of the juxtaglomerular cells surrounding the
afferent arterioles of glomeruli in the kidneys.
Renin acts on the basic substrate angiotensinogen (a
circulating α2 globulin made in the liver) to form the
decapeptide angiotensin I.
Angiotensin I is then then enzymatically transformed by the
converting enzyme (ACE), present in many tissues,
particularly in the pulmonary vascular endothelium, to the
octapeptide angiotensin II by splitting off the two C-terminal
amino acids.
Renin / Angiotensin
Angiotensin II is a potent pressor agent and exerts its action
by a direct effect on arteriolar smooth muscle. In addition,
Angiotensin II also stimulates production of aldosterone by
the zona glomerulosa of the adrenal cortex. The nonapeptide,
seen in occurrence, angiotensin III, may also stimulate
aldosterone production.
Renal Renin Release is governed by four interdependent
factors:
1. The juxaglomerular cells, which are specialized
myoepithelial cells cuffing the afferent arterioles act as
miniature pressure transducers, sensing renal perfusion
pressure.
Renin / Angiotensin
For example, under conditions of a reduction in circulating
blood volume, there is a corresponding reduction in renal
perfusion pressure and therefore, in afferent arteriolar
pressure.
This is perceived by the JG cells as a decreased stretch on
afferent arteriolar walls, leading to secretion of renin within
the kidney circulation.
This results in formation of angiotensin I which is converted
to angiotensin II by the converting enzyme.
Renin / Angiotensin
Angiotensin II influences sodium homeostasis by two major
mechanisms:
1. It changes renal blood flow so as to maintain a constant
glomerular filtration rate, thereby changing the filtration
fraction of sodium and it stimulates the adrenal cortex to
release aldosterone.
2. Increasing plasma levels of aldosterone lead to increasing
renal sodium retention and thus result in expansion of
extracellular fluid volume, which, in turn, dampens the
initiating signal for renin release.
Renin Release
2. A second control mechanism for renin release centers in
the macula densa cells, a group of distal convoluted
tubular epithelial cells in direct apposition to the
juxtaglomerular cells.
• They may function as chemoreceptors, monitoring the
sodium or chloride load presented to the distal tubule,
and such information may be conveyed to the
juxtaglomerular cells, where appropriate modifications
to renin release may occur.
• Under conditions of increased delivery of filtered
sodium to the macula densa, increasing release of
renin is capable of decreasing the glomerular filtration
rate, thereby reducing the filtered load of sodium.
Renin Release
3. The sympathetic nervous system regulates the release of
renin in response to assuming the upright posture.
The mechanism is either a direct effect on the
juxtaglomerular cell to increase adenyl cyclase activity or
an indirect effect on either the juxtaglomerular or the
macula densa cells by way of a vasoconstrictive action on
the afferent arteriole.
Renin Release
4. Finally, circulating factors also influence Renin release.
• Increasing dietary potassium directly decreases renin
release, decreasing potassium intake increases renin
release. The significance of this potassium effect is
unclear.
• Angiotensin II itself can exert a negative feedback
control on renin secretion.
• Atrial Natriuretic Peptide also inhibits renin secretion.
Thus, the secretion of renin is complex, consisting of both
intrarenal and extrarenal control mechanisms.
Hyperfunction of
Mineralocorticoids
Aldosteronism…
Introduction
Aldosteronism is a syndrome associated with hypersecretion
of the major adrenal mineralocorticoid aldosterone. Primary
aldosteronism signifies that the stimulus for the excessive
aldosterone production resides within the adrenal gland; in
secondary aldosteronism, the stimulus is extra-adrenal.
Small adenomas of the glomerulosa cells can result in
primary aldosteronism (Conn’s Syndrome), the classic
manifestations of which include hypertension, hypokalemia,
hypernatremia and alkalosis. Patients with primary
aldosteronism do not have evidence of glucocorticoid
hormone excess, and plasma renin / angiotensin II levels are
typically suppressed.
