Documenti di Didattica
Documenti di Professioni
Documenti di Cultura
Thermodynamics
Caricato da
sarathCopyright
Formati disponibili
Condividi questo documento
Condividi o incorpora il documento
Hai trovato utile questo documento?
Questo contenuto è inappropriato?
Segnala questo documentoCopyright:
Formati disponibili
Thermodynamics
Caricato da
sarathCopyright:
Formati disponibili
S.
13
Thermodynamics (November-2012, Set-2) JNTU-Kakinada
Code No.: R21032/R10
II B.Tech. I Semester Regular Examinations
November - 2012
Set-2
Solutions
THERMODYNAMICS
( Common to ME, AE, AME, MM )
Time: 3 Hours
Max. Marks: 75
Answer any FIVE Questions
All Questions carry equal marks
---
1.
2.
(a)
Establish the first law of thermodynamics from the Joules experiment. (Unit-II, Topic No. 2.4)
(b)
Briefly explain the macroscopic and microscopic view points. (Unit-I, Topic No. 1.3)
(c)
The energy in Joule (J) of a closed system can be expressed as E = 100 + 50 T + 0.04 T2. The heat absorbed is
given by Q (in Joule) = 5000 + 20 T. Temperature is in K. Calculate the work done during a process when the
temperature rises from 500 K to 1000 K. (Unit-I, Topic No. 1.4)
(a)
State and prove Carnots theorem. (Unit-III, Topic No. 3.3)
(b)
A house is to be maintained at a temperature of 20C by means of a heat pump pumping heat from the
atmosphere. Heat losses through the walls of the house are estimated at 0.65 kW per unit of temperature
difference between the inside of house and atmosphere.
(i)
If the atmospheric temperature is 10C, what is the minimum power required to drive the pump?
(ii) It is proposed to use the same heat pump to cool the house in the summer. For the same room temperature,
the same heat loss rate, and the same power input to the pump, what is the maximum permissible atmospheric
temperature? (Unit-II, Topic No. 2.5)
3.
Calculate the changes of entropy per kg of air in the following cases,
(i)
Air expands isothermally from 6 bar to 3 bar
(ii)
Air is compressed to half the volume at constant pressure and
(iii) Heat is supplied to air at constant volume till the pressure becomes three fold.
What would be the change in entropy of the air undergoes the above three processes in sequence. Take Cp = 1.005
kJ/kgK and Cv = 0.720 kJ/kgK. (Unit-III, Topic No. 3.4)
4.
5.
(a)
Using the Maxwells relations, obtain the expression for the difference of specific heats at constant pressure
and constant volume in terms of the derivatives of properties. Hence show that CP CV is always positive.
(Unit-V, Topic No. 5.3)
(b)
With the help of a schematic diagram, explain the measure of steam quality using the separating and throttling
calorimeter. (Unit-IV, Topic No. 4.5)
(a)
Explain the law of corresponding states. Hence briefly explain the significance of compressibility chart.
(Unit-V, Topic No. 5.3)
(b)
Two vessels A and B, each of volume 3 m3 may be connected together by a tube of negligible volume. Vessel
A contains air at 7 bar, 95C and vessel B contains air at 3.5 bar, 205C. Find the change of entropy when A is
connected to B. Assume the mixing to be complete and adiabatic. (Unit-VI, Topic No. 6.1)
( JNTU-Kakinada )
B.Tech. II-Year I-Sem.
Spectrum ALL-IN-ONE Journal for Engineering Students, 2013
S.14
6.
(a)
Define the following properties,
(i)
Humidity ratio
(ii) Dew point temperature and
(iii) Relative humidity (Unit-VI, Topic No. 6.2.1)
7.
(b)
Cooling water enters the cooling tower at a rate of 1000 kg/hr and 70C. Water is pumped from the base of the
tower at 24C and some makeup water is added afterwards. Air enters the tower at 15C, 50% RH, 1.01325 bar
and leaves the tower saturated at 34C, 1 bar. Calculate the flow rate of dry air in kg/hr and the makeup water
required per hour. (Unit-VIII, Topic No. 8.3)
(a)
An engine working on Otto cycle has an air standard efficiency of 56% and rejects 544 kJ/kg of air. The pressure
and temperature of air at the beginning of compression are 0.1 MPa and 60C respectively. Compute,
(i)
The compression ratio of the engine and
(ii) Maximum pressure in the cycle. (Unit-VII, Topic No. 7.1.1)
(b)
8.
