Documenti di Didattica
Documenti di Professioni
Documenti di Cultura
17
Caricato da
shail_k_86Descrizione originale:
Titolo originale
Copyright
Formati disponibili
Condividi questo documento
Condividi o incorpora il documento
Hai trovato utile questo documento?
Questo contenuto è inappropriato?
Segnala questo documentoCopyright:
Formati disponibili
17
Caricato da
shail_k_86Copyright:
Formati disponibili
FOUNDER’S DAY
S P E C I A L I S S U E
A COMPARATIVE STUDY OF 20 MOLE % Gd2O3
DOPED CeO2 PREPARED BY DIFFERENT SYNTHETIC
ROUTES
R.V. Wandekar, B.N. Wani and S.R. Bharadwaj
Chemistry Division
Bhabha Atomic Research Centre
This paper was selected for the 2nd Best Oral Presentation Award
at the 15th National Symposium on Thermal Analysis
held in February 2006 at Jaipur
Abstract
In recent years, a growing interest in properties of nanostructured ion conductors,
has been observed. Solid electrolytes for Solid Oxide Fuel cell (SOFC) should be highly dense material
due to gas-tight requirement. It is noted that the sintering and densification behavior of ceria-based
electrolytes rely on synthetic route and temperature of calcinations. Hence, gadolinium doped
ceria samples were prepared, by the cation complexation technique which is a variant of sol-gel technique,
using citric acid as complexing agent and also by ceramic route. X-ray diffraction (XRD) and simultaneous
TG/DTA technique were used, to characterize the samples. The purpose of this work was to prepare
the powders with 10-20 nm of crystallite size at a relatively low temperature. A comparative study of their
particle size, densification, thermal expansion and ionic conductivity was carried out.
Introduction
Sm 3+ or Gd 3+ cation. These materials have been
High oxide-ion conducting solid electrolytes, based recognized as leading electrolyte materials [1-3] for
application in solid oxide fuel cell. Traditional ceramic
on zirconia, have been extensively investigated at higher
temperatures (~1000°C). In case of SOFC operating at method for fabricating dense electrolytes, involves
intermediate temperature (800°C), ceria based electrolyte extensive heating of oxide mixture at higher temperature,
materials are more suitable. Out of various dopants in to yield a single phase solid solution. The materials derived
ceria, samarium and gadolinium showed maximum ionic by wet chemical methods show better reactivity than
conductivity at ~800°C. Samarium and gadolinium those obtained by solid state reaction, but these materials
doped ( 20 mol % ) cerium oxide is a solid solution, also require typical densification temperature of ~1300°C
formed by replacing Ce4+ sites of the CeO2 lattice by to reach maximum theoretical density. Nanocrystalline
116 Issue no. 285 October 2007
FOUNDER’S DAY
S P E C I A L I S S U E
powders, that can be fully densified at a lower powder was estimated using Scherrer equation. Their
temperature (~1000°C), can be obtained via sol gel linear thermal expansion behavior was studied using
route. A sol-gel technique has been considered as an dilatometer. AC impedance of pellets sintered at 1400oC
alternative method, for the preparation of homogeneous were measured from 300 to 450°C in air using SI 1260
material with the molecular level mixing than those Impedance Analyzer (Solartron, UK) between 10 MHz
prepared by ceramic route. In this communication, we to 1 Hz. Platinum paste (MaTeck GmbH, Germany) was
report a simple citrate gel procedure to prepare nano- applied on the pellets as electrodes.
sized ceria-based electrolyte material at low temperature.
The citrate gel procedure offers a number of advantages Results and Discussion
like low cost, is relatively simple, generates less carbon
residue and is quite effective in producing highly TG/DTA curve of the precursor metal complex is shown
sinterable ceramic powders [4]. in Fig. 1. TG curve shows two overlapping steps of
weight loss: first step starts at 50°C and completes at
Experimental 200°C and the loss in second step continues up to 720°C.