Introduction
Renal artery stenosis, with the attendant decrease in
perfusion pressure, can lead to hyperplasia and hyperfunction
of the juxtaglomerular cells and cause elevated levels of renin
and angiotensin II. This action leads to secondary
aldosteronism, which resembles the primary form, except for
the elevated renin and angiotensin II levels.
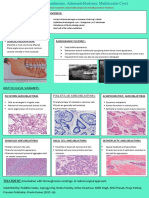
Primary Aldosteronism
• Originally, the disease was a aldosterone producing adrenal
adenoma (Conn’s Syndrome).
• Majority of cases involve a small and unilateral (with no side
predilection) adenoma. Rarely, there is an association with
adrenal carcinoma.
• Twice more common in women, (median age 30-50 years)
and comprise of 1% of unselected hypertensive patients
overall.
• Some patients have a Bilateral Cortical Nodular Hyperplasia
as the root cause, often referred to as idiopathic
hyperaldosteronism.
Signs / Symptoms
• The continual hypersecretion of aldosterone increases the
renal distal tubular exchange of intratubular sodium for the
secreted potassium and hydrogen ions, with progressive
depletion of body potassium and development of
hypokalemia.
• Most patients have diastolic hypertension, usually not of
marked severity and complain of headaches.
• Polyuria is present and associated with polydipsia.
• Muscle weakness and fatigue is related to the hypokalemia.
• Edema is characteristically absent in the absence of heart
failure or associated renal impairment.
Lab Findings
An overnight urinary concentration test often reveals the
inability to concentrate the urine, probably secondary to the
hypokalemia. Urinary pH is neutral to alkaline to compensate
for the metabolic alkalosis.
Hypokalemia may be severe (<3mmol/L) and reflects
significant potassium depletion, usually in excess of 300
mmols. Hypernatremia is due to both sodium retention and a
concomitant water loss from the polyuria.
Metabolic alkalosis is a result of hydrogen loss in the urine and
migration of hydrogen into potassium depleted cells. If
hypokalemia is severe, serum magnesium levels are also
reduced.
Total body sodium and total exchangeable sodium are usually
increased, while total exchangeable potassium is reduced.
Diagnosis
The diagnosis is suggested by a persistent hypokalemia in a
nonedematous patient on a normal sodium intake who is not
receiving potassium wasting diuretics.
The criteria for diagnosis of primary aldosteronism are:
1. Diastolic hypertension without edema
2. Hyposecretion of Renin that fail to rise despite volume loss
3. Hypersecretion of Aldosterone that fails to suppress
despite volume expansion (e.g. salt loading)
Patients with hypertension and hypokalemia may have primary
or secondary aldosteronism. The key to diagnosis is the plasma
renin activity.
Differential Diagnosis
• Hypermineralocorticoid states due to bilateral nodular
hyperplasia of the adrenals.
• DOC (Deoxycorticosterone) secreting adenomas.
• Glucocorticoid remediable aldosteronism (Defect in cortisol
biosynthesis)
• 11β Hydroxysteroid Dehydrogenase Deficiency
Treatment
• Primary aldosteronism due to an adenoma is treated by a
surgical excision. Dietary sodium restriction and
administration of an aldosterone antagonist like
spironolactone are also very effective.
• When idiopathic bilateral hyperplasia is suspected, surgery is
done only when hypokalemia is refractory. Hypertension
associated with idiopathic hyperplasia is not benefitted by
surgery.
• GRAs may be treated with glucocorticoids and
antimineralocorticoids. Patients with 11β hydroxysteroid
dehydrogenase deficiency are treated with low dose
dexamethasone.