Explain the simple Rankine cycle with the help of a schematic and T-S diagram. (Unit-VIII, Topic No. 8.1)
Write short notes on the following,
(a)
Clausius inequality (Unit-III, Topic No. 3.4)
(b)
Clausius - Clapeyron equation (Unit-IV, Topic No. 4.3)
(c)
Equivalence of Clausius and Kelvin-Plank statements of second law of thermodynamics. (Unit-III, Topic No. 3.2)
B.Tech. II-Year I-Sem.
( JNTU-Kakinada )
S.15
Thermodynamics (November-2012, Set-2) JNTU-Kakinada
SOLUTIONS TO NOVEMBER-2012, SET-2, QP
Q1.
(a)
Establish the first law of thermodynamics from the Joules experiment.
Nov.-12, Set-2, Q1(a)
Answer :
For answer refer Unit-II, Q10.
(b)
Briefly explain the macroscopic and microscopic view points.
Nov.-12, Set-2, Q1(b)
Answer :
For answer refer Unit-I, Q9.
(c)
The energy in Joule (J) of a closed system can be expressed as E = 100 + 50 T + 0.04 T2. The
heat absorbed is given by Q (in Joule) = 5000 + 20 T. Temperature is in K. Calculate the work
done during a process when the temperature rises from 500 K to 1000 K.
Nov.-12, Set-2, Q1(c)
Answer :
Given that,
T1 = 500 k
T2 = 1000 k
E = 100 + 50 T + 0.04 T2
Q = 5000 + 20 T
Workdone during the process is given by,
W = Q E
= Q E
W = (5000 + 20 T) dT (100 + 50 T + 0.04 T2) dT
Applying limits T1 to T2, We get
W =
T2
T1
(5000 + 20 T ) dT
T2
T1
(100 + 50 T + 0.04 T 2 ) dT
2
2
T2
T2
T3
T
T
5000
20
100
50
0
.
04
+
+
=
2
2
3
T1
T1
(T 2 T12 )
(T22 T12 )
(T 3 T13 )
+ 0.04 2
W = 5000 (T2 T1 ) + 20 2
100 (T2 T1 ) + 50
2
2
3
Substituting the values of T1 and T2 in above equation, we get,
(1000 2 500 2 )
(1000 2 500 2 )
(1000 3 500 3 )
5000
(
1000
500
)
+
20
100
(
1000
500
)
+
50
+
0
.
04
W =
2
2
3
= [10 106] [ 30.467 106]
= (10 30.467) 106
W = 20.467 10 3 kJ
Therefore, work done, W = 20.467 103 kJ.
( JNTU-Kakinada )
B.Tech. II-Year I-Sem.
Spectrum ALL-IN-ONE Journal for Engineering Students, 2013
S.16
Q2. (a) State and prove Carnots theorem.
Answer :
Nov.-12, Set-2, Q2(a)
For answer refer Unit-III, Q14.
(b) A house is to be maintained at a temperature of 20C by means of a heat pump pumping heat
from the atmosphere. Heat losses through the walls of the house are estimated at 0.65 kW
per unit of temperature difference between the inside of house and atmosphere.
(i) If the atmospheric temperature is 10C, what is the minimum power required to drive
the pump?
(ii) It is proposed to use the same heat pump to cool the house in the summer. For the same
room temperature, the same heat loss rate, and the same power input to the pump, what
is the maximum permissible atmospheric temperature?
Answer :
Nov.-12, Set-2, Q2(b)
Given that,
Ti = 20C
= 20 + 273
= 293 K
T0 = 10c
= 10 + 273
= 263 K
Qi = 0.65
kW
T
K
Sink (House)
To' = 20C
Sink (House)
Ti = 20C
Q' o
Qi
Heat
pump
Qo
Source
To = 10C
Figure (i): Supplying Heat to House
(i)
Heat
pump
Q'i
Source
Ti'
Figure (ii): Extracting Heat from House
The minimum power required is given by,
W=
Qi
(C . O . P). HP
... (1)
Where,
(C.O.P)H.P =
Ti
293
=
293 263
Ti To
(C.O.P)H.P = 9.767
B.Tech. II-Year I-Sem.
