Documenti di Didattica
Documenti di Professioni
Documenti di Cultura
Ethy Acetate Production US20140012037A1
Caricato da
sariDescrizione originale:
Copyright
Formati disponibili
Condividi questo documento
Condividi o incorpora il documento
Hai trovato utile questo documento?
Questo contenuto è inappropriato?
Segnala questo documentoCopyright:
Formati disponibili
Ethy Acetate Production US20140012037A1
Caricato da
sariCopyright:
Formati disponibili
US 20140012037A1
(19) United States
(12) Patent Application Publication (10) Pub. No.: US 2014/0012037 A1
(43) Pub. Date:
Gadewar et al.
(54)
ETHYL ACETATE PRODUCTION
(71) Applicant: Greenyug, LLC, Santa Barbara, CA
(Us)
Jan. 9, 2014
Publication Classi?cation
(51)
(52)
Int. C1.
0070 67/58
us. c1.
(2006.01)
(72) Inventors: Sagar B. GadeWar, Goleta, CA (US);
CPC .................................... .. C07C67/58(2013.01)
Peter K. Stoimenov, Goleta, CA (U S)
USPC .......................... .. 560/239; 560/248; 422/187
(57)
(21) App1.No.: 14/023,125
(22) Filed:
Sep. 10, 2013
ABSTRACT
A method of purifying an ethyl acetate stream comprises
contacting an inlet stream With a solvent, transferring at least
a portion of the impurity compound from the inlet stream into
the solvent to form an extract and a puri?ed product, separat
Related US. Application Data
ing the extract from the puri?ed product, separating the por
tion of the impurity compound from the extract, forming an
impurities stream and a regenerated solvent, and recycling at
(63) Continuation-in-part of application No. 12/925,460,
least a portion of the regenerated solvent to contact the inlet
stream. The inlet stream comprises ethyl acetate and an impu
?led on Oct. 20, 2010, noW Pat. No. 8,558,025.
(60)
Provisional application No. 61/253,349, ?led on Oct.
rity compound, and the extract comprises the solvent and the
portion of the impurity compound transferred from the inlet
20, 2009.
stream.
Patent Application Publication
Jan. 9, 2014 Sheet 1 0f 17
ETHANOL.
0.4
US 2014/0012037 A1
HYDROGEN
I!
a m
ETHYL ACE-TATE
V g
mi
1.0
ACETALDEHYDE
Figure 1: Reactive residue curve maps at pressures of1 atm and 5 atm.
Patent Application Publication
Jan. 9, 2014 Sheet 2 0f 17
ETHANOL
US 2014/0012037 A1
HYDROGEN
0.8
H15
0.4
EH3 _
0.4 P = 10 atm
as '
1n
I18
1mm. ACETATE
ACETALDEH Y'DE
ETHANOL
HYDROGEN
1713
"'AVN
""VJH I
x"
if:
0.3
P = 20 atm
(I, 6
a_ if
0.4
1):;
.s
IE:
I;
El,
DB
0.0
as '
ACETALDEH YDE
Figure 2: Reactive residue curve maps at pressures of 10 atm and 20 atm.
Patent Application Publication
Jan. 9, 2014 Sheet 3 0f 17
US 2014/0012037 A1
16
19
18
13
10
12
17
14
15
20
>
Figure 3: Single feed reactive distillation column schematic.
22
Patent Application Publication
Jan. 9, 2014 Sheet 4 0f 17
US 2014/0012037 A1
34
32
36
Figure 4: Schematic for a reactive distillation system with subsequent product
hydrogenation.
Patent Application Publication
Jan. 9, 2014 Sheet 5 0f 17
US 2014/0012037 A1
54
59
5O
56
46
42
4o
44
48
52
~
I
V
58
Figure 5: Double feed reactive distillation column schematic with an upper feed of
ethanol and lower feed of hydrogen.
Patent Application Publication
Jan. 9, 2014 Sheet 6 0f 17
US 2014/0012037 A1
80
9O
92
Figure 6: Double feed reactive distillation column schematic using dual catalyst
beds with an upper feed of ethanol and lower feed of hydrogen.
Patent Application Publication
Jan. 9, 2014 Sheet 7 0f 17
US 2014/0012037 A1
56
(b)
Figure 7: Side reactor configuration (a) upward flow feed to side reactor (b)
downward flow feed to side reactor.