Potrebbero piacerti anche
- ReninDocumento3 pagineReninAli GalaNessuna valutazione finora
- Renin-Angiotensin SystemDocumento5 pagineRenin-Angiotensin SystemZiedTrikiNessuna valutazione finora
- 101 OHara PDFDocumento11 pagine101 OHara PDFAntonio Lopez HernandezNessuna valutazione finora
- thuyết trìnhDocumento2 paginethuyết trìnhThanh TrúcNessuna valutazione finora
- Adrenal DisordersDocumento22 pagineAdrenal DisordersRashed ShatnawiNessuna valutazione finora
- Adrenal GlandDocumento46 pagineAdrenal GlandhSANNessuna valutazione finora
- Physiology RAASDocumento3 paginePhysiology RAASAulia Mahya FaradisaNessuna valutazione finora
- Renin - Angiotensin System RAS Renin - Angiotensin-Aldosterone System RaasDocumento3 pagineRenin - Angiotensin System RAS Renin - Angiotensin-Aldosterone System RaasReynaKatNessuna valutazione finora
- HyperkalemiaDocumento8 pagineHyperkalemiaرشاد خالد عماد Rashad Khaled EmadNessuna valutazione finora
- M5 - Electrolyte AbnormalitiesDocumento44 pagineM5 - Electrolyte AbnormalitiesVishal KumarNessuna valutazione finora
- HyperkalemiaDocumento6 pagineHyperkalemiaVijeyachandhar DorairajNessuna valutazione finora
- 6 Acute Kidney InjuryDocumento36 pagine6 Acute Kidney InjuryyaminmuhNessuna valutazione finora
- Renin-Angiotensin System: Navigation SearchDocumento5 pagineRenin-Angiotensin System: Navigation SearchArchit PatelNessuna valutazione finora
- Renal Funcetion Part 3Documento3 pagineRenal Funcetion Part 3Dr-Dalya ShakirNessuna valutazione finora
- Hyponatremia in Cirrhosis: An Update: Joseph J. Alukal, MD, Savio John, MD and Paul J. Thuluvath, MD, FRCPDocumento11 pagineHyponatremia in Cirrhosis: An Update: Joseph J. Alukal, MD, Savio John, MD and Paul J. Thuluvath, MD, FRCPMuzaffar MehdiNessuna valutazione finora
- Renal PhysiologyDocumento12 pagineRenal Physiologymina mounirNessuna valutazione finora
- ADRENAL GLAND X.PPTX - 1Documento23 pagineADRENAL GLAND X.PPTX - 1b29jcqnfn9Nessuna valutazione finora
- Jawaban PertanyaanDocumento13 pagineJawaban PertanyaantsruliantyNessuna valutazione finora
- Renin-Angiotensin System: I: (The Juxtaglomerular Apparatus)Documento1 paginaRenin-Angiotensin System: I: (The Juxtaglomerular Apparatus)reioctabianoNessuna valutazione finora
- 10 CLS 382 443 Electrolytes I Na and Fluid Balance FINALDocumento16 pagine10 CLS 382 443 Electrolytes I Na and Fluid Balance FINALamalNessuna valutazione finora
- NCM 3114 Electrolyte Imbalance-2Documento21 pagineNCM 3114 Electrolyte Imbalance-2Nicole Sherry M. CHEENessuna valutazione finora
- Lec 15 - Kidney ContinuationDocumento13 pagineLec 15 - Kidney ContinuationrajeshNessuna valutazione finora
- Control of Extracellular Fluid Volume by The KidneyppDocumento28 pagineControl of Extracellular Fluid Volume by The KidneyppLilian EdeniNessuna valutazione finora
- Med-Surg LEWIS 47 Final Foofed UpDocumento10 pagineMed-Surg LEWIS 47 Final Foofed Uphariniliaankhen100% (1)
- RAASDocumento5 pagineRAASJamaica VillonNessuna valutazione finora
- Hyperkalemia: Essentials of DiagnosisDocumento6 pagineHyperkalemia: Essentials of DiagnosisFreddy PanjaitanNessuna valutazione finora
- SiadhDocumento16 pagineSiadhGokul Rajan100% (1)