( JNTU-Kakinada )
S.17
Thermodynamics (November-2012, Set-2) JNTU-Kakinada
And
Ti ' = 30.003 + 293
Qi = 0.65
kW
T
K
T0' = 323.003 K
= 0.65 (Ti To)
= 0.65 (293 263)
Qi = 19.50 kW
Therefore, Substituting above values in equation (1),
We get,
Therefore, maximum permissible atmospheric
temperature, Ti ' = 323.003 K.
Q3.
19.50
W =
9.767
W = 1.997 kW
(ii)
(C.O.P)Ref =
(C.O.P)Ref =
[QT
'
si
Ti ' T0'
= 293 K
(
= 0.65 (T
Heat removed, Qo' = 0.65 Ti ' To'
Qo'
'
293
CP = 1.005 kJ/kg K
CV = 0.720 kJ/kg K
(i)
P1 = 6 bar
P2 = 3 bar
0.65(Ti 293)
1.997
Equating equations (2) and (3), we get
(Ti' 293) 2
... (3)
For isothermal process, temperature is constant, i.e.,
T1 = T2
From ideal gas equation, we have,
0.65(Ti' 293)
1.997
P1V1 P2V2
=
T1
T2
P1V1 =P2V2
= 0.65
(Ti' 293) 2 =
293 1.997
0.65
(Ti' 293) 2 = 900.186
(Ti' 293) =
900.186
Ti' 293 = 30.003
( JNTU-Kakinada )
Air Expands Isothermally from 6 Bar to 3 Bar
Given that,
'
293 1.997
Nov.-12, Set-2, Q3
Given that,
Qo'
(C.O.P)Ref =
W
(Ti 293)
Air is compressed to half the volume at
constant pressure and
m = 1 kg
Thus,
'
(ii)
What would be the change in entropy of the
air undergoes the above three processes in
sequence. Take C P = 1.005 kJ/kgK and
CV = 0.720 kJ/kgK.
... (2)
Ti' 293
293
Air expands isothermally from 6 bar to
3 bar
Answer :
293
(C.O.P)Ref =
(i)
(iii) Heat is supplied to air at constant
volume till the pressure becomes three
fold
Therefore, minimum power required, W = 1.997 kW
Heat pump for summer
We know that,
T0'
Calculate the changes of entropy per kg of
air in the following cases,
[Q T1 =T2]
P1 V2
=
P2 V1
Substituting P1 and P2 values in above equation,
6 V2
=
3 V1
V2 = 2 V1
B.Tech. II-Year I-Sem.
Spectrum ALL-IN-ONE Journal for Engineering Students, 2013
S.18
Therefore, change in entropy is given by,
V
P
(S)1 = S2 S1 = 2.3 m CV log 2 + C P log 2
V1
P1
2V
3
= 2.3 1 0.720 log + 1.005 log 1
6
V1
(ii)
= 2.3 1[ 0.217 + 0.303]
= 2.3 1 0.086
(S)1 = S2 S1 = 0.198 kJ/kg-K
Air is Compressed Half the Volume at Constant Pressure
Given that,
V2 = 0.5 V1
P1 =P2
Therefore, change in entropy is given by,
P
V
(S)2 = S2 S1 = 2.3 m CV log 2 + C P log 2
P1
V1
Substituting, m = 1, P1 = P2 and V2 = 0.5 V1 in above equation, we get,
P
(S)2 = S2 S1 = 2.3 1 0.720 log 2
P2
(iii)
0.5V1
+ 1.005 log
V1
= 2.3 1 [ 0 + ( 0.303)]
(S)2 = S2 S1 = 0.696 kJ/kg-K
Heat Supplied to Air at Constant Volume Till the Pressure Becomes Three Forth
Given that,
V1 = V2
P2 = 3P1
Change in entropy is given by,
P
(S)3 = S2 S1 = 2.3 m CV log 2
P1
V
+ C P log 2
Substituting, m = 1, V1 = V2 and P2 = 3P1 in above equation, we get,
3P
V
(S)3 = S2 S1 = 2.3 1 0.720 log 1 + 1.005 log 2
P
1
V1
= 2.3 1 [ 0.344 + 0]
(S)3 = S2 S1 = 0.791 kJ/kg-K
The change in entropy of the air if it undergoes all the above three processes in sequence is given by,
(S)T = (S)1 + (S)2 + (S)3
= 0.198 0.696 + 0.791
(S )T = 0.293 kJ/kg - K
B.Tech. II-Year I-Sem.
( JNTU-Kakinada )
Thermodynamics (November-2012, Set-2) JNTU-Kakinada
Q4.
