Documenti di Didattica
Documenti di Professioni
Documenti di Cultura
Assay Determination: General Index
Caricato da
David SanabriaDescrizione originale:
Titolo originale
Copyright
Formati disponibili
Condividi questo documento
Condividi o incorpora il documento
Hai trovato utile questo documento?
Questo contenuto è inappropriato?
Segnala questo documentoCopyright:
Formati disponibili
Assay Determination: General Index
Caricato da
David SanabriaCopyright:
Formati disponibili
HANDBOOK OF GENERIC DEVELOPMENT ANDA Development
A-126-01-129Y ANALYTICAL METHOD PAGES: 10
PROCEDURES
THIS SOP IS 'SWITCHED: ON'
AZITHROMYCIN POWDER FOR ORAL SUSPENSION - 200mg Azithromycin
Dihydrate per 5mL suspension after reconstitution as Anhydrous Azithromycin
ASSAY FOR STABILITY STUDIES
ASSAY DETERMINATION
General Index:
1. PRODUCT SPECIFICATIONS
2. ASSAY BY HPLC
3. ASSAY - HPLC SETUP
4. STANDARD PREPARATION
5. SYSTEM SUITABILITY TEST
6. SYSTEM SUITABILITY CHROMATOGRAM
7. SAMPLE PREPARATION
8. PROCEDURE
9. CALCULATION
10.TYPICAL GRAPHS
Method is suitable for:
In-process control
Product Release
Stability indicating analysis
ED. N0: Effective Date : APPROVED:
NEW
Ed. Status : DD / MM / 199Y C. Latha Edanna Holman C Frost Pat West
ORIGINAL
ANALYST SUPERVISOR QC HEAD
HANDBOOK OF GENERIC DEVELOPMENT Sect: 16.32
32 Oral AZITHROMYCIN SUSPENSION
HANDBOOK OF GENERIC DEVELOPMENT ANDA Development
A-126-01-129Y ANALYTICAL METHOD PAGES: 10
PROCEDURES
THIS SOP IS 'SWITCHED: ON'
AZITHROMYCIN POWDER FOR ORAL SUSPENSION - 200mg Azithromycin
Dihydrate per 5mL suspension after reconstitution as Anhydrous Azithromycin
ASSAY FOR STABILITY STUDIES
ASSAY SPECIFICATIONS
PRODUCT: AZITHROMYCIN SUSPENSION
200 mg/ 5mL
Description Color Odor & Clarity [White to light gray] uniform homogeneous
Suspension with banana / vanilla flavor
and odor
Identification A: The Chromatogram of the sample solution
exhibits a peak with the same retention time
as the standard solution
Fill Volume [15.0 mL] (5.0%) mL Target 15.0
Fill Volume [22.5 mL] (5.0%) mL Target 22.5
pH ( 1.0 unit) Target 10.0 Limit: 9.0 - 11.0
Viscosity Target 1200 Limit: 800 - 1800 cp
Brookfield, Spindle #[ LV2] After [6]rpm
Assay [for Stability] Limit: 90.0 - 110.0% of labeled amount
[180.0] - [220.0] mg / [5] mL
Impurity determination
- Any Individual NMT 0.5% of the labeled amount
- IMP.1 NMT 0.8% of the labeled amount
- IMP.2 NMT 0.5% of the labeled amount
- Total: NMT 2.0% of the labeled amount
Container Closure system 50 mL HDPE Bottle with CRC HDPP Cap
Security Cap has twist off-tamper evident
ring
IMPURITIES KEY:-
IMP1. N-Demethylazithromycin
IMP2. Desosaminylazithromycin
ED. N0: Effective Date : APPROVED:
NEW
Ed. Status : DD / MM / 199Y C. Latha Edanna Holman C Frost Pat West
ORIGINAL
ANALYST SUPERVISOR QC HEAD
HANDBOOK OF GENERIC DEVELOPMENT Sect: 16.33
33 Oral AZITHROMYCIN SUSPENSION
HANDBOOK OF GENERIC DEVELOPMENT ANDA Development
A-126-01-129Y ANALYTICAL METHOD PAGES: 10
PROCEDURES
THIS SOP IS 'SWITCHED: ON'
AZITHROMYCIN POWDER FOR ORAL SUSPENSION - 200mg Azithromycin
Dihydrate per 5mL suspension after reconstitution as Anhydrous Azithromycin
ASSAY FOR STABILITY STUDIES
HPLC / SETUP
REFERENCE USP 23 Page 141 - 142
Pre-Column : Alumina based hydrocarboneous spherical
(ES Industries cat.no. 115271-RP1/P) particles, 5, 80A pore size, C-18, 500 x
4.6mm
Column (Waters) : Alumina based hydrocarboneous spherical
(ES Industries cat.no. 115271-RP1/P) particles, 5, 80A pore size, C-18, 500 x
4.6mm
Column Temperature : (00 2C)
Pressure : 00.0 atm
Flow rate : 0.8 mL/min. (1.2mL/min with pre-column)
Detector : Coulometric electrochemical detector
Sample volume/Loop : 50 L
Mobile Phase : Mixture of 0.02M Potassium dihydrogen
(pH = 11.0) phosphate (KH2PO4):Acetonitrile (71 : 29,
V/V) with final pH=11.01
SPECIAL NOTE AND CAUTION:
n An in-line graphite filter element must be placed before every coulometric type
electrochemical cell.