- Aldo Ster OneDocumento6 pagineAldo Ster OneZiedTrikiNessuna valutazione finora
- Primary AldosteronismDocumento31 paginePrimary AldosteronismSteph100% (1)
- Pi Is 1548559517301222Documento7 paginePi Is 1548559517301222Obito AkatsukiNessuna valutazione finora
- Renal PhysiologyDocumento7 pagineRenal PhysiologyambiliNessuna valutazione finora
- Chapter 17 Vasoactive PeptidesDocumento15 pagineChapter 17 Vasoactive Peptidesghr ramosNessuna valutazione finora
- Adrenal Cortex: Functions & Disorders: By: Post Graduate Student Dept. of Oral Medicine, Diagnosis & RadiologyDocumento80 pagineAdrenal Cortex: Functions & Disorders: By: Post Graduate Student Dept. of Oral Medicine, Diagnosis & Radiologyvihang146Nessuna valutazione finora
- CKD Part 2Documento49 pagineCKD Part 2Pao AliNessuna valutazione finora
- Surgery YbDocumento67 pagineSurgery YbTamirat geletaNessuna valutazione finora
- Mineralocorticoid - AldosteoneDocumento17 pagineMineralocorticoid - AldosteoneExplore Life ScienceNessuna valutazione finora
- 25.8 Endocrine Regulation of Kidney Function - Anatomy and Physiology 2e - OpenStaxDocumento3 pagine25.8 Endocrine Regulation of Kidney Function - Anatomy and Physiology 2e - OpenStaxMarlene AngwaforNessuna valutazione finora
- HYPERALDOSTERONISMDocumento7 pagineHYPERALDOSTERONISMMarnee Justine ColladoNessuna valutazione finora
- The Hyponatremic Patient: A Systematic Approach To Laboratory DiagnosisDocumento7 pagineThe Hyponatremic Patient: A Systematic Approach To Laboratory DiagnosisJuen LohNessuna valutazione finora
- Questions From Exam 08.09.22Documento12 pagineQuestions From Exam 08.09.22Ismo SNessuna valutazione finora
- Neurointensivismo Metabolismo Del SodioDocumento11 pagineNeurointensivismo Metabolismo Del SodioAlanNessuna valutazione finora
- Uia 11 ANAESTHESIA AND CHRONIC RENAL FAILUREDocumento4 pagineUia 11 ANAESTHESIA AND CHRONIC RENAL FAILUREfadhiliNessuna valutazione finora
- Final Assignment Anatomy & Physiology 2Documento29 pagineFinal Assignment Anatomy & Physiology 2naem nadzriNessuna valutazione finora
- Lecture 10.2-Laboratory Aspect of Electrolyte and Acid Based, DR Ira Puspitawati, SP - PK (2021)Documento76 pagineLecture 10.2-Laboratory Aspect of Electrolyte and Acid Based, DR Ira Puspitawati, SP - PK (2021)NOVITA NUR ROHMA ROHMANessuna valutazione finora
- A Systematic Approach To The Hyponatremic Patient: Iva Ratkoviê-Gusiê, Petar Kes And Vanja Baπiê-KesDocumento10 pagineA Systematic Approach To The Hyponatremic Patient: Iva Ratkoviê-Gusiê, Petar Kes And Vanja Baπiê-KesTio Prima SNessuna valutazione finora
- Renal Function TestsDocumento23 pagineRenal Function TestsKer YehunNessuna valutazione finora
- Hal 106-108Documento6 pagineHal 106-108Dirgantari PademmeNessuna valutazione finora
- Renal System PPT (Pharmacology)Documento100 pagineRenal System PPT (Pharmacology)Cheska DillupacNessuna valutazione finora
- Dr.P.Sankaranarayanan MD: Emeritus Professor of Medicine Acs Medical College & HospitalDocumento81 pagineDr.P.Sankaranarayanan MD: Emeritus Professor of Medicine Acs Medical College & HospitalvaishnaviNessuna valutazione finora
