Documenti di Didattica
Documenti di Professioni
Documenti di Cultura
Solubility of Chlorine in Brine
Caricato da
Sandro GoisisDescrizione originale:
Copyright
Formati disponibili
Condividi questo documento
Condividi o incorpora il documento
Hai trovato utile questo documento?
Questo contenuto è inappropriato?
Segnala questo documentoCopyright:
Formati disponibili
Solubility of Chlorine in Brine
Caricato da
Sandro GoisisCopyright:
Formati disponibili
356
Bott & Schulz: Solubility of Chlorine in Brine at Duerent p H Values
SOLUBILITY OF CHLORINE IN A BRINE SOLUTION
AT DIFFERENT pH VALUES
By T. R. BOTT and S. SCHULZ
The solubilities of chlorine in a solution containing 250 g/litre of sodium chloride, at different pH values (in
the range 1-2.5) have been experimentally determined at different chlorine partial pressures and at temperatures of 50" and 75". The results show some slight divergence from data previously reported in the
literature but these were obtained by a different procedure.
Introduction
Absorption of chlorine in water involves not only physical
solution, C12(as), but also chemical reaction. The reaction
which takes place is:
CI2 (as) 2H20
HOCl H 3 0 + C1-. ...........(1)
The 'active chlorine', or the chlorine present in solution due to
absorption, C12 (act), is therefore made up of a proportion
each of Clz (aq), HOCl and C1-.
If, however, the solvent is not pure water but water containing a quantity of dissolved sodium chloride, the conditions become more complex since the common ion gains
importance. Several reactions are considered to take place
in addition to the physical absorption of chlorine (as with
pure water) i.e.:Cl5+2H50
+ HOCl+H,O++Cl- ...............(2)
..........(3)
HOCl+H20 k H 3 0 + +CIO- ..................... (4)
Clz + CI- % (213- ....................................... ( 5 )
3C12 9H20
% 6H,O++
5Clk-t CIOs-..
The different reactions suggest that there is a strong dependence of chlorine solubility on hydrogen ion concentration, since the equilibrium of the various reactions will be
affected by pH.
Similarly, sodium chloride being a strong electrolyte is
virtually completely dissociated producing C1- ions in solution which in turn at least affects the equilibrium position
of some of the reactions. The solubility of chlorine in brine
will be dependent upon partial pressure of chlorine, salt
concentration and p H in the salt solution and the temperature. Barr' has made theoretical investigations of this problem.
Solubility data of chlorine in brine without reference to
pH are given in the l i t e r a t ~ r e . ~A- ~paper by Yokota5 gives
data which makes allowance for hydrogen ion concentration.
Data on the solubility of chlorine in salt solution are important for the efficient operation of electrolytic cells producing chlorine, and in the design, and operation, of absorption towers for the recovery of chlorine with brine.
Experimental
A dynamic method for the determination of the chlorine
solubility was adopted, since it had the advantage of steady
conditions. The stirring action of bubbling the gas through
the liquid in the absorption cell avoided the possibility of
supersaturation.
Fig. I shows the apparatus in diagrammatic form. In
order to be able to vary the partial pressure of the chlorine
in the apparatus, nitrogen could be supplied from a pressurised
supply and mixed with gaseous chlorine from a liquified
supply. Before mixing, the nitrogen and chlorine could be
metered separately.
After mixing, the combined gas stream passed through a
heater and a series of water saturators maintained at constant
temperature in a bath. The temperature of this well-stirred
bath was maintained at a fixed temperature below the absorption cell temperature (to within +0*15"), so that the water
vapour content of the gas stream entering the absorption
-&N2
'I
1. Needle valve
5. Wash-bottle
9. 3-way tap
13. Manometer
Fig. 1. Apparatus for
2. Flow meter
6. Water reservoir
10. Side vessel
14. Trap for condensed water
the determination of chlorine solubility
3. Safety device
4. Heating coil
7. Heating jacket
8 . Heating coil
11. Tap
12. Absorption vessel
16. Water reservoir for adjusting water level in 15
15. Seal pot
J. appl. Chem., 1967, Vol. 172December
Bott h Schulz: Solubility of Chlorine in Brine at Different p H Values
cell was the same as that leaving the cell. If these precautions
had not been taken it would have meant that the solution in
the cell would have gained or lost water, according to conditions, and this would have led to erroneous results. The
partial pressure of the water vapour in the gas stream leaving
the test cell was quite considerable at the higher temperatures. Allowance was made for this water vapour content in
the subsequent calculation of the chlorine partial pressure.