n Flow rate may be varied in order to achieve the required System Suitability
n Due to slight batch to batch variation in column packing, retention times may
change. In that case, the other system suitability parameters should be met.
n All solvents used must be of HPLC grade.
n All samples and standard solution should be protected from Light.
n Working Standard Solution can be used for 48 hrs when stored in a refrigerator.
NOTE: Degas the mobile phase by sonication for 30 minutes.
n If a Guard cell (Model 5020) is used, it is placed between the pump and the
injector, and operated at the potential of 0.05V higher than E2.
n If a conditioning cell (Model 5021) is used, it is placed immediately before the
analytical cell and operated at the same potential as E1.
n It is necessary to ensure that the column is kept at constant temperature
through the analysis time to prevent changes in the retention time
ED. N0: Effective Date : APPROVED:
NEW
Ed. Status : DD / MM / 199Y C. Latha Edanna Holman C Frost Pat West
ORIGINAL
ANALYST SUPERVISOR QC HEAD
HANDBOOK OF GENERIC DEVELOPMENT Sect: 16.34
34 Oral AZITHROMYCIN SUSPENSION
HANDBOOK OF GENERIC DEVELOPMENT ANDA Development
A-126-01-129Y ANALYTICAL METHOD PAGES: 10
PROCEDURES
THIS SOP IS 'SWITCHED: ON'
AZITHROMYCIN POWDER FOR ORAL SUSPENSION - 200mg Azithromycin
Dihydrate per 5mL suspension after reconstitution as Anhydrous Azithromycin
ASSAY FOR STABILITY STUDIES
HPLC Assay of Azithromycin
SCOPE:-
Azithromycin assay is determination by HPLC with electrochemical detection after
extraction of active Azithromycin from powder for oral suspension with Diluent.
Detection
Azithromycin is detected by dual porous graphite coulometric electrodes operated in
the oxidative screen mode.
Detector:
Coulometric electrochemical detector with dual series capability using a ESA
Coulochem II Multi-Electrode Detector with dual series porous graphite coulometric
electrodes.
Analytical Cell:
Model 5010 standard analytical cell.
Or/alternative - high sensitivity cell
Model 5011 analytical cell [high sensitivity] operated in the oxidative screen mode at
the following parameters:
Cell potential:
E1 (first electrode): 0.600-0.730V
E2 (working electrode): 0.800-0.870V
Full scale range: 1,2 or 5A
Filter: 2,5 or 10 sec
Signal output voltage: 100mV
ED. N0: Effective Date : APPROVED:
NEW
Ed. Status : DD / MM / 199Y C. Latha Edanna Holman C Frost Pat West
ORIGINAL
ANALYST SUPERVISOR QC HEAD
HANDBOOK OF GENERIC DEVELOPMENT Sect: 16.35
35 Oral AZITHROMYCIN SUSPENSION
HANDBOOK OF GENERIC DEVELOPMENT ANDA Development
A-126-01-129Y ANALYTICAL METHOD PAGES: 10
PROCEDURES
THIS SOP IS 'SWITCHED: ON'
AZITHROMYCIN POWDER FOR ORAL SUSPENSION - 200mg Azithromycin
Dihydrate per 5mL suspension after reconstitution as Anhydrous Azithromycin
ASSAY FOR STABILITY STUDIES
Diluent for internal standard:
Mixture of 0.02M Potassium dihydrogen phosphate (KH2PO4):Acetonitrile (71:29, v/v)
with final pH = 8.01.