- Fluid and Electrolytes Assignment 1. What Are The Different Processes of Body Fluid and Solutes Movement? and Give at Least 2 Examples EachDocumento8 pagineFluid and Electrolytes Assignment 1. What Are The Different Processes of Body Fluid and Solutes Movement? and Give at Least 2 Examples EachAngelicaNessuna valutazione finora
- Fluid and Electrolytes Assignment 1. What Are The Different Processes of Body Fluid and Solutes Movement? and Give at Least 2 Examples EachDocumento8 pagineFluid and Electrolytes Assignment 1. What Are The Different Processes of Body Fluid and Solutes Movement? and Give at Least 2 Examples EachAngelicaNessuna valutazione finora
- Adrenal Gland - Physiology, Pathology, and Pharmacology - Lecturio PDFDocumento8 pagineAdrenal Gland - Physiology, Pathology, and Pharmacology - Lecturio PDFfaiz alamNessuna valutazione finora
- Drugs and The Kidney: Continuing Medical EducationDocumento6 pagineDrugs and The Kidney: Continuing Medical EducationJANINE PASIONNessuna valutazione finora
- Vasopressin Is A Nonapeptide Antidiuretic Hormone Involved in Modulating Various Physiological ProcessesDocumento7 pagineVasopressin Is A Nonapeptide Antidiuretic Hormone Involved in Modulating Various Physiological ProcessesyoungjbNessuna valutazione finora
- Hepato Renal SyndromeDocumento3 pagineHepato Renal SyndromepraveenbhavniNessuna valutazione finora
- ElectrolyteDocumento86 pagineElectrolyteFaryalBalochNessuna valutazione finora
- Search Termsearch Database: Limits Advanced Journal List HelpDocumento21 pagineSearch Termsearch Database: Limits Advanced Journal List Helpحسام جبار مجيدNessuna valutazione finora
- Hepatorenal Syndrome, A Simple Guide To The Condition, Diagnosis, Treatment And Related ConditionsDa EverandHepatorenal Syndrome, A Simple Guide To The Condition, Diagnosis, Treatment And Related ConditionsNessuna valutazione finora
- FHHBBCFDocumento53 pagineFHHBBCFAayush AdhikariNessuna valutazione finora
- Presented By: Aaryan Dahal (1607) Aayush Adhikari (1608)Documento50 paginePresented By: Aaryan Dahal (1607) Aayush Adhikari (1608)Aayush AdhikariNessuna valutazione finora
- Jobrpt SBONFACP2 CON01 996074935410058164384325 PDFDocumento2 pagineJobrpt SBONFACP2 CON01 996074935410058164384325 PDFAayush AdhikariNessuna valutazione finora
- ShigellaDocumento23 pagineShigellaAayush AdhikariNessuna valutazione finora
- Struktur Sel ProkariotDocumento101 pagineStruktur Sel ProkariotRizkalNessuna valutazione finora
- HandBook of Pathology and PathoPhysiology of CardioVascularDocumento321 pagineHandBook of Pathology and PathoPhysiology of CardioVascularNurwahidah Moh WahiNessuna valutazione finora
- Circulatory SystemDocumento31 pagineCirculatory SystemGeonardo100% (3)
- Bone Marrow and Stem Cell TranspplantDocumento28 pagineBone Marrow and Stem Cell TranspplantVILLEJO JHOVIALENNessuna valutazione finora
- Cell Cell InteractionDocumento45 pagineCell Cell InteractionErick Ello RodelNessuna valutazione finora
- 46 MusclesDocumento5 pagine46 Musclesuddim004100% (2)
- Blood Conservation StrategiesDocumento62 pagineBlood Conservation StrategiesHarika BandaruNessuna valutazione finora
- Chapter 4Documento17 pagineChapter 4JohnNessuna valutazione finora