The saturated gas mixture from the saturators was passed
immediately through heaters maintained at the second bath
temperature so that condensation was avoided, and then into
the cell where it bubbled through a sintered glass diffuser
into the salt solution. The bath was well stirred and maintained at a temperature within the limits of t0.05".At the
exit from the cell, a trap was provided to collect any water
condensed out due to the lower temperature and to prevent
interference with steady gas flow conditions-condensation
at this stage was not important provided the condensate did
not run back into the cell. Finally the gas stream was
allowed to bubble through a level device (Fig. 1, 15). The
height of the bottle (Fig. 1, 16) was adjusted to allow for
changes in atmospheric pressure, thereby maintaining a
constant pressure in the apparatus.
The cell was providcd with means of measuring the pH
in situ. The side vessel (Fig. 1, 10) was equipped with a
conical joint at the top which was designed to take the glass
electrode system of the pH meter when in use. The pressure
in the absorption cell (Fig. 1 , 12) was sufficiently high to
push the liquid into the side vessel with the tap (Fig. 1, 11)
open and the tap (Fig. 1,9) connected to atmosphere. The pH
measurement could be carried out while the temperature of
the brine was the same as for saturation and while the composition of the gas in contact with the solution in the cell.
Equilibrium conditions therefore were not disturbed. Closing
the valve (Fig. 1, 9) allowed the liquid in the side vessel to be
pushed back into the cell after measurement of pH. The
hydrogen ion concentration determination did not interfere
with the course of the experiment and could be made
periodically during the 'run'. A Marconi pM nicter with
miniature electrodes was used for the pH measurements.
Care was exercised according to the maker's instructions and
particularly as far as keeping the sensitive calomel electrodc
free from temperature fluctuations. Standardisation was
carried out outside the cell, and the electrodes only inserted
into the side vessel when experiments were being conducted.
The absorption cell was fitted with a rubber serum cap at
the top so that samples of brine could be taken by hypodermic syringe without leakage of chlorine or change in cell
pressure. As the ncedle was made to reach the bottom of the
brine cell it was possible to take samples while an experiment
was in progress, and working conditions were not affected.
The chlorinated brine rose into the syringe by virtue of the
pressure in the cell which was allowed to push the plunger
back slowly. After the needle was withdrawn, it was sealed
by pushing it into a rubber bung. The solubility of the
chlorine falls with temperature rise, and therefore as the
solution cooled there was no tendency to lose chlorine.
Saniples (usually three) were taken periodically and
analysed. When consecutive samples showed similar chlorine
contents it was considered that equilibrium had been established. The 'active chlorine' (i.e. Clz (a@, HOCl and C13-)
content of the chlorinated brine was determined by titration,
the basis of the method being the oxidation of arsenite to
arsenate by dissolved CIz and HOCI-as the reaction pro-
J. appl. Chem., 1967, VsI. 17, December
357
ceeds the equilibria of Equations 2-5 will be disturbed so that
eventually all the dissolved chlorine will react. A few
drops of phenolphthalein indicator were added to 25 ml
0.1 N-sodium arsenite solution, followed by a small quantity
of 2 N-sodium hydroxide so that thc solution remained
alkaline when the cooled 25 ml sample of chlorinated brine
from the test cell was added. 0.5 N-Sulphuric acid was
carefully added until thc colour of the phenolphthalein was
discharged, followed by 2-3 g of sodium bicarbonate and
starch indicator. The excess sodium arsenite was backtitrated with 0.1 N-iodine solution.
The test brine solutions were made up from distilled water
and 'Analar' sodium chloride. The initial pH was adjusted
by adding 0.01 N-HCI solution to the brine.