Patent Application Publication
Jan. 9, 2014 Sheet 8 0f 17
US 2014/0012037 A1
84
86
VA I 106
70
(b)
107
108
82
:2
110
109
90
92
Figure 8: Double Side reactor configuration (a) upward flow feed to multiple side
reactors (b) downward flow feed to multiple side reactors.
Patent Application Publication
Jan. 9, 2014 Sheet 9 0f 17
US 2014/0012037 A1
216
DC
1-1
212\
FIG.9
206f
208
202
Patent Application Publication
Jan. 9, 2014 Sheet 10 0f 17
UZEmH
US 2014/0012037 A1
Patent Application Publication
Jan. 9, 2014 Sheet 11 0f 17
Em
a
\Sm
8m
Mam//
US 2014/0012037 A1
A|
DEN$60
A|4\|Sm8m a/
oLmwcz?aE2iwmbx
\
Em\\
0
ln
m
8N
amEm3:51
l
2
m
29 %
.595%
/Sm
102
m3
A962
Em286 2E8<
Patent Application Publication
Jan. 9, 2014 Sheet 12 0f 17
US 2014/0012037 A1
364,\
A362
360
PSf1Fctriho2egmapu.rsicneg
358
3\56
307
256
Patent Application Publication
Jan. 9, 2014 Sheet 14 0f 17
UQES 269
US 2014/0012037 A1
EN
2.oLcz6m5:Eb-_23c9ox.0wm
v?
@320 628i
f
2m
Ewzom
Patent Application Publication
Jan. 9, 2014 Sheet 15 0f 17
US 2014/0012037 A1
66
w
=
5&8
(I)
(d
aof7Nd0mH1Se8UditQ
?gure
Patent Application Publication
Jan. 9, 2014 Sheet 16 0f 17
13mm uogaexmawm wauemqwz
US 2014/0012037 A1
Patent Application Publication
Jan. 9, 2014 Sheet 17 0f 17
US 2014/0012037 A1
Jan. 9, 2014
US 2014/0012037 A1
ETHYL ACETATE PRODUCTION
CROSS-REFERENCE TO RELATED
APPLICATIONS
[0001] This application is a continuation-in-part of and
claims priority to US. patent application Ser. No. 12/925,460
?led on Oct. 20, 2010 to Sagar B. GadeWar and entitled Ethyl
Acetate Production, Which is a Non-Provisional application
of and claims priority to US. Provisional Application No.
61/253,349, ?led Oct. 20, 2009 to Sagar B. GadeWar and
entitled Ethyl Acetate Production, both of Which are incor
porated herein by reference in their entirety.
STATEMENT REGARDING FEDERALLY
SPONSORED RESEARCH OR DEVELOPMENT
[0002] Not applicable.
REFERENCE TO A MICROFICHE APPENDIX
[0003] Not applicable.
BACKGROUND
[0004]
The following discussion is provided solely to assist
the understanding of the reader, and does not constitute an
admission that any of the information discussed or references
cited constitute prior art to the present invention.
[0005] Ethyl acetate can be produced from acetaldehyde
according to the Tischenko reaction:
With a co-precipitate composition comprising silver-cad
mium-Zinc-Zirconium Which is substantially in the free metal
form.
[0013] Use of the Tischenko reaction for the production of
mixed esters from aldehydes is described in US. Pat. No.
3,714,23 6.
[0014] US. Pat. No. 5,334,751 describes production of
ethyl acetate by reaction of ethanol and oxygen in the pres
ence of a solid catalyst that contains crystalline TiP2O7 and
has the formula PdaMbTiPcOx, Where M is Cd, Au, Zn, T1, or
an alkali metal or alkaline earth metal, a is 0.0005-0.2, b is
0.3a, c is 0.5-2.5, x has a value to satisfy the valencies, and Ti
and P of the crystalline TiP2O7 represent part of the crystal
line TiP2O7.
[0015] BR-A-91/04652 describes pre-treatment of a palla
dium on a silica carrier catalyst for production of ethyl acetate
by direct oxidation of ethanol With air.
[0016]
Production of esters from primary alcohols by dehy
drogenation using bromous acid or a salt thereof in acid
medium is described in JP-A-59/025334.
[0017] In SU-A-362814 there is described a process for
production of ethyl acetate by dehydrogenation of ethanol at
180 C. to 300 C. in the presence of a copper catalyst con
taining Zinc as an activator With an ethanol feed rate of 250 to
700 liters per liter of catalyst per hour.