- Case Report: Metastatic Parosteal Osteosarco Maina DogDocumento8 pagineCase Report: Metastatic Parosteal Osteosarco Maina DogAdrian ShakellNessuna valutazione finora
- 4LIVfunctions ElizabethDocumento23 pagine4LIVfunctions ElizabethLunaLure100% (1)
- Cells To Tissues To Organs Graphic Organizer ExampleDocumento2 pagineCells To Tissues To Organs Graphic Organizer Exampleapi-431603797Nessuna valutazione finora
- Williams Obstertics, Twenty-Second Edition - Page 619 630Documento57 pagineWilliams Obstertics, Twenty-Second Edition - Page 619 630Bharat ThapaNessuna valutazione finora
- Autonomic Nervous System - Anatomy - LecturioDocumento20 pagineAutonomic Nervous System - Anatomy - LecturioAlfred ZhikNessuna valutazione finora
- Histological Variants:: PathogenesisDocumento2 pagineHistological Variants:: PathogenesisPravin KumarNessuna valutazione finora
- Neutralization, Hemolysis and Complement Fixation: Group 9 Megino, Elvin Maturgo, Kenneth Reyes, Patricia Vergara, CyrilDocumento42 pagineNeutralization, Hemolysis and Complement Fixation: Group 9 Megino, Elvin Maturgo, Kenneth Reyes, Patricia Vergara, CyrilPatty ReyesNessuna valutazione finora
- Modul Bronkoesofagologi - 6. Disfagia OrofaringDocumento31 pagineModul Bronkoesofagologi - 6. Disfagia OrofaringNi Putu Apriliantini Arleni Putri 1802511045Nessuna valutazione finora
- 14-16 Juli 2022, Jayapura I Post Test (Jawaban)Documento10 pagine14-16 Juli 2022, Jayapura I Post Test (Jawaban)Abdul Charis KonorasNessuna valutazione finora
- A Care Study On Normal Spontaneous Vaginal Delivery Submitted To: Ms. Maria Lowela V. ElopreDocumento8 pagineA Care Study On Normal Spontaneous Vaginal Delivery Submitted To: Ms. Maria Lowela V. ElopreJhoi CastroNessuna valutazione finora
- The Plant and Animal CellsDocumento2 pagineThe Plant and Animal CellsLeny MacraNessuna valutazione finora
- Maternal Physiology During PregnancyDocumento28 pagineMaternal Physiology During PregnancyPutra MahautamaNessuna valutazione finora
- Pathophysiology of Ectopic PregnancyDocumento3 paginePathophysiology of Ectopic PregnancyJulienne Sanchez-Salazar100% (1)
- Morrell Notes (Updated)Documento129 pagineMorrell Notes (Updated)darrentse100% (1)
- Stele Types - The Arrangement of Xylem and Phloem in Stems, Rooots, and AxesDocumento30 pagineStele Types - The Arrangement of Xylem and Phloem in Stems, Rooots, and AxesYashica LalNessuna valutazione finora
- Sculpting Tut01Documento27 pagineSculpting Tut01SliverBirth86% (14)
- Bio Cheat SheetDocumento2 pagineBio Cheat Sheetboardermikie69100% (1)
- Business Communication Building Critical Skills 6th Edition Locker Test BankDocumento34 pagineBusiness Communication Building Critical Skills 6th Edition Locker Test Bankrubepazuqo88% (8)
- 0.2 Anatomía Facial - BorisDocumento11 pagine0.2 Anatomía Facial - BorisRonald Steven Reyes TellezNessuna valutazione finora
- Igcse AccommodationDocumento3 pagineIgcse AccommodationChr1zFX4Nessuna valutazione finora
- Binder 1Documento489 pagineBinder 1alzubairiyNessuna valutazione finora
- Nervous System Mutliple Choice W KeyDocumento2 pagineNervous System Mutliple Choice W KeyArvie NasayaoNessuna valutazione finora