Diluent:-
Mixture of 0.01M Potassium dihydrogen phosphate (KH2PO4):
2-propanol:Ethanol:Acetonitrile (53 : 20 : 16 : 11, V/V) with final pH = 8.42
1
Mobile phase and Diluent for internal standard preparation
n Weigh 11.6gr of Potassium dihydrogen phosphate into a large glass container (6 liter
capacity),
n Add 4260mL of HPLC grade Water and mix to dissolve.
n Add 1740mL Acetonitrile and mix well.
n Divide the solution into two portions (2:1) and adjust the pH of the larger portion (4L)
to pH=11.00.1 with 10M Potassium hydroxide.
n Filter through 0.2 membrane filter.
n This is the Mobile phase.
n Adjust the pH of the smaller portion (2L) to pH=8.0 0.1 with 10M Potassium
hydroxide.
n This is the Diluent for internal standard.
2
Diluent preparation
n Weigh 4.3gr of Potassium dihydrogen phosphate into a 6 liters glass container
n Add 3180mL of HPLC grade water and mix to dissolve.
n Add 1200mL of 2-propanol, 960mL of Ethanol and 660mL of Acetonitrile and mix
well.
n Adjust the final pH of Diluent to 8.4 with 10M Potassium hydroxide.
n This is the diluent.
NOTE: Flow rate and mobile phase proportions may be varied in order to achieve the
required system suitability.
ED. N0: Effective Date : APPROVED:
NEW
Ed. Status : DD / MM / 199Y C. Latha Edanna Holman C Frost Pat West
ORIGINAL
ANALYST SUPERVISOR QC HEAD
HANDBOOK OF GENERIC DEVELOPMENT Sect: 16.36
36 Oral AZITHROMYCIN SUSPENSION
HANDBOOK OF GENERIC DEVELOPMENT ANDA Development
A-126-01-129Y ANALYTICAL METHOD PAGES: 10
PROCEDURES
THIS SOP IS 'SWITCHED: ON'
AZITHROMYCIN POWDER FOR ORAL SUSPENSION - 200mg Azithromycin
Dihydrate per 5mL suspension after reconstitution as Anhydrous Azithromycin
ASSAY FOR STABILITY STUDIES
STANDARD PREPARATION
The Analytical Standard refers to one of the following:
Primary Reference Standard (A.S.);
Active Drug Substance analyzed by the Analytical laboratory according to a
specific monograph in comparison with current Reference Standard. After release this
material becomes an in-house working standard.
SOLUTION PREPARATION
Solution A: - Internal Standard Solution
Accurately weigh and dissolve 3 mg of Diphenhydramine hydrochloride A.S. into 2L of
diluent for internal standard.
Standard Preparation
n Accurately weigh about 23.5mg of Azithromycin dihydrate A.S. into a 50mL
volumetric flask.
n Dissolve in and dilute to volume with Diluent. Sonicate for 5 minutes.
n Pipet 5mL of the above solution into a 50mL volumetric flask and bring up to volume
with the internal standard solution.
n Dilute 5mL of the resulting solution to a 50 mL with the internal standard solution.
This is the Standard Solution.
SYSTEM SUITABILITY TEST
SYSTEM SUITABILITY TEST
n Inject the Standard Solution.
n The number of theoretical plates (calculated according to USP) for the Azithromycin
peak should be greater or equal to 1500.
n The tailing factor for both analyte peaks, Diphenhydramine and Azithromycin (when
calculated according to USP) should be not more than 2.0
n The relative standard deviation for 5 replicate injections is not more than 2.0%.