[0018]
The dehydrogenation of ethanol to form ethyl
acetate is described in GB-A-287846. This proposes use of a
dehydrogenating agent, such as a copper catalyst, a tempera
ture of from 250 C. to 500 C., and a pressure of more than
10 atmospheres (1 .013><106 Pa).
[0006] When acetaldehyde is produced from ethanol, ethyl
acetate can be produced from ethanol according to the fol
[0019] Vapor phase contact of ethanol at a temperature
above its critical temperature With a catalyst comprising cop
loWing reaction:
per and a dif?cultly reducible oxide, such as Zinc oxide or
[0007] Altemately, ethanol can react With acetaldehyde
according to the folloWing reaction:
Mpa) being suggested.
manganese oxide, is proposed in GB-A-312345, use of a
temperature of 375 C. and a pressure of 4000 psi (27.58
[0020] GB-A-470773 teaches a process for conversion of
ethanol to ethyl acetate by dehydrogenating ethanol over a
catalyst consisting of a reduced metal, for example, copper on
[0008] Conversion of primary alcohols into esters and
ketones using copper catalyst is described by K. Takeshita in
the Bulletin of the Chemical Society of Japan, (1978) vol.
51(9), pp 2622-2627.
[0009] US. Pat. No. 4,996,007 describes a process for the
oxidation of primary alcohols to aldehydes, esters and acids.
A primary alcohol is reacted With oxygen, With a catalyst
selected from ruthenium, rhodium, platinum, palladium, rhe
nium and mixtures thereof, With the option of quaternary C1
to C20 alkyl ammonium co-catalyst, and dihydrodihydrox
ynaphthalene, dihydroxyanthracene or a mixture thereof as
an oxygen activator.
[0010] In US. Pat. No. 4,220,803 catalytic dehydrogena
tion of ethanol for the production of acetaldehyde and acetic
acidusing a supported copper oxide essentially free of barium
is proposed.
[0011] US. Pat. No. 4,052,424 suggesteda silver-cadmium
alloy catalyst for use in production of alkyl alkanoate esters,
by contacting a primary alkanol in the vapor phase With the
catalyst at a temperature of betWeen about 250 C. and 6000
C.
[0012] In US. Pat. No. 4,440,946 there is described a pro
cess for producing a carboxylate ester Which comprises con
tacting a mixture of alcohol and aldehyde in the vapor phase
infusorial earth With 10% uranium oxide as promoter, main
tained at a temperature of 220 C. to 260 C., removing by
condensation some of the gas-vapor product rich in hydrogen
resulting from the reaction, and returning the gaseous remain
der rich in hydrogen to the catalyZing Zone.
[0021] EP-A-0151886 describes a process for the prepara
tion of C2+ esters of alkyl carboxylic acids from C2+ primary
alcohols Which comprises contacting a vaporous mixture
containing a primary C2+ alkanol and hydrogen in an alkanol:
hydrogen molar ratio of from 1:10 to about 1000:1 at a com
bined partial pres sure of alkanol and hydrogen of from about
0.1 bar (103 Pa) up to about 40 bar (4><106 Pa) and at a
temperature in the range of from about 180 C. to about 300
C. in a catalytic reaction Zone With a catalyst consisting
essentially of a reduced mixture of copper oxide and Zinc
oxide, and recovering a reaction product mixture containing a
primary C2+ alkyl ester of an alkyl carboxylic acid Which
ester contains tWice as many carbon atoms as the primary C2+
alkanol.
[0022] In EP-A-0201105 there is described a method for
converting primary alcohols, such as ethanol, to their corre
sponding alkanoate esters Which involves the regulation of
the mole feed ratio of hydrogen gas to alkanol in the reaction
Zone of a copper-chromite containing catalyst.
Jan. 9, 2014
US 2014/0012037 A1
[0023]
One method of separating ethyl acetate from ethanol
and Water involves extractive distillation With an extractive
agent comprising polyethylene glycol and dipropylene gly
col, diethylene glycol, or triethylene glycol as described in
US. Pat. No. 4,569,726 or With an extractive agent containing
dimethyl sulphoxide as described in US. Pat. No. 4,379,028.
[0024] Separation of ethyl acetate from a composition com
prising ethyl acetate, ethanol and Water is described in JP-A
05/186392 by feeding the composition to a distillation col
umn to obtain a quasi-aZeotropic mixture comprising ethyl
acetate, ethanol and Water, condensing it, separating the con
densate into an organic layer and an aqueous layer, returning
the organic layer to the column, and recovering ethyl acetate
as a bottom product from the column.