The 2.03 % weight loss in first step is probably due to
Gadolinium doped ceria of composition Ce0.8Gd0.2 O2-d dehydration. During second step ~ 1.45 % continuous
(GDC) was synthesized, by sol gel and ceramic route. In weight loss is seen which can be due to decomposition
sol gel route, cerium nitrate and gadolinium nitrate was of precursor metal complex. The DTA curve exhibits an
mixed in the required stoichiometric ratio. Citric acid endothermic peak around 100°C and a large exothermic
was added to this mixture so as to maintain the total peak at ~ 320°C which extends up to 400°C.
metal to citric acid molar ratio of 1:2 and final mixture Endothermic peak can be assigned for dehydration of
was dissolved in a minimum amount of water. On heating adsorbed water from the precursor. Exothermic peak can
this solution mixture at 150°C a gel was formed after be ascribed to the burning of organic material in the
evaporation of water. Subsequently the gel was dried at
same temperature and the gases were
released to form a fine yellow color
precursor, which is a citric acid complex
of rare earth metal. The gel initially
started to swell and a foamy precursor
is formed. This foam contains very light
and homogeneous flacks of very small
particle size. Precursor was found to be
hygroscopic in nature. For comparison
these materials were also prepared by
standard ceramic route using CeO2 and
Gd2O3 as reactants and calcined at
900°C.
Powder XRD patterns of all the samples
were recorded (Phillips X-ray
Diffractometer, PW 1710, Ni filter and Fig. 1: TG/DTA scans of precursor
Cu Ka radiation and 2q range 10-70°).
The crystallite size of the calcined
Issue no. 285 October 2007 117
FOUNDER’S DAY
S P E C I A L I S S U E
sample and to the gradual crystallizing process of GDC. pattern corresponds to CeO2. The peaks are relatively
broader for the sol gel synthesized samples, indicating
In order to compare it with TG/DTA data, the X-ray that the powder is composed of smaller crystallites. The
diffraction patterns of GDC, heated at different broadening of the peaks gradually decreases with
temperatures during sol gel synthesis as well as by increasing temperature of pyrolysis.
ceramic route, were examined (Fig. 2). XRD data shows
no peaks due to impurity at 600°C and the observed The average crystallite size of GDC powder, calculated
by the Scherrer equation using d220
is given in Table 1. It clearly
indicates that sol gel synthesized
samples have smaller crystallite
size as compared to solid state
route sample.
The densities and the thermal
expansion coefficients of the
samples sintered at 1400°C are also
given in Table 1. It can be seen that
the density sol gel prepared sample
is marginally higher as compared
to that from the solid state
preparation. The thermal expansion
coefficients are, however,
comparable for the two samples
(Fig. 3).
Fig. 2: XRD patterns of GDC prepared by different
routes and calcined at different temperatures
Table 1 : Physical properties of GDC prepared by two different routes
SS = solid state SG = Sol gel route
118 Issue no. 285 October 2007
FOUNDER’S DAY
S P E C I A L I S S U E
Impedance spectra of GDC prepared by sol gel as
well as solid state route were recorded at different
temperatures in the frequency range of 10 MHz to
1 Hz. The electrical conductivity of GDC for grain
and grain boundary was obtained from impedance
spectra and the activation energy was calculated
from the linear Arrhenius plot of log (σ T) vs 1/T
(Fig. 4).
The conductivities of sol gel synthesized samples
are higher as compared to solid state synthesized
samples and the activation energies Ea, derived
from the log (σ T) vs 1/T plots are lower for sol-
Fig. 3: Dilatometry curve of GDC prepared by
sol gel ( ) and solid state ( • ) gel synthesized samples (Table 2).
Conclusion
Nanosized powders of gadolinium doped ceria were
prepared, by heating the metal complex precursor
using citric acid as complexing agent. Because of
the smaller particle size ~12- 30 nm, the
densification increases and the activation energy for
conduction reduces.