Results and discussion
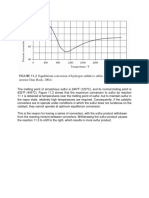
Data on pH and chlorine solubility have been obtained for
brine of concentration 250 g/1 at SO" and different chlorine
partial pressures. At 75", results at one partial pressure only
are recorded. The results are shown in Figs. 2 and 3.
x-fl -I
2mc3
-x-x
I
0
I
2
PH
Fig. 2. Solubility of chlorine in brine (250 g / l ) at 5 0 " ~
Chlorine partial pressure: Curve 1 0'93 atm.; curve 2 0.5 atm.;
curve 3 0.1 atm.
ii
1001 0
20
1'5
25
PH
Fig. 3. Solubility of chlorine in brine (250 g / l ) at 15"c and a chlorine
partial pressure of 0-67atm.
358 Blake & Randle: Removal of Zn2+from the Ternary System Zn2+-Na+-H+ by Cation-exchange Resin Column
=l
E 1600
I
0
k5 1200
/
,
PH
Fig. 4. Sulubiliiy of chlorine in brine (250 g / l ) at 50C arid a chloriiie
partial pressure of 0.93 atm.
Bott & Schulz
- _ _ _ _ YokotaS
Over the range studied, the solubility of the chlorine decreases slightly with increasing pH up to values 1-1-5 but
thereafter increases with increasing pH. The solubility decreases with chlorine partial pressure in the gas mixture as
would be expected.
Yokota5 presented a nomograph based on his experimental
work for the prediction of the amount of Clz(as) and CISpresent in solution at different temperatures, partial pressures
and salt concentration. The effect of the pH of the brine
solution on solubility, however, is taken into account by the
addition of a third term allowing for the amount of HOCl
present. Correction factors are given for a chlorine partial
pressure of one atmosphere only. The curves given in Fig. 4
compare the results of the present work (data at a chlorine
partial pressure of 0-93 atmospheres and a temperature of
50) and data taken from Y ~ k o t a .The
~ two sets of results
show slight divergence over the range of pH studied. The
difference is only approximately 5 % at the greatest deviation.
The measurement of pH in situ, as in this work, is considered
to give a more reliable result than an inferred value.
Department of Chemical Engineering,
University of Birmingham,
Edgbaston, Birmingham, 15
Received 3 April, 1967;
amended manuscript 13 July, 1967
References
Barr, L., J. electro. chen?. Soc., 1954, 101, 497
Seidell, A., Solubilities of Inorganic and Metal Organic Compounds, Vol. 1, 1940, 3rd edn, p. 397 (New York: Van
Nostrand)
Ullmann, F, Encyklopadie der Technischen Chemie, Vol. 5,
1954, p. 291 (Munchen-Berlin: Urban & Schwarzenberg)
Washburn, E. W., ed., International Critical Tables, Vol. 3,
1928, 1st edn, p. 256 (New York: McGraw-Hill)
Yokota, N., Chem. Engng, Tokyo, 1958,22,476
REMOVAL OF Zn FROM THE TERNARY SYSTEM
Zn -Na+ -H+ BY CATION-EXCHANGE RESIN COLUMN
By W. E. BLAKE and J. RANDLE
Very dilute solutions containing zinc sulphate, sodium and sulphuric acid were passed down a column of
Zeo-Karb 225 cation-exchange resin, and the volume at which zinc first appeared in the effluent (breakthrough volume), together with the volume at which the concentration of zinc in the effluent became equal
to its concentration in the influent (equivalent volume), were determined.
For solution concentrations of: Na2S04, 0.1-0*9 %; ZnSO,, 0.07-0.3 %; H,S04, 0.1-0.3 %, the
following equations applied:
log (breakthrough volume) = - 0.124 (log% Na,SO,) - 1.57 (log % HZS04)- 0.987 (log% ZnSO,) 0.780(k 0.045)
and
log (equilibrium volume) = -0,139 (log% Na2S04) -0-987 (log% H,S04) -0.663 (log% h S 0 4 ) f
0.482( f0.021)
Nomograms were constructed relating breakthrough volume and equilibrium volume to solution concentration.