[0025]
EP-A-0331021 describes hoW carbonylation of ole
?ns to produce monocarboxylate esters causes formation of
aldehydes and acetals as byproducts. Monocarboxylate esters
produced in this Way are subjected to a three step puri?cation
process involving treatment With a strongly acidic agent, fol
loWed by hydrogenation and distillation. The initial treatment
With a strongly acidic agent is intended to convert acetals to
vinyl ethers and aldehydes and acetals to aldols. The subse
drogenation catalyst, an ethanol inlet con?gured to pass an
ethanol feed over the dehydrogenation catalyst, a top product
gaseous hydrogen removal passage, and a bottoms product
liquid ethyl acetate removal passage. The extraction unit is
con?gured to receive a liquid ethyl acetate product stream
from the reactive distillation column through the bottoms
product liquid ethyl acetate removal passage, contact a liquid
solvent feed stream With the liquid ethyl acetate product
stream, provide an extract stream comprising a portion of any
impurities in the liquid ethyl acetate product stream, and
provide a puri?ed product stream. The stripping unit is con
?gured to receive the extract stream from the extraction unit,
separate the portion of the impurities from the extract stream,
provide an outlet impurities stream, and provide a regener
ated solvent stream back to the extraction unit as at least a
portion of the liquid solvent feed stream.
[0030]
In an embodiment, a reactive distillation process
producing high purity ethyl acetate from ethanol comprises
feeding a feed stream comprising ethanol to a reactive distil
lation column, contacting the ethanol With a catalyst, dehy
drogenating ethanol over the catalyst in the liquid phase dur
ing the distillation process, removing ethyl acetate during the
quent hydrogenation step then converts these compounds to
distillation process as a bottoms product, and removing
byproducts Which are more easily separated from the desired
hydrogen during the distillation process as a top product.
[0031] These and other features Will be more clearly under
stood from the folloWing detailed description taken in con
monocarboxylate ester.
[0026]
EP-A-0101910 contains a similar disclosure regard
ing carbonylation of ole?ns to give monocarboxylate esters. It
junction With the accompanying draWings and claims.
proposes treatment of the monocarboxylate ester With hydro
gen at elevated temperature in the presence of an acidic ion
exchanger or Zeolite doped With one or more metals of Group
VIII of the Periodic Table, folloWed by hydrogenation. It is
stated that acetals present as byproducts are converted to vinyl
ethers Which are converted by hydrogenation to loW boiling
esters or the aldehydes and acetals are converted to high
boilers by an aldol reaction. Unsaturated ketones are con
verted to saturated ketones.
[0027]
US. Pat. No. 4,435,595 describes the use ofreactive
distillation to produce high-purity methyl acetate, by esteri
fying methanol and acetic acid. The process provides for the
use of acetic acid and methanol Which How in counter-current
through a single reactive distillation column in the presence
of an acid catalyst. The acetic acid, in addition to being a
reagent, also acts as an extracting agent for the non-reacted
methanol and for the Water Which has been produced. There
BRIEF DESCRIPTION OF THE DRAWINGS
[0032] For a more complete understanding of the present
disclosure and the advantages thereof, reference is noW made
to the folloWing brief description, taken in connection With
the accompanying draWings and detailed description.
[0033] FIG. 1 shoWs reactive residue maps for conversion
of ethanol to ethyl acetate at pressures of 1 and 5 atmospheres
(atm) for an embodiment.
[0034] FIG. 2 shoWs reactive residue maps for conversion
of ethanol to ethyl acetate at pressures of 10 and 20 atm for an
embodiment.
[0035] FIG. 3 shoWs a simpli?ed schematic of a reactive
distillation system according to an embodiment.
[0036] FIG. 4 shoWs a simpli?ed schematic of a reactive
distillation system according to another embodiment.
fore, the methyl acetate is separated continuously from the
[0037]
acetic acid and removed from the top of the column.
distillation system according to still another embodiment.
[0038]
SUMMARY
[0028]
In an embodiment, a method of purifying an ethyl
acetate stream comprises contacting an inlet stream With a
solvent, transferring at least a portion of the impurity com
pound from the inlet stream into the solvent to form an extract
and a puri?ed product, separating the extract from the puri?ed
product, separating the portion of the impurity compound
from the extract, forming an impurities stream and a regen
erated solvent, and recycling at least a portion of the regen
erated solvent to contact the inlet stream. The inlet stream
comprises ethyl acetate and an impurity compound, and the
extract comprises the solvent and the portion of the impurity
compound transferred from the inlet stream.