n The resolution factor between Azithromycin and Diphenhydramine peaks(when
calculated according to USP) should be not less than 1.5
ED. N0: Effective Date : APPROVED:
NEW
Ed. Status : DD / MM / 199Y C. Latha Edanna Holman C Frost Pat West
ORIGINAL
ANALYST SUPERVISOR QC HEAD
HANDBOOK OF GENERIC DEVELOPMENT Sect: 16.37
37 Oral AZITHROMYCIN SUSPENSION
HANDBOOK OF GENERIC DEVELOPMENT ANDA Development
A-126-01-129Y ANALYTICAL METHOD PAGES: 10
PROCEDURES
THIS SOP IS 'SWITCHED: ON'
AZITHROMYCIN POWDER FOR ORAL SUSPENSION - 200mg Azithromycin
Dihydrate per 5mL suspension after reconstitution as Anhydrous Azithromycin
ASSAY FOR STABILITY STUDIES
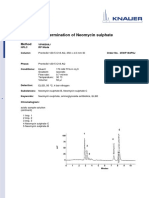
SYSTEM SUITABILITY CHROMATOGRAM
(Representative Chromatogram)
RT [1] = 5.62 min RT [2] = 9.83 min
ED. N0: Effective Date : APPROVED:
NEW
Ed. Status : DD / MM / 199Y C. Latha Edanna Holman C Frost Pat West
ORIGINAL
ANALYST SUPERVISOR QC HEAD
HANDBOOK OF GENERIC DEVELOPMENT Sect: 16.38
38 Oral AZITHROMYCIN SUSPENSION
HANDBOOK OF GENERIC DEVELOPMENT ANDA Development
A-126-01-129Y ANALYTICAL METHOD PAGES: 10
PROCEDURES
THIS SOP IS 'SWITCHED: ON'
AZITHROMYCIN POWDER FOR ORAL SUSPENSION - 200mg Azithromycin
Dihydrate per 5mL suspension after reconstitution as Anhydrous Azithromycin
ASSAY FOR STABILITY STUDIES
SAMPLE PREPARATION:
n Test THREE bottles individually for Azithromycin content as BELOW:
n Tap the Azithromycin container to initially loosen the powder.
n Reconstitute by weighing the correct volume of water into the bottle that is 9mL for
15mL final volume (after reconstitution) or 12mL for 22.5 mL final volume (after
reconstitution.)
n Shake the reconstituted container for approximately 15 minutes.
n Allow the contents of the container to stand for 30 minutes.
n Repeat Shaking procedure of the container for an additional 15 minutes.
n Shake the bottle for 20 seconds immediately prior to sampling in order to accomplish
homogeneity of suspension.
n Via a plastic syringe calibrated to 0.2mL, transfer 5mL suspension into a 500mL
volumetric flask (for the 200mg/5mL dosage constituted oral suspension)
Use a 250mL volumetric flask (for the 100mg/5mL dosage constituted oral suspension).
n When sampling with syringe, avoid inclusion of large air bubbles.
n Record the weight of the suspension aliquot delivered into the volumetric flask.
n Add about 70% volume of Diluent to the flask and shake for 30 minutes.
n Make up to volume with Diluent.
n Pipet 5mL of above solution into a 50mL volumetric flask and make up to volume with
the internal standard solution.
n Pipet 5mL of the resulting solution into a 50mL volumetric flask and make up to
volume with internal standard solution. This is the Sample solution.
n Determine the density of the freshly prepared suspension using a calibrated
pycnometer.
ED. N0: Effective Date : APPROVED:
NEW
Ed. Status : DD / MM / 199Y C. Latha Edanna Holman C Frost Pat West
ORIGINAL
ANALYST SUPERVISOR QC HEAD
HANDBOOK OF GENERIC DEVELOPMENT Sect: 16.39
39 Oral AZITHROMYCIN SUSPENSION
HANDBOOK OF GENERIC DEVELOPMENT ANDA Development
A-126-01-129Y ANALYTICAL METHOD PAGES: 10
PROCEDURES
THIS SOP IS 'SWITCHED: ON'
AZITHROMYCIN POWDER FOR ORAL SUSPENSION - 200mg Azithromycin
Dihydrate per 5mL suspension after reconstitution as Anhydrous Azithromycin
ASSAY FOR STABILITY STUDIES
PROCEDURE:
n Separately - inject equal volumes of Standard and Sample solutions into the
chromatograph.
n Record the chromatograms
n Determine the resulting heights for both the Diphenhydramine peak and the
Azithromycin peak using a suitable integrator Measure the responses for the major
peaks
n Retain chromatograms showing the calculate values in mg/mL
n NOTE: Inject the Standard solution after each third injection of the Sample solution
CALCULATION.
P e a k h e i g h t o f A z ith r o m y c in s m p
= Rsm p
P e a k h e i g h t o f D ip h e n h y d r a m in e s m p
Peak height of Azithromycin Std
= Rstd
Peak height of Diphenhydramine Std
Rsmp x Std wt 1 (mg) x F 3 x 5 x 100
= % Azithromycin of labeled claim
Rstd x Vsmp 2 (mL) x Dosage (mg / 5mL)
1
Std wt is corrected according to % Assay1and % Water in the standard.