J. appl. Chem., 1967, Vol. 17, December
Potrebbero piacerti anche
- The Secrets of Breakpoint ChlorinationDocumento26 pagineThe Secrets of Breakpoint Chlorinationmailmaverick8167Nessuna valutazione finora
- ORP Vs FRCDocumento12 pagineORP Vs FRCShesharam Chouhan100% (1)
- Solvent Extraction of Palladium From Chloride Media by TBPDocumento6 pagineSolvent Extraction of Palladium From Chloride Media by TBPMoreno MarcatiNessuna valutazione finora
- Equilibria in The Ammonium Carbamate Urea-Water System: K. G. Clark, L. C. DDocumento5 pagineEquilibria in The Ammonium Carbamate Urea-Water System: K. G. Clark, L. C. DjcortezcNessuna valutazione finora
- Absorption of CO2 in Aqueous DiglycolamineDocumento10 pagineAbsorption of CO2 in Aqueous DiglycolamineLê Vinh HảoNessuna valutazione finora
- Oxidation Reduction Potential (Orp) :redox and Free ChlorineDocumento2 pagineOxidation Reduction Potential (Orp) :redox and Free ChlorinePraneeth SrivanthNessuna valutazione finora
- Understanding Oxidising Biocides and ORP ControlDocumento6 pagineUnderstanding Oxidising Biocides and ORP Controljfisher2534100% (1)
- HCL ScrubberDocumento2 pagineHCL Scrubbersundhar100% (2)
- Colloids - 15-11Documento51 pagineColloids - 15-11Vo Trung Kien B2100780Nessuna valutazione finora
- h2s Naoh Equilibrium CurveDocumento22 pagineh2s Naoh Equilibrium Curvesmartleo_waloNessuna valutazione finora
- Preparation of Catalysts II: Scientific Bases for the Preparation of Heterogeneous CatalystsDa EverandPreparation of Catalysts II: Scientific Bases for the Preparation of Heterogeneous CatalystsNessuna valutazione finora
- Desulfurization of Natural Gas LiquidsDocumento21 pagineDesulfurization of Natural Gas LiquidsZoran ČeralinacNessuna valutazione finora
- Stripping Notes PDFDocumento28 pagineStripping Notes PDFnimi khalidNessuna valutazione finora
- Case Study 109 Ammonia OxidationDocumento9 pagineCase Study 109 Ammonia OxidationkaalkankNessuna valutazione finora
- Hydrochloric Acid Plant Design: The Copperbelt University School of Technology Chemical Engineering DepartmentDocumento86 pagineHydrochloric Acid Plant Design: The Copperbelt University School of Technology Chemical Engineering DepartmentAndre Winata100% (1)
- Chlorine: International Thermodynamic Tables of the Fluid StateDa EverandChlorine: International Thermodynamic Tables of the Fluid StateNessuna valutazione finora
- Group 3Documento31 pagineGroup 3vdfbhgNessuna valutazione finora
- Day 4 - Iron Control PDFDocumento37 pagineDay 4 - Iron Control PDFMohanned Khairy100% (1)
- Sulfuric Acid - Wikipedia PDFDocumento94 pagineSulfuric Acid - Wikipedia PDFrajesh indukuriNessuna valutazione finora
- Pool Equations PHDocumento84 paginePool Equations PHMariusCapraNessuna valutazione finora
- Vapor-Liquid Equilibria. Ethylene Oxide - Acetaldehyde and Ethylene Oxide - Water SystemsDocumento5 pagineVapor-Liquid Equilibria. Ethylene Oxide - Acetaldehyde and Ethylene Oxide - Water SystemsGie0% (1)
- Room Temperature Synthesis of Copper Oxide Nanoparticles Morphological Evaluation and Their Catalytic Applications For Degradation of Dyes and C-N Bond Formation Reaction PDFDocumento11 pagineRoom Temperature Synthesis of Copper Oxide Nanoparticles Morphological Evaluation and Their Catalytic Applications For Degradation of Dyes and C-N Bond Formation Reaction PDFAshpavi ArunNessuna valutazione finora