[0029]
In an embodiment, a reactive distillation system for
FIG. 5 shoWs a simpli?ed schematic of a reactive
FIG. 6 shoWs a simpli?ed schematic of a reactive
distillation system according to yet another embodiment.
[0039] FIGS. 7(a) and 7(b) shoWs a simpli?ed schematic of
a reactive distillation system according to an embodiment.
[0040]
FIGS. 8(a) and 8(1)) shoWs a simpli?ed schematic of
a reactive distillation system according to another embodi
ment.
[0041] FIG. 9 illustrates a schematic ?oW diagram of a
reactive distillation system With a recycle according to an
embodiment.
[0042] FIG. 10 illustrates a schematic ?oW diagram of a
product separation system according to an embodiment.
[0043]
FIG. 11 illustrates a schematic ?oW diagram of a
product separation system according to another embodiment.
[0044]
FIG. 12 illustrates a schematic ?oW diagram of a
producing high purity ethyl acetate from ethanol comprises a
stripping section according to another embodiment.
reactive distillation column, an extraction unit, and a strip
[0045] FIG. 13 illustrates a schematic co-current extraction
?oW scheme according to an embodiment.
ping unit. The reactive distillation column comprises a dehy
Potrebbero piacerti anche
- Industrial Process Plant Construction Estimating and Man-Hour AnalysisDa EverandIndustrial Process Plant Construction Estimating and Man-Hour AnalysisValutazione: 5 su 5 stelle5/5 (1)
- Chemical Resistance GuideDocumento40 pagineChemical Resistance GuidepablopinNessuna valutazione finora
- Combustion Engines: An Introduction to Their Design, Performance, and SelectionDa EverandCombustion Engines: An Introduction to Their Design, Performance, and SelectionNessuna valutazione finora
- Process Steam Systems: A Practical Guide for Operators, Maintainers, and DesignersDa EverandProcess Steam Systems: A Practical Guide for Operators, Maintainers, and DesignersNessuna valutazione finora
- MS Protokol Nenad RosoDocumento2 pagineMS Protokol Nenad RosoМилош ЈовановићNessuna valutazione finora
- Turton AppBDocumento114 pagineTurton AppBAdesuwa O'sae0% (1)
- Synthetic Natural Gas: From Coal, Dry Biomass, and Power-to-Gas ApplicationsDa EverandSynthetic Natural Gas: From Coal, Dry Biomass, and Power-to-Gas ApplicationsTilman J. SchildhauerNessuna valutazione finora
- Well Integrity for Workovers and RecompletionsDa EverandWell Integrity for Workovers and RecompletionsValutazione: 5 su 5 stelle5/5 (3)
- Biochemistry PDFDocumento431 pagineBiochemistry PDFMichael PiducaNessuna valutazione finora
- The Steam Engine Explained and Illustrated (Seventh Edition) With an Account of its Invention and Progressive Improvement, and its Application to Navigation and Railways; Including also a Memoir of WattDa EverandThe Steam Engine Explained and Illustrated (Seventh Edition) With an Account of its Invention and Progressive Improvement, and its Application to Navigation and Railways; Including also a Memoir of WattNessuna valutazione finora
- Practical Synthetic Organic Chemistry: Reactions, Principles, and TechniquesDa EverandPractical Synthetic Organic Chemistry: Reactions, Principles, and TechniquesNessuna valutazione finora
- Case Studies in Mechanical Engineering: Decision Making, Thermodynamics, Fluid Mechanics and Heat TransferDa EverandCase Studies in Mechanical Engineering: Decision Making, Thermodynamics, Fluid Mechanics and Heat TransferValutazione: 5 su 5 stelle5/5 (2)
- Sustainable Energy Conversion for Electricity and Coproducts: Principles, Technologies, and EquipmentDa EverandSustainable Energy Conversion for Electricity and Coproducts: Principles, Technologies, and EquipmentNessuna valutazione finora