S m p w t(g)
2
Vsmp =
D e n s ity (g / m L )
3
F - factor = 5 for dosage 100mg per 5mL
= 10 for dosage 200mg per 5mL
ED. N0: Effective Date : APPROVED:
NEW
Ed. Status : DD / MM / 199Y C. Latha Edanna Holman C Frost Pat West
ORIGINAL
ANALYST SUPERVISOR QC HEAD
HANDBOOK OF GENERIC DEVELOPMENT Sect: 16.40
40 Oral AZITHROMYCIN SUSPENSION
HANDBOOK OF GENERIC DEVELOPMENT ANDA Development
A-126-01-129Y ANALYTICAL METHOD PAGES: 10
PROCEDURES
THIS SOP IS 'SWITCHED: ON'
AZITHROMYCIN POWDER FOR ORAL SUSPENSION - 200mg Azithromycin
Dihydrate per 5mL suspension after reconstitution as Anhydrous Azithromycin
ASSAY FOR STABILITY STUDIES
CONCLUSION.
Method AMP-126-01-1298 is suitable for the assay of the active ingredient in the
dosage form.
TYPICAL CHROMATOGRAMS
(Representative Chromatogram)
RT [1] = 5.62 min RT [2] = 9.83 min
Diphenhydramine 5.62
Azithromycin 9.83
DEGRADATION PROFILE:-
Decomposition Products Found
IMP1. N-Demethylazithromycin NMT 0.8 %
IMP2. Desosaminylazithromycin NMT 0.5 %
ED. N0: Effective Date : APPROVED:
NEW
Ed. Status : DD / MM / 199Y C. Latha Edanna Holman C Frost Pat West
ORIGINAL
ANALYST SUPERVISOR QC HEAD
HANDBOOK OF GENERIC DEVELOPMENT Sect: 16.41
41 Oral AZITHROMYCIN SUSPENSION
Potrebbero piacerti anche
- Microbial EnumerationDocumento7 pagineMicrobial Enumerationvivek pattaniNessuna valutazione finora
- s1005 Cagewash OperationDocumento7 pagines1005 Cagewash OperationMalathi SathivelNessuna valutazione finora
- Print 24.1.18 Sop LlistDocumento11 paginePrint 24.1.18 Sop Llistkalaiarasi ravichandranNessuna valutazione finora
- Toxicology Procedures ManualDocumento246 pagineToxicology Procedures Manualdiyanalungu89Nessuna valutazione finora
- Specification FormDocumento5 pagineSpecification Formpowdertech100% (2)
- Sop ASP HPLCDocumento12 pagineSop ASP HPLCAngel PalominoNessuna valutazione finora
- 001 AbbrevationsDocumento86 pagine001 AbbrevationssreeniNessuna valutazione finora
- Certificate of Quality: Cayman Pharma S.R.O. Ul. Práce 657, 277 11 NERATOVICE Czech Republic Tel. +420 315 665 360Documento3 pagineCertificate of Quality: Cayman Pharma S.R.O. Ul. Práce 657, 277 11 NERATOVICE Czech Republic Tel. +420 315 665 360amin138irNessuna valutazione finora
- SOP: Retro-Orbital Blood Collection in The RatDocumento5 pagineSOP: Retro-Orbital Blood Collection in The RatSipend AnatomiNessuna valutazione finora
- Chemical Analysis of Soft WaterDocumento4 pagineChemical Analysis of Soft WaterBhawani singhNessuna valutazione finora
- Lovex RegistrDocumento64 pagineLovex RegistrbishopshehadehNessuna valutazione finora
- LASA Blood SamplingDocumento4 pagineLASA Blood SamplinggursinNessuna valutazione finora
- RabbitDocumento5 pagineRabbitapi-385448540Nessuna valutazione finora
- Is 14543. 2004 Packaged Drinking Water SpecificationsDocumento35 pagineIs 14543. 2004 Packaged Drinking Water SpecificationsRahmathNessuna valutazione finora
- DOEH Pesticide FilesDocumento20 pagineDOEH Pesticide FilesMichael_Lee_RobertsNessuna valutazione finora
- Coding - of - Pipes - 20170611 - Purified WaterDocumento3 pagineCoding - of - Pipes - 20170611 - Purified Waterreflectprakash3610Nessuna valutazione finora
- IncurredDocumento2 pagineIncurredVictor DattaNessuna valutazione finora
- Attachment 13 Analytical Equipment Calibration Certificate v3 PDFDocumento40 pagineAttachment 13 Analytical Equipment Calibration Certificate v3 PDFGnanaSunderNessuna valutazione finora