- Claus ProcessDocumento5 pagineClaus ProcesssatishchemengNessuna valutazione finora
- ConclusionDocumento2 pagineConclusionfrhslmn100% (1)
- Part 1: Design, Modeling and Simulation of Post-Combustion CO Capture Systems Using Reactive SolventsDocumento24 paginePart 1: Design, Modeling and Simulation of Post-Combustion CO Capture Systems Using Reactive SolventsBenjamin F ZavalaNessuna valutazione finora
- Packed Columns: Design and Performance: Further ReadingDocumento18 paginePacked Columns: Design and Performance: Further ReadingSergei JvjvjgNessuna valutazione finora
- CO2 SoluBilityDocumento8 pagineCO2 SoluBilityAntonio José Luque CarmonaNessuna valutazione finora
- Density and Viscosity of BrineDocumento33 pagineDensity and Viscosity of BrineJerrydawangNessuna valutazione finora
- Cooling Towers Information PackageDocumento25 pagineCooling Towers Information Packagemdawg467Nessuna valutazione finora
- CO2 Capture With ILs and DESsDocumento33 pagineCO2 Capture With ILs and DESsMasroorAbroNessuna valutazione finora
- Flow Instabilities in A Horizontal Thermosyphon Reboiler LoopDocumento22 pagineFlow Instabilities in A Horizontal Thermosyphon Reboiler Loopwylie01Nessuna valutazione finora
- Assignment 2 With SolutionsDocumento4 pagineAssignment 2 With SolutionsVenkat MacharlaNessuna valutazione finora
- Steam To Carbon Ratio Calculations: InstructionsDocumento8 pagineSteam To Carbon Ratio Calculations: InstructionsSuresh Ramakrishnan100% (1)
- Phthalic AnhydrideDocumento1 paginaPhthalic AnhydrideShashikant PanchalNessuna valutazione finora
- Chapter 3 Separation of Oil and Gas - 1987 - Developments in Petroleum ScienceDocumento50 pagineChapter 3 Separation of Oil and Gas - 1987 - Developments in Petroleum ScienceFalokid RaboNessuna valutazione finora
- Chee3004: Project 2Documento43 pagineChee3004: Project 2faizzi100% (3)
- Understanding and Eliminating Pressure Fluctuations in 2010 Process SafetyDocumento6 pagineUnderstanding and Eliminating Pressure Fluctuations in 2010 Process SafetyandriNessuna valutazione finora
- High-Pressure Fluid Phase Equilibria: Phenomenology and ComputationDa EverandHigh-Pressure Fluid Phase Equilibria: Phenomenology and ComputationNessuna valutazione finora
- Desulfurization v1.2 (Final)Documento35 pagineDesulfurization v1.2 (Final)Talha JamilNessuna valutazione finora
- Ammonium Carbamte P, T - BASFDocumento4 pagineAmmonium Carbamte P, T - BASFVinh Do ThanhNessuna valutazione finora
- Koya University Faculty of Engineering School of Petroleum and Chemical Reservior LabDocumento16 pagineKoya University Faculty of Engineering School of Petroleum and Chemical Reservior LabBakomora Evans WilliamsNessuna valutazione finora
- Ammonia Production PDFDocumento5 pagineAmmonia Production PDFJustine LagonoyNessuna valutazione finora
- Producción Del Ácido SulfúricoDocumento102 pagineProducción Del Ácido SulfúricoLuis Diego Cardenas PadillaNessuna valutazione finora
- AdsoptionDocumento17 pagineAdsoptionArun Sharma100% (1)
- Plant Design Kojic Acid Beta 1Documento41 paginePlant Design Kojic Acid Beta 1Milton Dels RosaNessuna valutazione finora