(a)
S.19
Using the Maxwells relations, obtain the expression for the difference of specific heats at
constant pressure and constant volume in terms of the derivatives of properties. Hence show
that CP CV is always positive.
Nov.-12, Set-2, Q4(a)
Answer :
From entropy relations, we have,
V
dP
Tds = CPdT T
T P
... (1)
P
dv
Tds = CVdT + T
T V
... (2)
Equating equations (1) and (2), we get,
P
V
CPdT T
dP = CVdT + T dv
T V
T P
P
V
(CP CV)dT = T
dv + T
dP
T V
T P
P
v
T
T
dP
T V
T P
+
dv
dT =
(C P CV )
(C P CV )
We know that,
T
T
dV +
dP
dT =
V P
P V
Comparing the coefficients of dV and dP in above two dT equations, we get,
P
T
T V
T
=
(C P CV )
V P
V
T
T
T P
=
(C P CV ) P V
... (3)
... (4)
From equation (4),
V
T
T P
C P CV =
T
P V
V P
CP C V = T
T P T V
( JNTU-Kakinada )
... (5)
B.Tech. II-Year I-Sem.
Spectrum ALL-IN-ONE Journal for Engineering Students, 2013
S.20
But, from relations of thermodynamic variables, we have,
P T V
= 1
T V v P P T
P
=
T V T
1
V
P P T
P
V P
T V
T P v T
... (6)
Substitute equation (6) in equation (5), we get,
2
v P
C P CV = T
T P V T
As volume expansivity () =
... (7)
1 V
1 V
and isothermal compressibility (K ) =
T
V T P
V P T
... (8)
Equation (7) can be expressed in terms of and K as,
TV 2 2
vK T
C P CV =
C P CV =
[Use equation (8)]
TV 2
KT
V
P
is always positive and
is negative for a substance.
Since
T P
V T
Thus, CP CV will be always positive.
(b)
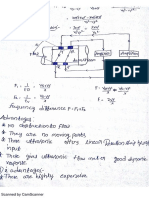
With the help of a schematic diagram, explain the measure of steam quality using the separating and throttling calorimeter.
Answer :
Nov.-12, Set-2, Q4(b)
Control valve
Thermometer
Inlet of
wet steam
Throttling calorimeter
Water
Gauge
Separating
calorimeter
Manometer
Outlet of
dry steam
Figure: Separating and Throttling Calorimeter
B.Tech. II-Year I-Sem.
( JNTU-Kakinada )
Thermodynamics (November-2012, Set-2) JNTU-Kakinada
S.21
The combined separating and throttling calorimeter is shown in figure. It employs a steam which passes through a
tube directed by a control valve. Initially, steam enters the separating calorimeter where in maximum amount of moisture is
separated and is calibrated by a water gauge. Then, the dry steam coming from a separating calorimeter is sent to a throttling
calorimeter where it is throttled. The exhaust steam is collected from an outlet port and its dryness fraction is measured.
Let, x = Actual dryness fraction of steam
x1 = Dryness fraction of steam at separating calorimeter
x2 = Dryness fraction of steam at throttling calorimeter
Then, actual dryness fraction of steam,
x = x1 x2
x=
M . x2
M +m
Q x1 = M + m
Merit
This method is employed for measurement of quality even though the wet steam is not sufficient to take it to a super
heated region.
Demerits
1.
In this method, the separating calorimeter cannot separate water particles from wet steam completely.
2.
This method is not suitable for very wet steam.
Q5. (a) Explain the law of corresponding states. Hence briefly explain the significance of compressibility
chart.
Answer :
Nov.-12, Set-2, Q5(a)
Law of Corresponding States
It states that gases behave in similar direction if they are applied with reduced temperature and pressures beyond
their critical point.
Compressibility charts are used for determining the compressibility factor Z which is a function of temperature and
pressure.
Significance of Compressibility Chart
For answer refer Unit-VI, Q26, Topic: Uses of Compressibility Chart.
(b) Two vessels A and B, each of volume 3 m3 may be connected together by a tube of negligible
volume. Vessel A contains air at 7 bar, 95C and vessel B contains air at 3.5 bar, 205C. Find
the change of entropy when A is connected to B. Assume the mixing to be complete and
adiabatic.