σT) vs 1/T for grain (gr)
Fig. 4. Plot of log (σ
and grain boundary (gb) of GDC prepared
by sol gel (sg) and solid state (ss).
Table 2: Energy of activation of GDC obtained from impedance studies
prepared by two different routes
Issue no. 285 October 2007 119
FOUNDER’S DAY
S P E C I A L I S S U E
References
1. M.Mogensen in Catalysis by Ceria and Related 3. Ji-Guang Li, T. Ikegami, Y. Wang and T. Mori, J.
Materials A.Trovarelli (Ed.) Imperial College Press Am. Ceram. Soc., 86 (2003) 915.
(2002) p 453. 4. R.A. Rocha and E.N.S. Muccillo, Mat. Res. Bull
2. C.Peng, Y.Wang, K. Jiang, B.Q.Bin, H.W.Liang, J. 38 (2003) 197.
Feng and J. Meng, J Alloys Compd., 349 (2003)
273.
About the Author
Ms. Rohini V. Wandekar, is pursuing her Ph.D in Chemistry at Bhabha Atomic
Research Centre (BARC) Mumbai. She obtained her M.Sc. degree in Chemistry from
University of Mumbai in 2003. Her research work involves the development of
various cell components in Solid Oxide Fuel Cell at intermediate temperature
(IT-SOFC) and study of thermodynamic properties of these materials
Dr (Ms) Bina N. Wani, Ph.D (Mumbai University) joined Chemistry Division of
BARC in 1979. She has about 28 years of experience in utilization of various thermo-
analytical techniques related to solid –state chemistry, studies on oxyfluoro
compound including superconducting materials and catalysts based on mixed
oxides. She has 49 publications in various reputed international journals to her
credit. She worked as Visiting Scientist at Ames Laboratory, Iowa, USA in 1995. Her
current interest lies in the development of a) various cell components in Solid
Oxide Fuel Cell at intermediate temperature (IT-SOFC) and b) mixed oxide based
catalyst for pollution control.
Dr (Ms) Shyamla Bharadwaj, M.Sc. and Ph.D. from Mumbai University joined
BARC in 1977 through 20th Batch Training School. She has been working in the
area of high temperature thermodynamics of nuclear as well as other refractory
materials for the past 30 years. During the year 2000-01, she worked as Guest
Scientist at Juelich Research Centre, Juelich, Germany. Presently, she is heading
the Fuel Cell Materials and Catalysis Section of Chemistry Division, BARC. She is a
Ph.D guide of Mumbai University and professor of Homi Bhabha National Institute,
Mumbai. She has been honoured with the NETSH-ITAS Award 2006, for her
contribution in the field of thermodynamics and thermal analysis.