- 01 Disintegration Test For Tablets and CapsulesDocumento30 pagine01 Disintegration Test For Tablets and CapsulesmefroNessuna valutazione finora
- QC TestsDocumento12 pagineQC Testsembermich100% (1)
- Salom Pharmacy LTD: Quality Control Laboratory SPL-SOP-QC-DISS-001 Operation of Dissolution Rate ApparatusDocumento2 pagineSalom Pharmacy LTD: Quality Control Laboratory SPL-SOP-QC-DISS-001 Operation of Dissolution Rate ApparatusPaolo Peps100% (1)
- SOP For Karl Fisher Titration-Sophie - RadwanDocumento3 pagineSOP For Karl Fisher Titration-Sophie - RadwanzhobeysNessuna valutazione finora
- Check List Regarding Documents Submitted For Drug Registration PurposesDocumento8 pagineCheck List Regarding Documents Submitted For Drug Registration PurposesMayson Bali100% (1)
- Content: Standard Operating Procedure (SOP) Use and Maintenance of A FreezerDocumento7 pagineContent: Standard Operating Procedure (SOP) Use and Maintenance of A FreezerPrincess PiaNessuna valutazione finora
- Ashok ResumeDocumento5 pagineAshok ResumeRamboNessuna valutazione finora
- Related Substances of Cefipime IPDocumento4 pagineRelated Substances of Cefipime IPBhawani singhNessuna valutazione finora
- Rajiv Gandhi University of Health Sciences: ST NDDocumento10 pagineRajiv Gandhi University of Health Sciences: ST NDkumar_chemicalNessuna valutazione finora
- Raw Data Report of TabletDocumento11 pagineRaw Data Report of TabletShubam SharmaNessuna valutazione finora
- # Observations Corrective Actions: Manufacturing ControlsDocumento4 pagine# Observations Corrective Actions: Manufacturing ControlsMasthan GMNessuna valutazione finora
- Pyriproxyfen Papaya Draft ProtocolDocumento16 paginePyriproxyfen Papaya Draft ProtocolprecauteNessuna valutazione finora
- Appendix-I PQ #. 1 Sampling Point: SP001 (Feed Water Inlet) : G Io N CH Lo Rid e Ni Tra TeDocumento14 pagineAppendix-I PQ #. 1 Sampling Point: SP001 (Feed Water Inlet) : G Io N CH Lo Rid e Ni Tra TeaKureishiNessuna valutazione finora
- CEFOTAXIME Injection 1 GMDocumento119 pagineCEFOTAXIME Injection 1 GMiloveit52252Nessuna valutazione finora
- Meclizine HCLDocumento10 pagineMeclizine HCLChEng_Nessuna valutazione finora
- XYZ Tablet 20mg FCTDocumento123 pagineXYZ Tablet 20mg FCTAnonymous jzlKNORNessuna valutazione finora
- MNT 008-03 Cleaning of Overhead Potable Water Storage TankDocumento3 pagineMNT 008-03 Cleaning of Overhead Potable Water Storage TankAniket kopate100% (1)
- Annexure - VIDocumento19 pagineAnnexure - VIKiran KumarNessuna valutazione finora
- OQ Raw Materials Sample TentDocumento18 pagineOQ Raw Materials Sample TentAnup BajracharyaNessuna valutazione finora
- BME-01 Equipment Iddentification TagDocumento4 pagineBME-01 Equipment Iddentification TagPAPPU RANJITH KUMARNessuna valutazione finora
- Cefepime For Injection USP (Dossier) PDFDocumento66 pagineCefepime For Injection USP (Dossier) PDFsofiaNessuna valutazione finora
- Amendment 1: Replacement of 3Kl Uf Storage Tank & Installation of New 500 Ltr. Brine TankDocumento8 pagineAmendment 1: Replacement of 3Kl Uf Storage Tank & Installation of New 500 Ltr. Brine TankprakashNessuna valutazione finora
- Eskag Pharma Pvt. LTD., Haridwar, Unit-I Department: Quality Assurance Product MatrixDocumento20 pagineEskag Pharma Pvt. LTD., Haridwar, Unit-I Department: Quality Assurance Product MatrixMohit ChauhanNessuna valutazione finora
- Annexure - II Product DevelopmentDocumento50 pagineAnnexure - II Product DevelopmentLife PearlNessuna valutazione finora