- Water Index CalculationsDocumento24 pagineWater Index CalculationsGustavo Adolfo Piñero Borges100% (1)
- Lecture 1 - Sulfur and Sulfuric AcidDocumento9 pagineLecture 1 - Sulfur and Sulfuric AcidNayan HalderNessuna valutazione finora
- Production of Pearl Caustic Soda-653343 PDFDocumento62 pagineProduction of Pearl Caustic Soda-653343 PDFhardajhbfNessuna valutazione finora
- Bubble Column ReactorDocumento21 pagineBubble Column ReactorMuhammad Hamzah SyahrirNessuna valutazione finora
- Ozone PDFDocumento4 pagineOzone PDFShesharam ChouhanNessuna valutazione finora
- Caustic Soda Industry-1Documento3 pagineCaustic Soda Industry-1Muhammad KhurramNessuna valutazione finora
- Cyanide Treatment TechnologiesDocumento3 pagineCyanide Treatment Technologiesdei_sandeep7994Nessuna valutazione finora
- Solubility Ammonium Sulfate in Sulfuric Acid SolutionDocumento2 pagineSolubility Ammonium Sulfate in Sulfuric Acid SolutionBagus Surya Saputra0% (1)
- Guidelines For Sulfide Generation PDFDocumento24 pagineGuidelines For Sulfide Generation PDFnawajhaNessuna valutazione finora
- Packed-Bed Microreactor For Continuous-FlowDocumento9 paginePacked-Bed Microreactor For Continuous-Flowkarpanai100% (1)
- Liquid-Phase Chlorination of Ethylene and 1,2-DichloroethaneDocumento7 pagineLiquid-Phase Chlorination of Ethylene and 1,2-DichloroethaneAntonio AraujoNessuna valutazione finora
- TEP03 Part4 AbsorptionDocumento28 pagineTEP03 Part4 AbsorptionEK63Nessuna valutazione finora
- OverviewDocumento21 pagineOverviewgreenhen15Nessuna valutazione finora
- Formic Acid Plant: A Brief OverviewDocumento17 pagineFormic Acid Plant: A Brief OverviewMuzzamilNessuna valutazione finora
- 1 - SO2 and NOx PDFDocumento13 pagine1 - SO2 and NOx PDFAaron David ReidNessuna valutazione finora
- Pump Handbook by GrundfossDocumento156 paginePump Handbook by GrundfossSyam Tawakkal100% (1)
- Pressure Drops Two PhasesDocumento13 paginePressure Drops Two PhasesSandro Goisis100% (1)
- Brochure - On-Site Nitrogen MembraneDocumento2 pagineBrochure - On-Site Nitrogen MembraneSandro GoisisNessuna valutazione finora
- Curve Fitting of Ammonia-Water Mixture PropertiesDocumento18 pagineCurve Fitting of Ammonia-Water Mixture PropertiesSandro GoisisNessuna valutazione finora
- Chem Eng May 2014Documento84 pagineChem Eng May 2014Sandro GoisisNessuna valutazione finora
- Building Blocks of Life Unit Book Guide: Chapter 1: Reviewing Introductory BiologyDocumento6 pagineBuilding Blocks of Life Unit Book Guide: Chapter 1: Reviewing Introductory Biologyapi-521781723Nessuna valutazione finora
- Handbook of Metal EtchantDocumento3 pagineHandbook of Metal EtchantLilian RoseNessuna valutazione finora
- The Cook Book of Chemistry: Rajat Kalia - Alpha ClassesDocumento299 pagineThe Cook Book of Chemistry: Rajat Kalia - Alpha ClassesRajat KaliaNessuna valutazione finora
- Agnique PG 8105-G: Technical InformationDocumento4 pagineAgnique PG 8105-G: Technical InformationADITYA MAHANessuna valutazione finora
- RPT Science F2 KSSM 2020Documento20 pagineRPT Science F2 KSSM 2020intanNessuna valutazione finora
- Differential EquationsDocumento19 pagineDifferential EquationsDinah Jane MartinezNessuna valutazione finora
- Estimation of PHENOLSDocumento2 pagineEstimation of PHENOLSGajaNand Mallick100% (1)