120 Issue no. 285 October 2007
Potrebbero piacerti anche
- Shoe Dog: A Memoir by the Creator of NikeDa EverandShoe Dog: A Memoir by the Creator of NikeValutazione: 4.5 su 5 stelle4.5/5 (537)
- Crack Propagation of Rolling Contact Fatigue - 2007Documento12 pagineCrack Propagation of Rolling Contact Fatigue - 2007Geraldo Rossoni SisquiniNessuna valutazione finora
- The Yellow House: A Memoir (2019 National Book Award Winner)Da EverandThe Yellow House: A Memoir (2019 National Book Award Winner)Valutazione: 4 su 5 stelle4/5 (98)
- Solid State Devices Notes Pages 28-54Documento27 pagineSolid State Devices Notes Pages 28-54Mohamed Mahmoud Saad HassanNessuna valutazione finora
- The Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeDa EverandThe Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeValutazione: 4 su 5 stelle4/5 (5794)
- Performance of Reinforced Thermosetting Resin Pipe Systems in Desalination Applications: A Long-Term Solution To Corrosion - The Arabian Gulf ExampleDocumento6 paginePerformance of Reinforced Thermosetting Resin Pipe Systems in Desalination Applications: A Long-Term Solution To Corrosion - The Arabian Gulf ExamplejatecconsultNessuna valutazione finora
- High-Performance Direct Conversion X-Ray Detectors Based On Sintered Hybrid Lead Triiodide Perovskite WafersDocumento6 pagineHigh-Performance Direct Conversion X-Ray Detectors Based On Sintered Hybrid Lead Triiodide Perovskite WafersDiegoNessuna valutazione finora
- The Little Book of Hygge: Danish Secrets to Happy LivingDa EverandThe Little Book of Hygge: Danish Secrets to Happy LivingValutazione: 3.5 su 5 stelle3.5/5 (400)
- Design of Lacing Face To Face (Toe To Toe)Documento3 pagineDesign of Lacing Face To Face (Toe To Toe)kiranNessuna valutazione finora
- Grit: The Power of Passion and PerseveranceDa EverandGrit: The Power of Passion and PerseveranceValutazione: 4 su 5 stelle4/5 (588)
- Undrained Behaviour (And Modelling) : Computational Geotechnics CourseDocumento46 pagineUndrained Behaviour (And Modelling) : Computational Geotechnics Courselphuong_20Nessuna valutazione finora
- Elon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureDa EverandElon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureValutazione: 4.5 su 5 stelle4.5/5 (474)
- 1 s2.0 S2452321616302220 MainDocumento8 pagine1 s2.0 S2452321616302220 MainDaay VlixNessuna valutazione finora
- A Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryDa EverandA Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryValutazione: 3.5 su 5 stelle3.5/5 (231)
- 40nicrmo7-3: Steel GradeDocumento3 pagine40nicrmo7-3: Steel GradeGanesh K CNessuna valutazione finora
- Hidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceDa EverandHidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceValutazione: 4 su 5 stelle4/5 (895)
- Design properties of HEA profiles for S235 steel class (γ = 1.00, units = mm)Documento18 pagineDesign properties of HEA profiles for S235 steel class (γ = 1.00, units = mm)scarto08Nessuna valutazione finora
- Team of Rivals: The Political Genius of Abraham LincolnDa EverandTeam of Rivals: The Political Genius of Abraham LincolnValutazione: 4.5 su 5 stelle4.5/5 (234)
- Retrofitting of StructuresDocumento28 pagineRetrofitting of StructuresKapil DhabuNessuna valutazione finora
- Never Split the Difference: Negotiating As If Your Life Depended On ItDa EverandNever Split the Difference: Negotiating As If Your Life Depended On ItValutazione: 4.5 su 5 stelle4.5/5 (838)
- AR03 Studies On Failure Behaviour of Wire Rope Used in Underground Coal MinesDocumento21 pagineAR03 Studies On Failure Behaviour of Wire Rope Used in Underground Coal MinesmaddogoujeNessuna valutazione finora
- The Emperor of All Maladies: A Biography of CancerDa EverandThe Emperor of All Maladies: A Biography of CancerValutazione: 4.5 su 5 stelle4.5/5 (271)
- Base Plate DesignDocumento32 pagineBase Plate DesignGautam Sharma50% (2)