- Sampling Dispensing BoothDocumento8 pagineSampling Dispensing BoothAndreas StathatosNessuna valutazione finora
- Inspection QueriesDocumento3 pagineInspection Queriesmuhammad qasimNessuna valutazione finora
- VeerDocumento19 pagineVeerRam KprNessuna valutazione finora
- Internal Quality Audit FormDocumento8 pagineInternal Quality Audit Formgharba putraNessuna valutazione finora
- Siphar Bod-03-06-2011Documento4 pagineSiphar Bod-03-06-2011Indra NeelNessuna valutazione finora
- Observation 1: Department of Health A!'Id Hu Ian ServicesDocumento3 pagineObservation 1: Department of Health A!'Id Hu Ian ServicesNevin SmithNessuna valutazione finora
- Revital Heathcare (Epz) Limited Quality Control Department Sterility Test Raw Data SheetDocumento2 pagineRevital Heathcare (Epz) Limited Quality Control Department Sterility Test Raw Data SheetMusyoka UrbanusNessuna valutazione finora
- Raya Pharmaceuticals Co. LTD: Cataflam®Documento8 pagineRaya Pharmaceuticals Co. LTD: Cataflam®Mayson BaliNessuna valutazione finora
- Journal of Controlled Release: Review ArticleDocumento26 pagineJournal of Controlled Release: Review ArticleUday BaruahNessuna valutazione finora
- Limit TestsDocumento20 pagineLimit TestsBMohdIshaqNessuna valutazione finora
- In-Process Testing of Infusions: Novamed Pharmaceutical Quality Control Department Standard Operating ProcedureDocumento5 pagineIn-Process Testing of Infusions: Novamed Pharmaceutical Quality Control Department Standard Operating Procedurenaeem186Nessuna valutazione finora
- Volumetric Measurement in The LaboratoryDocumento43 pagineVolumetric Measurement in The LaboratoryKiran ChokshiNessuna valutazione finora
- WI No. QC 003Documento2 pagineWI No. QC 003Khrystin BerdosNessuna valutazione finora
- Air Sampler: Quality Control DepartmentDocumento1 paginaAir Sampler: Quality Control DepartmentbilalmasNessuna valutazione finora
- Water Treatment System (Sistem Pengolahan Air Spa)Documento38 pagineWater Treatment System (Sistem Pengolahan Air Spa)Tjen MolynaNessuna valutazione finora
- Ag009 05 01Documento1 paginaAg009 05 01Nishit SuvaNessuna valutazione finora
- Cleaning and disinfection of food factories: a practical guideDa EverandCleaning and disinfection of food factories: a practical guideNessuna valutazione finora
- Manual Isocratic Pump - YL9200ADocumento33 pagineManual Isocratic Pump - YL9200ADavid SanabriaNessuna valutazione finora
- Adapaleno - Clindamicina Gel HPLCDocumento5 pagineAdapaleno - Clindamicina Gel HPLCDavid SanabriaNessuna valutazione finora
- N2 Generator ENG ManualDocumento48 pagineN2 Generator ENG ManualDavid SanabriaNessuna valutazione finora
- Intruction Manual ChromNav JascoDocumento128 pagineIntruction Manual ChromNav JascoDavid Sanabria100% (2)
- Azitromicina Metanol PDFDocumento5 pagineAzitromicina Metanol PDFDavid SanabriaNessuna valutazione finora
- Alginato Por IrDocumento3 pagineAlginato Por IrDavid SanabriaNessuna valutazione finora
- Determinación de NeomicinaDocumento1 paginaDeterminación de NeomicinaDavid Sanabria100% (1)
- Alginato HPLCDocumento7 pagineAlginato HPLCDavid SanabriaNessuna valutazione finora
- Efectos de Picolinato en La Fecundidad Del CerdoDocumento5 pagineEfectos de Picolinato en La Fecundidad Del CerdoDavid SanabriaNessuna valutazione finora
- HAMILTON Microlab 700 Flyer enDocumento15 pagineHAMILTON Microlab 700 Flyer enYannik OppligerNessuna valutazione finora