- 6 Chem PackDocumento5 pagine6 Chem PackCody YangNessuna valutazione finora
- Vapor Pressure LoweringDocumento10 pagineVapor Pressure LoweringMelvin CabonegroNessuna valutazione finora
- Direct Quantification of Sulfur Dioxide in Wine by Surface Enhanced RamanSpectroscopy - Mandrile2020Documento7 pagineDirect Quantification of Sulfur Dioxide in Wine by Surface Enhanced RamanSpectroscopy - Mandrile2020Kateřina InachisNessuna valutazione finora
- 5070 s12 QP 11Documento16 pagine5070 s12 QP 11Saad ArsalanNessuna valutazione finora
- CHM 123L Lab 1 Due September 9, 2013 Gravimetric Analysis of Phosphorus in Plant FoodDocumento6 pagineCHM 123L Lab 1 Due September 9, 2013 Gravimetric Analysis of Phosphorus in Plant FoodAhmed IsmailNessuna valutazione finora
- PDFDocumento39 paginePDFEdna Lip AnerNessuna valutazione finora
- 15.1 Energy CyclesDocumento28 pagine15.1 Energy CyclesNico Theodorus SimamoraNessuna valutazione finora
- Nucleation and Growth Kinetics of Biochemicals Measured at High SupersaturationsDocumento10 pagineNucleation and Growth Kinetics of Biochemicals Measured at High SupersaturationsDuong TuNessuna valutazione finora
- StoichiometryDocumento60 pagineStoichiometryneerajtrip123Nessuna valutazione finora
- MC 2 Lab Prelim 2Documento4 pagineMC 2 Lab Prelim 2Diana Joy Ancheta CldheiNessuna valutazione finora
- ASTM G205-16 Standard Guide For Determining Emulsion Properties, Wetting Behavior, and Corrosion-Inhibitory Properties of Crude Oils PDFDocumento10 pagineASTM G205-16 Standard Guide For Determining Emulsion Properties, Wetting Behavior, and Corrosion-Inhibitory Properties of Crude Oils PDFSergio DiazNessuna valutazione finora
- Matter: Learning Competencies Code Date UnpackedDocumento4 pagineMatter: Learning Competencies Code Date UnpackedJUDITH M. MABAO100% (1)
- Unitech Bituboard DataDocumento2 pagineUnitech Bituboard DataSantosh Patil100% (1)
- 1.4 Material and Energy BalanceDocumento51 pagine1.4 Material and Energy Balancesmith100% (1)
- Metallic Bonding: Theodore L. Brown H. Eugene Lemay, Jr. and Bruce E. BurstenDocumento12 pagineMetallic Bonding: Theodore L. Brown H. Eugene Lemay, Jr. and Bruce E. BurstenAngelica Maeriz MindoroNessuna valutazione finora
- Biochemistry BOOKDocumento82 pagineBiochemistry BOOKShimmering MoonNessuna valutazione finora
- 2 Physico-Chemical Principles of Steelmaking Processes: Heterogeneous Vs Homogeneous SystemsDocumento11 pagine2 Physico-Chemical Principles of Steelmaking Processes: Heterogeneous Vs Homogeneous SystemsMir RafsanNessuna valutazione finora
- Exp 6 Phase Equilibrium Eutectic SystemDocumento6 pagineExp 6 Phase Equilibrium Eutectic SystemNur Fadhilah33% (3)
- Top 22 Chemistry Interview Questions & AnswersDocumento7 pagineTop 22 Chemistry Interview Questions & AnswersSreedevi KrishnakumarNessuna valutazione finora
- 460 Bai Tap Dien HoaDocumento69 pagine460 Bai Tap Dien Hoavanhiepk52a100% (1)
- PHC340Y Lab ManualDocumento237 paginePHC340Y Lab ManualAdrika Chowdhary50% (2)
- Inter 1 Chemistry Success Series 2019 by Ambitious PDFDocumento17 pagineInter 1 Chemistry Success Series 2019 by Ambitious PDFArslan Sattar100% (2)
- Crospovidone PDFDocumento2 pagineCrospovidone PDFstselvaNessuna valutazione finora