Answer :
Nov.-12, Set-2, Q5(b)
Given that,
VA = 3 m3
PA = 7 bar
TA = 95C = 95 + 273
= 368 K
PB = 3.5 bar
TB = 205C = 205 + 273
= 478 K
Assume,
CP = 1.005 kJ/kg K
Cv = 0.720 kJ/kg K
R = 0.287 kJ/kg K
( JNTU-Kakinada )
B.Tech. II-Year I-Sem.
Spectrum ALL-IN-ONE Journal for Engineering Students, 2013
S.22
From gas equation, we have,
For vessel A
Mass, mA =
mA =
PAV A
RT A
7 3 10 2
0.287 368
mA = 19.883 kg
For Vessel B
mB =
PBVB
RTB
[Q VA = VB = 3 m3]
3.5 3 102
0.287 478
mB = 7.654 kg
Total mass, m = mA + mB
= 19.883 + 7.654
m = 27.537 kg
Since, there is adiabatic mixing the internal energy must be equal.
UA= mA CV TA
= 19.883 0.720 368
UA = 5268.2 kJ
UB = mB CV TB
= 7.654 0.720 478
UA= 2634.201 kJ
We know that,
UA + UB = (mA + mB) CV T
5268.2 + 2634.201 = (19.883 + 7.654) 0.720 T
7902.401 = 19.827 T
T = 398.575 K
Therefore, final temperature, T = 398.575 K
Final pressure can be obtained by equation,
P V = mRT
P=
mRT
(V A + V B )
27.537 0.287 398.575
3+ 3
P = 524.998 kPa
Therefore pressure, P = 524.998 kPa
B.Tech. II-Year I-Sem.
( JNTU-Kakinada )
Thermodynamics (November-2012, Set-2) JNTU-Kakinada
S.23
Change in entropy is given by,
For Vessel A
T
P
(S)A = mA C P ln T R ln P
A
A
398.575
524.998
= 19.883 1.005 ln 368 0.287 ln
7 10 2
= 19.883 (0.163)
(S)A = 3.241 kJ/kg K
For Vessel B
T
(S)B = mB C P ln T
B
P
R ln
PB
398.575
524.998
0.287 ln
= 7.654 1.005 ln
478
3.5 10 2
= 7.654 ( 0.299)
(S ) B = 2.289 kJ/kg - K
Total change in entropy is,
(S) = (S)A + (S)B
= 3.241 + ( 2.289)
(S ) = 0.952 kJ/kg - K .
Q6.
(a)
Define the following properties,
(i)
Humidity ratio
(ii) Dew point temperature and
(iii) Relative humidity
Nov.-12, Set-2, Q6(a)
Answer :
(i)
Humidity Ratio
For answer refer Unit-IV, Q20, Topic: Specific Humidity.
(ii)
Dew Point Temperature
For answer refer Unit-VI, Q19, Topic: Dew Point Temperature.
(iii)
Relative Humidity
For answer refer Unit-VI, Q20, Topic: Relative Humidity.
(b)
Cooling water enters the cooling tower at a rate of 1000 kg/hr and 70C. Water is pumped
from the base of the tower at 24C and some makeup water is added afterwards. Air enters
the tower at 15C, 50% RH, 1.01325 bar and leaves the tower saturated at 34C, 1 bar. Calculate
the flow rate of dry air in kg/hr and the makeup water required per hour.
Nov.-12, Set-2, Q6(b)
( JNTU-Kakinada )
B.Tech. II-Year I-Sem.
Spectrum ALL-IN-ONE Journal for Engineering Students, 2013
S.24
Q7.
Answer :
Given that,
Mw = 1000 kg/hr
T1 = 15C, T3 = 70C
T2 = 34C T4 = 24C
P1 = 1.01325 bar
P2 = 1 bar
R.H = 50%
From psychrometric chart,
At, T1 = 15C, R. H = 50%
h1 = 28.44 kJ/kg, w1 = 0.0052
v1 = 0.822 m3/kg
At, T2 = 34C
h2 = 122.46 kJ/kg
w2 = 0.0344 kg/kg dry air
2 = 0.917 m3/kg
At, T3 = 70C
h 3 = hf
= 293 kJ/kg
At, T4 = 24C
h 4 = hf
= 100.6 kJ/kg
(i)
An engine working on Otto cycle has
an air standard efficiency of 56% and
rejects 544 kJ/kg of air. The pressure
and temperature of air at the beginning
of compression are 0.1 MPa and 60C
respectively. Compute,
(i) The compression ratio of the engine
and
(ii) Maximum pressure in the cycle.