- Is 5403 1999Documento9 pagineIs 5403 1999akNessuna valutazione finora
- 4 - Lab Consumables Specification 20180604Documento5 pagine4 - Lab Consumables Specification 20180604martinNessuna valutazione finora
- Chem 11 - Lab 1aDocumento6 pagineChem 11 - Lab 1amisstsangNessuna valutazione finora
- EvaluationDocumento3 pagineEvaluationDennisNessuna valutazione finora
- E439-10 Standard Test Methods For Chemical Analysis of BerylliumDocumento8 pagineE439-10 Standard Test Methods For Chemical Analysis of Berylliumjavier perezNessuna valutazione finora
- TP1 Basic Concept For Determining The Concentration of The SolutionDocumento14 pagineTP1 Basic Concept For Determining The Concentration of The SolutionSor SoknaNessuna valutazione finora
- Specific Criteria For Calibration Laboratories in Mechanical DisciplineDocumento20 pagineSpecific Criteria For Calibration Laboratories in Mechanical DisciplineshahNessuna valutazione finora
- HACH Ammonia Salicylate Method No. 8155 DOC316.53.01077Documento6 pagineHACH Ammonia Salicylate Method No. 8155 DOC316.53.01077Balas43Nessuna valutazione finora
- Analysis of Grape and WineDocumento116 pagineAnalysis of Grape and WineElenaTrofimNessuna valutazione finora
- PV 3922 enDocumento3 paginePV 3922 enzerenchen666Nessuna valutazione finora
- HW Uncertainty in MeasurementDocumento5 pagineHW Uncertainty in MeasurementDaniel RoopchandNessuna valutazione finora
- CT 338Documento17 pagineCT 338Assane RoqueNessuna valutazione finora
- Chem ch3Documento20 pagineChem ch3ChandlerNessuna valutazione finora
- Phenol Test: SpectroquantDocumento1 paginaPhenol Test: Spectroquantrnd labNessuna valutazione finora
- Practical RevisionDocumento2 paginePractical RevisionJust HeyNessuna valutazione finora
- Multipette® Plus Repeater® Plus: Operating ManualDocumento18 pagineMultipette® Plus Repeater® Plus: Operating ManualJheraldyn CastroNessuna valutazione finora
- Commonly Encountered Equipment in The Chemistry LaboratoryDocumento11 pagineCommonly Encountered Equipment in The Chemistry LaboratoryAudrey MendozaNessuna valutazione finora
- A Microscale Acid-Base TitrationDocumento3 pagineA Microscale Acid-Base TitrationMark ReadNessuna valutazione finora
- Hydroxyl Value: Lubrizol Standard Test ProcedureDocumento4 pagineHydroxyl Value: Lubrizol Standard Test ProcedureAvadhut PatilNessuna valutazione finora
- AOAC 961.02 Spray Products As DesinfectantsDocumento5 pagineAOAC 961.02 Spray Products As DesinfectantsMario Rodrigo Ramirez MesaNessuna valutazione finora
- AllDocumento490 pagineAllStarla Ellison100% (1)
- Calibrating Pasteur PipetteDocumento4 pagineCalibrating Pasteur PipetteJanielle FajardoNessuna valutazione finora
- Eppendorf - Serological PipetsDocumento1 paginaEppendorf - Serological Pipetsenrique santillanNessuna valutazione finora
- Lab Manual - PHP-109 Pharma Analysis 1st Sem 10 ExpDocumento17 pagineLab Manual - PHP-109 Pharma Analysis 1st Sem 10 ExpAnkesh PalNessuna valutazione finora
- Kit Colina 2Documento4 pagineKit Colina 2Rodrigo Acuña LeivaNessuna valutazione finora
- ChemistryDocumento282 pagineChemistryTiger Alive100% (6)
- Raizada Lab Equipment InventoryDocumento8 pagineRaizada Lab Equipment Inventoryapi-256130935Nessuna valutazione finora
- SNI 2012 - in EnglishDocumento39 pagineSNI 2012 - in EnglishElwan Hafwan HadiwijayaNessuna valutazione finora
- CatálogoDocumento1.928 pagineCatálogoMaría José Cortez RamírezNessuna valutazione finora