- Process For Ethyl Acetate ProductionDocumento9 pagineProcess For Ethyl Acetate ProductionMuhammad Yanuar AnantaNessuna valutazione finora
- Hydrogen Production TechnologiesDa EverandHydrogen Production TechnologiesMehmet SankirNessuna valutazione finora
- Combustion of Pulverised Coal in a Mixture of Oxygen and Recycled Flue GasDa EverandCombustion of Pulverised Coal in a Mixture of Oxygen and Recycled Flue GasNessuna valutazione finora
- Us 20120035390Documento18 pagineUs 20120035390sariNessuna valutazione finora
- Electrochemical FertilizerDocumento24 pagineElectrochemical Fertilizerpakde jongkoNessuna valutazione finora
- United States: (12) Patent Application Publication (10) Pub. No.: US 2015/0018589 A1Documento7 pagineUnited States: (12) Patent Application Publication (10) Pub. No.: US 2015/0018589 A1Lily DianaNessuna valutazione finora
- Cooper. Process For The Production of Acetic Anhydride and Acetic Acid - European Patent Office - EP 0087870 B1Documento9 pagineCooper. Process For The Production of Acetic Anhydride and Acetic Acid - European Patent Office - EP 0087870 B1John Patrick DagleNessuna valutazione finora
- Acetaldehido A01 031Documento4 pagineAcetaldehido A01 031DwiPurwatiNessuna valutazione finora
- United States: (12) Patent Application Publication (10) Pub. No.: US 2010/0197959 A1Documento19 pagineUnited States: (12) Patent Application Publication (10) Pub. No.: US 2010/0197959 A1sariNessuna valutazione finora
- EP2066611B1Documento1 paginaEP2066611B1Baruc Martinez SantiagoNessuna valutazione finora
- United States Patent (19) 11 Patent Number: 6,093,845: Van Acker Et Al. (45) Date of Patent: Jul. 25, 2000Documento8 pagineUnited States Patent (19) 11 Patent Number: 6,093,845: Van Acker Et Al. (45) Date of Patent: Jul. 25, 2000falya aronaNessuna valutazione finora
- Us 20080089986Documento30 pagineUs 20080089986MohamadDanielBinYaacobNessuna valutazione finora
- IPR2018-00159 PTAB Permits SK Innovation To Swap Unpatentable Claim 2 With New Claim in Its Molecular Weight Method PatentDocumento44 pagineIPR2018-00159 PTAB Permits SK Innovation To Swap Unpatentable Claim 2 With New Claim in Its Molecular Weight Method PatentTim BillickNessuna valutazione finora
- Patent Asam Asetat 2015Documento22 paginePatent Asam Asetat 2015Nyimas Ulfatry100% (1)
- Production of Sodium DithioniteDocumento10 pagineProduction of Sodium DithioniteDhaval PadaliaNessuna valutazione finora
- Acido PropionicoDocumento6 pagineAcido Propionicoromina orellanaNessuna valutazione finora
- Acido Propionico ImpurezasDocumento14 pagineAcido Propionico Impurezasromina orellanaNessuna valutazione finora
- Ulllted States Patent (10) Patent N0.: US 8,193,396 B2Documento6 pagineUlllted States Patent (10) Patent N0.: US 8,193,396 B2Adelina Boru SianturiNessuna valutazione finora
- Pendukung 1LIU ET ALL 2018 US20180201568A1Documento8 paginePendukung 1LIU ET ALL 2018 US20180201568A1nysa arientikaNessuna valutazione finora
- Turton AppBDocumento114 pagineTurton AppBamms9988Nessuna valutazione finora
- Ulllted States Patent (19) (11) Patent Number: 6,013,597: Karim Et Al. (45) Date of Patent: Jan. 11, 2000Documento10 pagineUlllted States Patent (19) (11) Patent Number: 6,013,597: Karim Et Al. (45) Date of Patent: Jan. 11, 2000shcnmartinNessuna valutazione finora
- Us 20120090534Documento28 pagineUs 20120090534Victor Von DoomNessuna valutazione finora
- Catalyst (DMC Type)Documento3 pagineCatalyst (DMC Type)Visarut RujirawanichNessuna valutazione finora
- United States: (12) Patent Application Publication (10) Pub. No.: US 2013/0015405 A1Documento27 pagineUnited States: (12) Patent Application Publication (10) Pub. No.: US 2013/0015405 A1edwinNessuna valutazione finora