Nov.-12, Set-2, Q7(a)
Answer :
Given that,
P1 = 0.1 MPa
P1 = 100 kPa
T1 = 60C = 60 + 273
= 333 K
Q = 544 kJ/kg
cycle = 56%
= 0.56
We know that,
(i)
Compression Ratio
cycle= 1
1
(rp ) 1
0.56 = 1
1
(rp ) 1
1
[Q = 1.4]
Mass flow rate of dry air is given by,
m& a =
m& w (h3 h4 )
( h2 h1 ) ( w2 w1 )h4
0.56 = 1
m& a =
1000 (293 100.6)
(122.46 28.44) (0.0344 0.0052) 100.6
0.56 = 1
m& a = 2112 .371 kg hr
Therefore, mass flow rate dry air, m& a= 2112.371 kg/hr
(ii)
(a)
Make up water required,
m& make up = m& a(w2 w1)
= 2112.271 (0.0344 0.0052)
m& make up = 61.678 kg hr
Therefore, mass flow rate of make-up water,
m& make up = 61.678 kg/hr
B.Tech. II-Year I-Sem.
1
(r p ) 0.4
(rp )1.41
1
(rp ) 0.4
= 1 0.56
1
= 0.44
(r p ) 0.4
(rp)0.4 =
1
0.44
(rp)0.4 = 2.273
rp = 7.789
Therefore, compression ratio, rp = 7.789
( JNTU-Kakinada )
S.25
Thermodynamics (November-2012, Set-2) JNTU-Kakinada
Since, we have,
But,
Assume
Q R = 0.287 kJ kgK
Cv = 0.72 kJ kg -
RT1
V1 = P
1
V1 =
P2 V1
=
P1 V2
P2
= (rp)
P1
0.287 333
100
V1 = 0.956 m3/kg
P2
= (7.789)1.4
100
V1
Compression ratio, rp =
V2
7.789 =
P2 = 1770.412 kPa
0.956
V2
Substituting the above values in equation (1), we get,
V2 = 0.123 m3/kg
P3 0.123 1.770 0.123
=
756.236
1511.792
The temperature at the heat supplied region is given
by,
756.236 P3 = 1.770 1511.792
P3 = 3.538 MPa
1.41
P2 = 1.770 MPa
For Otto cycle, V3 = V2 = 0.123 m3/kg
V2 7.789 = 0.956
T2 V1
=
T1 V2
0.956
T2
=
333 0.123
Therefore, maximum pressure, P3 = 3.538 MPa
T2 = 333 (7.772)0.4
Pressure (P)
T2 = 756.236 K
Heat rejected by the engine,
Q = CV (T3 T2)
544 = 0.720 (T3 756.236)
755.556 = T3 756.236
3
2
Pr = C
T3 = 755.556 + 756.236
T3 = 1511.792 K
Volume (V)
Therefore, T3 = Tmax = 1511.792 K
Figure: P.V Diagram for Otto Cycle
Maximum temperature, T3 = 1511.792 K
(ii)
(b)
Maximum Pressure
We know that,
P3V3 P2V2
=
T2
T3
( JNTU-Kakinada )
... (1)
Explain the simple Rankine cycle with
the help of a schematic and T-S diagram.
Nov.-12, Set-2, Q7(b)
Answer :
For answer refer Unit-VIII, Q1.
B.Tech. II-Year I-Sem.
Spectrum ALL-IN-ONE Journal for Engineering Students, 2013
S.26
Q8.
Write short notes on the following,
(a)
Clausius inequality
(b)
Clausius Clapeyron equation
(c)
Equivalence of Clausius and Kelvin-Plank statements of second law of thermodynamics.
Nov.-12, Set-2, Q8
Answer :
(a)
Clausius Inequality
For answer refer Unit-III, Q20.
(b)
Clausius-Clapeyron Equation
For answer refer Unit-IV, Q18.
(c)
Equivalence of Clausius and Kelvin-Planck Statements of Second Law of Thermodynamics
For answer refer Unit-III, Q13, Topic: Equivalence of Kelvin-Planck and Clausius Statements.
B.Tech. II-Year I-Sem.
( JNTU-Kakinada )
Potrebbero piacerti anche
- Heat Transfer Homework 3 Solutions ME 335Documento10 pagineHeat Transfer Homework 3 Solutions ME 335David SebulskyNessuna valutazione finora
- Thermodynamics and Applications - