Documenti di Didattica
Documenti di Professioni
Documenti di Cultura
Copper Bullet Lab
Caricato da
api-314065532Titolo originale
Copyright
Formati disponibili
Condividi questo documento
Condividi o incorpora il documento
Hai trovato utile questo documento?
Questo contenuto è inappropriato?
Segnala questo documentoCopyright:
Formati disponibili
Copper Bullet Lab
Caricato da
api-314065532Copyright:
Formati disponibili
Anna Chiprean
Haberman
AP Chemistry
March 16 2018
Determination of Copper in Brass Lab
Purpose:
The goal of this experiment is to determine the spectroscopically of the amount of
copper in brass shot.
Procedure:
Create .4 M Cu (NO3)2 (stock solution) out of water and copper nitrate. Dissolve the Cu
(NO3)2 in H20 and add it to a volumetric flask. Determine the value of each diluted solution,
making sure that one cuvette is filled with 10 mL of water and one cuvette is filled with 10 mL
stock solution. M1V1=M2V2 is used to find the rest of your diluted solution contents. Use the
spectrometer to measure the absorbance of you solutions. Fill the cuvettes ¾ of the way full and
record absorbance. Graph your results. To dissolve the brass bullet, determine the amount of
nitric acid. Do this step under a fume hood. Determine the amount of copper present in the brass
sample. Use the spectrometer to determine the absorbance.
Data:
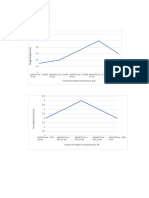
Calibration Chart for Cu(NO3)2
1.4
1.2 y = 2.9862x
R² = 0.9969
1
Absorbance
0.8
0.6
0.4
0.2
0
0 0.05 0.1 0.15 0.2 0.25 0.3 0.35 0.4 0.45
Concentration (M)
Solution Concentration Absorption Stock/Water
1 0.4 M 1.190 10 mL Stock Solution
2 0.2 M 0.582 5 mL Stock Solution
+ 5 mL H20
3 0.1 M 0.333 2.5 mL Stock
Solution + 7.5 mL
H20
4 0.05 M 0.158 1.25 mL Stock
Solution + 8.75 ml
H20
5 0.025 M 0.113 0.625 mL Stock
Solution + 9.375ml
H20
6 0.00 M 0.000 10 mL H20
Potrebbero piacerti anche
- Chemistry WorkbookDocumento18 pagineChemistry WorkbookSaleem Momin100% (1)
- Earth Mat Design GeneralDocumento62 pagineEarth Mat Design GeneralShah AlamNessuna valutazione finora
- Molarity, Molality, Normality, and Mass Percent Worksheet II Answer Key 11-12 PDFDocumento3 pagineMolarity, Molality, Normality, and Mass Percent Worksheet II Answer Key 11-12 PDFrevie100% (1)
- Normality and Molarity 3 PDFDocumento3 pagineNormality and Molarity 3 PDFMayra FlorNessuna valutazione finora
- TriboElectric SeriesDocumento3 pagineTriboElectric SeriesApurwand JfrNessuna valutazione finora
- Exercises in Introductory Physics PDFDocumento2 pagineExercises in Introductory Physics PDFChadNessuna valutazione finora
- Exp 4result Discussion For FaDocumento4 pagineExp 4result Discussion For FanasuhaNessuna valutazione finora
- Molarity, Molality, Normality, and Mass Percent Worksheet II Answer Key 11-12 PDFDocumento3 pagineMolarity, Molality, Normality, and Mass Percent Worksheet II Answer Key 11-12 PDFGerald KamulanjeNessuna valutazione finora
- Handbook On Biogas UtilizationDocumento148 pagineHandbook On Biogas UtilizationRusty MacCharles100% (3)
- CHM13P Exit Exam ReviewerDocumento9 pagineCHM13P Exit Exam ReviewerPaolo GochingcoNessuna valutazione finora
- Chm580 Experiment 1Documento9 pagineChm580 Experiment 1ohhiNessuna valutazione finora
- Molarity, Molality, Normality, and Mass Percent Worksheet II Answer Key 11-12 PDFDocumento3 pagineMolarity, Molality, Normality, and Mass Percent Worksheet II Answer Key 11-12 PDFZeferinix100% (2)
- A F (Concentratie) : 0.6 F (X) 0.107621917808219 X + 0.007476712328767 R 0.984180901225002Documento2 pagineA F (Concentratie) : 0.6 F (X) 0.107621917808219 X + 0.007476712328767 R 0.984180901225002Nastase DamianNessuna valutazione finora
- Example 1.1 Example 1.1 Example 1.1 Example 1.1 Example 1.1 Solution Solution Solution Solution SolutionDocumento1 paginaExample 1.1 Example 1.1 Example 1.1 Example 1.1 Example 1.1 Solution Solution Solution Solution Solutionjamal khanNessuna valutazione finora
- Name: Orio, Psyber V. Date: October 11, 2020 Degree/Year: BSED SCIENCE 1B NIGHT ScoreDocumento4 pagineName: Orio, Psyber V. Date: October 11, 2020 Degree/Year: BSED SCIENCE 1B NIGHT ScoreChanie Baguio Pitogo100% (1)
- Libro 1Documento2 pagineLibro 1YovanniAbadMoreiraNessuna valutazione finora
- Informe AnalisisDocumento3 pagineInforme AnalisisJesus Daniel OrtizNessuna valutazione finora
- PP Molarity and DilutionsDocumento15 paginePP Molarity and DilutionsLuisa Gardênia FariasNessuna valutazione finora
- Worksheet 2 - Chapter 07 (SOLUTION AND SUSPENSION)Documento2 pagineWorksheet 2 - Chapter 07 (SOLUTION AND SUSPENSION)Aisha AnwarNessuna valutazione finora
- Volume Pereaksi Konsentrasi 0,1 MDocumento2 pagineVolume Pereaksi Konsentrasi 0,1 MJariyah AmiliaNessuna valutazione finora
- Problem Set For PH and BuffersDocumento3 pagineProblem Set For PH and BuffersRegine Coeli Menta LansanganNessuna valutazione finora
- Lab Experiment 4Documento10 pagineLab Experiment 4Nor FazilahNessuna valutazione finora
- H-060 Thermo. Titr. Application Note No.: Title: Standardization of 0.1M Perchloric Acid in Glacial Acetic AcidDocumento3 pagineH-060 Thermo. Titr. Application Note No.: Title: Standardization of 0.1M Perchloric Acid in Glacial Acetic AcidGunjan KalyaniNessuna valutazione finora
- Iron Determination Calibration GraphsDocumento17 pagineIron Determination Calibration Graphs510418106 ARITRYAGHOSHNessuna valutazione finora
- Lab Report (Spectroscopy)Documento6 pagineLab Report (Spectroscopy)Levison Kasenga100% (2)
- Molarity, Molality, Normality, and Mass Percent Worksheet II Answer Key 11-12Documento3 pagineMolarity, Molality, Normality, and Mass Percent Worksheet II Answer Key 11-12Md HossainNessuna valutazione finora
- Analytical Chemistry First LaboratoryDocumento6 pagineAnalytical Chemistry First LaboratoryMELANIE ANTOLINNessuna valutazione finora
- Practical 1 - Spectrophotometry TechniquesDocumento13 paginePractical 1 - Spectrophotometry TechniquesDhanen DranNessuna valutazione finora
- Chem Enzyme KineticsDocumento2 pagineChem Enzyme KineticsSebastian SmytheNessuna valutazione finora
- Courbe D'etalonnageDocumento1 paginaCourbe D'etalonnageSara LovaNessuna valutazione finora
- Lab Report A2Documento7 pagineLab Report A2SumayyahNessuna valutazione finora
- Report Organic Chemistry 18125292 Liu Ni QuynhDocumento8 pagineReport Organic Chemistry 18125292 Liu Ni QuynhLe HieuNessuna valutazione finora
- EXP 5 KoloidDocumento12 pagineEXP 5 KoloidLau Yong HuiNessuna valutazione finora
- T2 Stoichiometry ET4Documento24 pagineT2 Stoichiometry ET4qishzz pqazzNessuna valutazione finora
- V V C C: Mass Ion ConcentratDocumento2 pagineV V C C: Mass Ion ConcentratJoshua RuizNessuna valutazione finora
- Results and Calculation: 3 - (Aq) - (Aq) + (Aq) 3 - (Aq) 2 (Aq)Documento20 pagineResults and Calculation: 3 - (Aq) - (Aq) + (Aq) 3 - (Aq) 2 (Aq)myzna_husna_90788547Nessuna valutazione finora
- Lab Report CHM 256Documento9 pagineLab Report CHM 256mhd sssyamilNessuna valutazione finora
- Basic Analytical Chemistry: Laboratory ReportDocumento12 pagineBasic Analytical Chemistry: Laboratory ReportCassyNessuna valutazione finora
- Medias: Yeast Mannitol AgarDocumento7 pagineMedias: Yeast Mannitol AgarKhadijaNessuna valutazione finora
- Analytical Chemistry - Experiment 2 CHM 256Documento12 pagineAnalytical Chemistry - Experiment 2 CHM 256mhd sssyamilNessuna valutazione finora
- Foster Cole 101230199 Malaïka Zarrouki 2021-01-29Documento7 pagineFoster Cole 101230199 Malaïka Zarrouki 2021-01-29Cole FosterNessuna valutazione finora