- Devil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaDa EverandDevil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaValutazione: 4.5 su 5 stelle4.5/5 (266)
- Effect of Temperature Loads On Beam Design in Thermal AnalysisDocumento4 pagineEffect of Temperature Loads On Beam Design in Thermal AnalysisMohammad AasimuddinNessuna valutazione finora
- On Fire: The (Burning) Case for a Green New DealDa EverandOn Fire: The (Burning) Case for a Green New DealValutazione: 4 su 5 stelle4/5 (74)
- Recent Development of High-Strength and Tough Welding Consumables For Offshore StructuresDocumento8 pagineRecent Development of High-Strength and Tough Welding Consumables For Offshore StructuresDiNessuna valutazione finora
- Fracture MechanicsDocumento17 pagineFracture Mechanicscal2_uniNessuna valutazione finora
- The Unwinding: An Inner History of the New AmericaDa EverandThe Unwinding: An Inner History of the New AmericaValutazione: 4 su 5 stelle4/5 (45)
- Arch Wires Used in OrthodonticsDocumento47 pagineArch Wires Used in Orthodonticsavanthika krishnaraj100% (1)
- Vtu Syllabus 02 PDFDocumento6 pagineVtu Syllabus 02 PDFAmarNessuna valutazione finora
- Homework No.3 - RC I - CEE 351 - Fall 2022Documento2 pagineHomework No.3 - RC I - CEE 351 - Fall 2022Elvis KarayigaNessuna valutazione finora
- The Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersDa EverandThe Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersValutazione: 4.5 su 5 stelle4.5/5 (345)
- D 4123-Indirect Tension Test For Resilient Modulus of BitumiDocumento4 pagineD 4123-Indirect Tension Test For Resilient Modulus of BitumianjanaNessuna valutazione finora
- Inelastic Bending of Singly-Reinforced Concrete Beam (Rectangular Section)Documento13 pagineInelastic Bending of Singly-Reinforced Concrete Beam (Rectangular Section)Joshua Callahan UangNessuna valutazione finora
- The World Is Flat 3.0: A Brief History of the Twenty-first CenturyDa EverandThe World Is Flat 3.0: A Brief History of the Twenty-first CenturyValutazione: 3.5 su 5 stelle3.5/5 (2259)
- GAP AnalysisDocumento5 pagineGAP AnalysisRosendo RizoNessuna valutazione finora
- Sherman 2002Documento66 pagineSherman 2002alden cayagaNessuna valutazione finora
- Process Equipment Design Materials Selection 7-02-13Documento91 pagineProcess Equipment Design Materials Selection 7-02-13May TampusNessuna valutazione finora
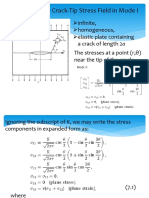
- Details of The Crack-Tip Stress Field in Mode IDocumento22 pagineDetails of The Crack-Tip Stress Field in Mode IFarid N SNessuna valutazione finora
- The Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreDa EverandThe Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreValutazione: 4 su 5 stelle4/5 (1090)
- What Happens To Load at YieldingDocumento14 pagineWhat Happens To Load at YieldingWaqas Anjum100% (2)
- GY360 Structural Geology: Lecture Notes On The Dynamic Analysis of StressDocumento9 pagineGY360 Structural Geology: Lecture Notes On The Dynamic Analysis of StressBLACKLIST 00Nessuna valutazione finora
- Class Notes For Making RugsDocumento2 pagineClass Notes For Making RugsdakNessuna valutazione finora
- (Shirvani 1998) Behavior of Tensile Anchors in Concrete Statistical Analysis and Design RecommendationsDocumento450 pagine(Shirvani 1998) Behavior of Tensile Anchors in Concrete Statistical Analysis and Design RecommendationsmanoelmangabeiraNessuna valutazione finora
- The Sympathizer: A Novel (Pulitzer Prize for Fiction)Da EverandThe Sympathizer: A Novel (Pulitzer Prize for Fiction)Valutazione: 4.5 su 5 stelle4.5/5 (121)
- Viscoelasticity 01 IntroDocumento4 pagineViscoelasticity 01 IntroJohn Mario CanroNessuna valutazione finora
- Jamaluddin 2019. Cooling Rapid MartensiteDocumento8 pagineJamaluddin 2019. Cooling Rapid MartensiteHabybur RahmanNessuna valutazione finora
- Her Body and Other Parties: StoriesDa EverandHer Body and Other Parties: StoriesValutazione: 4 su 5 stelle4/5 (821)