- TEPZZ 65 46ZA - T: European Patent ApplicationDocumento24 pagineTEPZZ 65 46ZA - T: European Patent ApplicationIrfan LutfiantoNessuna valutazione finora
- Ulllted States Patent (19) (11) Patent Number: 6,114,278: Karim Et Al. (45) Date 0f Patent: Sep. 5, 2000Documento8 pagineUlllted States Patent (19) (11) Patent Number: 6,114,278: Karim Et Al. (45) Date 0f Patent: Sep. 5, 2000federico paNessuna valutazione finora
- US8269048Documento6 pagineUS8269048Lara MartinezNessuna valutazione finora
- Asetaldehid (Recovered)Documento15 pagineAsetaldehid (Recovered)Mario VensfiseCrewNessuna valutazione finora
- FGFHGHJHJKDocumento9 pagineFGFHGHJHJKMary Grace VelitarioNessuna valutazione finora
- TOLUENEDocumento7 pagineTOLUENEAlzahra AliNessuna valutazione finora
- Patent EsDocumento8 paginePatent EsWalter RaulNessuna valutazione finora
- United States: (12) Patent Application Publication (10) Pub. No.: US 2009/0005588 A1Documento10 pagineUnited States: (12) Patent Application Publication (10) Pub. No.: US 2009/0005588 A1sariNessuna valutazione finora
- Gicc Us8631660Documento9 pagineGicc Us8631660Ricardo RamsesNessuna valutazione finora
- Integrated Process For The Production of Vinyl Acetate From Acetic Acid Via Ethyl AcetateDocumento44 pagineIntegrated Process For The Production of Vinyl Acetate From Acetic Acid Via Ethyl Acetatearif ihwandaNessuna valutazione finora
- EP2952536A2Documento45 pagineEP2952536A2Yahya AlmundzirNessuna valutazione finora
- Iii Iihiiii: United States Patent (19) 11 Patent Number: 5,780,656Documento5 pagineIii Iihiiii: United States Patent (19) 11 Patent Number: 5,780,656Mari Luz Hermoza MamaniNessuna valutazione finora
- United States Patent: 4,837,360 Patent Number: Date of Patent: EtDocumento6 pagineUnited States Patent: 4,837,360 Patent Number: Date of Patent: EtorogenidaNessuna valutazione finora
- United States Patent: Bartlett Patent No.: Date of PatentDocumento34 pagineUnited States Patent: Bartlett Patent No.: Date of PatentSameh El-SabbaghNessuna valutazione finora
- Japan Sea WaterDocumento8 pagineJapan Sea WaterVikk SuriyaNessuna valutazione finora
- Advances in Solid Oxide Fuel Cells XDa EverandAdvances in Solid Oxide Fuel Cells XMihails KusnezoffNessuna valutazione finora
- TEPZZ 4 8 - ZZA - T: European Patent ApplicationDocumento23 pagineTEPZZ 4 8 - ZZA - T: European Patent ApplicationMaru LinNessuna valutazione finora
- United States Patent (191: Z2 20) Woter - I 18JDocumento7 pagineUnited States Patent (191: Z2 20) Woter - I 18JZulfi ZumalaNessuna valutazione finora
- United States Patent (10) Patent No.: US 8,143,340 B2Documento5 pagineUnited States Patent (10) Patent No.: US 8,143,340 B2Awais KhanNessuna valutazione finora
- Light Monoolefins From Methanol And/or Dimethyl Ether: BonifazDocumento12 pagineLight Monoolefins From Methanol And/or Dimethyl Ether: BonifazSp4rkZ87Nessuna valutazione finora
- US8349170 - Components of Riser and Reactor SectionDocumento25 pagineUS8349170 - Components of Riser and Reactor SectionPraveen KumarNessuna valutazione finora
- Ceramic Materials for Energy Applications IV: A Collection of Papers Presented at the 38th International Conference on Advanced Ceramics and Composites, January 27-31, 2014, Daytona Beach, FLDa EverandCeramic Materials for Energy Applications IV: A Collection of Papers Presented at the 38th International Conference on Advanced Ceramics and Composites, January 27-31, 2014, Daytona Beach, FLHua-Tay LinNessuna valutazione finora
- Fuels, Chemicals and Materials from the Oceans and Aquatic SourcesDa EverandFuels, Chemicals and Materials from the Oceans and Aquatic SourcesFrancesca M. KertonNessuna valutazione finora