- Solution Concentration - Unit 4 ChemDocumento32 pagineSolution Concentration - Unit 4 ChemSiskaWahyuniNessuna valutazione finora
- OBS! For All Solutions, Use Sterilized Distilled Deionized Water!Documento1 paginaOBS! For All Solutions, Use Sterilized Distilled Deionized Water!Ingeniero AnonimoNessuna valutazione finora
- Alat Haspeng AndatDocumento13 pagineAlat Haspeng AndatHakamNessuna valutazione finora
- A. Perhitunganpada Proses Pembuatan Arang Aktif 1. Pembuatan Larutan H PO 10%Documento15 pagineA. Perhitunganpada Proses Pembuatan Arang Aktif 1. Pembuatan Larutan H PO 10%saidin fahmi fadillahNessuna valutazione finora
- Chem 262 Lab#2 ReportDocumento2 pagineChem 262 Lab#2 ReportAhmedNessuna valutazione finora
- ch14 PDFDocumento17 paginech14 PDFMyke AguinaldoNessuna valutazione finora
- Lab Report 4 - Dissolved Oxygen Content of Water: Winkler MethodDocumento6 pagineLab Report 4 - Dissolved Oxygen Content of Water: Winkler MethodATHALIAH JENINE TABUCLIN BANTUGNessuna valutazione finora
- Determination of Nitrate in Wastewater Using Sodium SalicylateDocumento13 pagineDetermination of Nitrate in Wastewater Using Sodium SalicylateJuan ManuelNessuna valutazione finora
- Centre For Diploma Studies (Ceds) Universiti Tun Hussein Onn Malaysia (Uthm)Documento6 pagineCentre For Diploma Studies (Ceds) Universiti Tun Hussein Onn Malaysia (Uthm)Jasni PagarautomaticNessuna valutazione finora
- Feo Determination in Rocks and Minerals: by Wet ChemistryDocumento5 pagineFeo Determination in Rocks and Minerals: by Wet ChemistryCésar VargasNessuna valutazione finora
- Practical 14 Iron Wool by Redox TitrationDocumento4 paginePractical 14 Iron Wool by Redox TitrationAngnes PoliskaNessuna valutazione finora
- Introduction To General Organic and Biochemistry 10Th Edition Bettelheim Solutions Manual Full Chapter PDFDocumento33 pagineIntroduction To General Organic and Biochemistry 10Th Edition Bettelheim Solutions Manual Full Chapter PDFarthur.hendricks257100% (14)
- Molarity - Molality and DilutionsDocumento13 pagineMolarity - Molality and DilutionsMuhammad AhmedNessuna valutazione finora
- Bab Iv.1 HasilDocumento6 pagineBab Iv.1 HasilCitra KencanaNessuna valutazione finora
- TITLEDocumento2 pagineTITLETishonna DouglasNessuna valutazione finora
- Spectrophotometric Study of Complexes by Job's Method: Report of The Minor UGC Project EntitledDocumento19 pagineSpectrophotometric Study of Complexes by Job's Method: Report of The Minor UGC Project EntitledM Irfan Khan100% (1)
- Density LabDocumento6 pagineDensity LabAlex GalangNessuna valutazione finora
- Determinarea CromuluiDocumento2 pagineDeterminarea Cromuluiramafares47Nessuna valutazione finora
- Lab Report AASDocumento8 pagineLab Report AASMohamad Saiful Mohd RaffiahNessuna valutazione finora
- 6th ChapDocumento15 pagine6th ChapAYESHA MUMTAZNessuna valutazione finora
- chm421 Exp 2 FixedDocumento8 paginechm421 Exp 2 Fixedhannannnjihh2002Nessuna valutazione finora
- Pengujian Air LimbahDocumento6 paginePengujian Air LimbahMuhammad Ilham RabbaniNessuna valutazione finora
- Experiment 5 - Data TreatmentDocumento6 pagineExperiment 5 - Data TreatmentShawn Ann SilanNessuna valutazione finora
- Religion Reflection 1Documento3 pagineReligion Reflection 1api-314065532Nessuna valutazione finora
- Walk The LineDocumento3 pagineWalk The Lineapi-314065532Nessuna valutazione finora
- Care For CreationDocumento7 pagineCare For Creationapi-314065532Nessuna valutazione finora
- Relic PaperDocumento7 pagineRelic Paperapi-314065532Nessuna valutazione finora
- Lindberg KidnappingDocumento6 pagineLindberg Kidnappingapi-314065532Nessuna valutazione finora
- Vanessa ParadisDocumento5 pagineVanessa Paradisapi-314065532Nessuna valutazione finora
- Anna ResumeDocumento1 paginaAnna Resumeapi-314065532Nessuna valutazione finora
- Napoleon BonaparteDocumento5 pagineNapoleon Bonaparteapi-314065532Nessuna valutazione finora
- ST LuciaDocumento7 pagineST Luciaapi-314065532Nessuna valutazione finora
- Publication 3Documento12 paginePublication 3api-314065532Nessuna valutazione finora
- Math HistoryDocumento3 pagineMath Historyapi-314065532Nessuna valutazione finora
- Publication 2Documento3 paginePublication 2api-314065532Nessuna valutazione finora
- Hamlet SoundtrackDocumento11 pagineHamlet Soundtrackapi-314065532Nessuna valutazione finora
- Lecture 5 2023Documento44 pagineLecture 5 2023Rana AliiNessuna valutazione finora
- Introduction To System of Linear EquationDocumento44 pagineIntroduction To System of Linear EquationEzekiel LapitanNessuna valutazione finora
- Jyothikiran TotalRecallDocumento11 pagineJyothikiran TotalRecallHARITHA H.PNessuna valutazione finora
- United States: Patent OfficeDocumento3 pagineUnited States: Patent OfficePappu PandeyNessuna valutazione finora
- Elimination ReactionDocumento89 pagineElimination ReactionManahil FatimaNessuna valutazione finora
- BS en 12671-2009Documento26 pagineBS en 12671-2009Ramakrishnan Hari50% (2)
- Week 1module StudentDocumento12 pagineWeek 1module Studentkylie jonesNessuna valutazione finora
- Post Lab FC 2Documento2 paginePost Lab FC 2Adv Sandeep SinghNessuna valutazione finora
- Study Area: 37 Page 2 of 21Documento1 paginaStudy Area: 37 Page 2 of 21Hana HananeNessuna valutazione finora
- MSDS Hypersperse MDC704Documento9 pagineMSDS Hypersperse MDC704Abdullah AbedNessuna valutazione finora
- 9 Enhanced Oil Recovery-1Documento23 pagine9 Enhanced Oil Recovery-1Muneeb AhmadNessuna valutazione finora
- Thermodynam TasksDocumento1 paginaThermodynam TasksEhtıram SeyıdovNessuna valutazione finora
- GPSA Propiedades Termodinamicas 24 PDFDocumento42 pagineGPSA Propiedades Termodinamicas 24 PDFDavid Cortez PeraltaNessuna valutazione finora
- The Problem and Its Background: Nueva Vizcaya State University Senior High School DepartmentDocumento9 pagineThe Problem and Its Background: Nueva Vizcaya State University Senior High School DepartmentJoemar SubongNessuna valutazione finora
- ConductionDocumento42 pagineConductionwassli0% (1)
- Lapocque - Transport Phenomena in Electrochemical Systems - Charge and Mass Transport in Electrochemical CellsDocumento18 pagineLapocque - Transport Phenomena in Electrochemical Systems - Charge and Mass Transport in Electrochemical CellsgiyagirlsNessuna valutazione finora
- Chemical Engineering - Fluid Flow, Heat Transfer and Mass Transfer - Vol. 1 - J. M. Coulson, J. F. Richardson, J. R. Backhurst and J. H. HarkerDocumento128 pagineChemical Engineering - Fluid Flow, Heat Transfer and Mass Transfer - Vol. 1 - J. M. Coulson, J. F. Richardson, J. R. Backhurst and J. H. HarkerFlyNarutoFly27100% (2)
- 22 15Documento4 pagine22 15Ricardo VergelNessuna valutazione finora
- Humidity Measurement: Instrumentation Engineering DepartmentDocumento24 pagineHumidity Measurement: Instrumentation Engineering DepartmentLakshmiNessuna valutazione finora
- Ameroid RSR - MSDSDocumento9 pagineAmeroid RSR - MSDSASHWINNessuna valutazione finora
- Solubility of NaClDocumento5 pagineSolubility of NaClGustavo ZagoNessuna valutazione finora
- Body Water, Osmolarity and Ionic Composition of Body Fluids: NotesDocumento12 pagineBody Water, Osmolarity and Ionic Composition of Body Fluids: NotesSuci IlhamiNessuna valutazione finora
- Are We Really Made of StarsDocumento7 pagineAre We Really Made of StarsMerlita TuralbaNessuna valutazione finora
- ESA SME Initiative Course D:MaterialsDocumento64 pagineESA SME Initiative Course D:MaterialsJoseph JonathanNessuna valutazione finora