- GROUP 3 Qualitative Test For LipidsDocumento2 pagineGROUP 3 Qualitative Test For LipidsJennifer SorianoNessuna valutazione finora
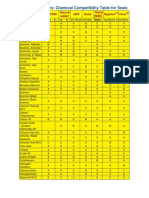
- Chemical Compatibility Table For SealsDocumento3 pagineChemical Compatibility Table For SealsThanh Tuyên VõNessuna valutazione finora
- IUPAC Provisional Recommendations: Table VII Ligand AbbreviationsDocumento12 pagineIUPAC Provisional Recommendations: Table VII Ligand AbbreviationsSalih PaşaNessuna valutazione finora
- PCD Pharma CompanyDocumento14 paginePCD Pharma Companyprince jainNessuna valutazione finora
- CHE232 - Course Outline 2022Documento4 pagineCHE232 - Course Outline 2022MayibongweNessuna valutazione finora
- United States Patent (10) Patent No.:: Huynh-Ba (45) Date of Patent: Jan. 13, 2004Documento11 pagineUnited States Patent (10) Patent No.:: Huynh-Ba (45) Date of Patent: Jan. 13, 2004Alexander Franco CastrillonNessuna valutazione finora
- HM 11Documento51 pagineHM 11body fayezNessuna valutazione finora
- Lec 8 Protein QualityDocumento6 pagineLec 8 Protein QualityABISAGANA B CS&BS studentNessuna valutazione finora
- Worksheet Modul 7Documento15 pagineWorksheet Modul 7Naufal FasaNessuna valutazione finora
- Lab 4 - Lipid AnalysisDocumento7 pagineLab 4 - Lipid Analysisapi-383698554Nessuna valutazione finora
- Alkena Dan Reaksi AdisiDocumento22 pagineAlkena Dan Reaksi AdisiSalsha meifitra agnaNessuna valutazione finora
- Haloalkanes and Haloarenes-Anil-hssliveDocumento3 pagineHaloalkanes and Haloarenes-Anil-hssliveNikhil MathewNessuna valutazione finora
- Pre TestDocumento7 paginePre TestRONALD ARTILLERONessuna valutazione finora
- Novabiochem: Derivatives For Enhancing Peptide SynthesisDocumento4 pagineNovabiochem: Derivatives For Enhancing Peptide SynthesisValentina D BrunaNessuna valutazione finora
- Organic Derivatives of WaterDocumento6 pagineOrganic Derivatives of WaterCelyn Ann RamosNessuna valutazione finora
- Class 12 Org Che CHAPTER 10Documento10 pagineClass 12 Org Che CHAPTER 10DeekshaNessuna valutazione finora
- Nama ObatDocumento3 pagineNama ObatrestiNessuna valutazione finora
- Biokimia Sistem Endokrin Metabolik 2017Documento48 pagineBiokimia Sistem Endokrin Metabolik 2017putuNessuna valutazione finora
- Dosis Obat Igd PDFDocumento3 pagineDosis Obat Igd PDFRiyadila FajarizaNessuna valutazione finora
- Lipids and Fatty Acids-Pages-1-12Documento12 pagineLipids and Fatty Acids-Pages-1-12Cristel Mae De GuzmanNessuna valutazione finora
- Solanum Alkaloids and Their Pharmaceutical Roles - A ReviewDocumento14 pagineSolanum Alkaloids and Their Pharmaceutical Roles - A ReviewHuy BakNessuna valutazione finora
- Plastics VocabularyDocumento2 paginePlastics VocabularyLautaro FalchiniNessuna valutazione finora
- Tabel Antoine 1Documento3 pagineTabel Antoine 1atikaindrnNessuna valutazione finora
- ,, ,, - 3, - Omega-Q AriixDocumento3 pagine,, ,, - 3, - Omega-Q AriixOlga VoicuNessuna valutazione finora
- Second Self-Assessment TestDocumento2 pagineSecond Self-Assessment TestAbhay VishwakarmaNessuna valutazione finora
- งานเคมีบ าบัด กลุ่มงานเภสัชกรรม โรงพยาบาลราชวิถี ตาราง การเตรียมยา และความคงตัวของยาเคมีบ าบัด Reconstitution PreparationDocumento11 pagineงานเคมีบ าบัด กลุ่มงานเภสัชกรรม โรงพยาบาลราชวิถี ตาราง การเตรียมยา และความคงตัวของยาเคมีบ าบัด Reconstitution Preparationtotoil22Nessuna valutazione finora
- Assignment-1-Cbse Question Bank Chapter-12-Aldehydes, Ketones & Carboxylic AcidsDocumento9 pagineAssignment-1-Cbse Question Bank Chapter-12-Aldehydes, Ketones & Carboxylic AcidsSHUBHAMNessuna valutazione finora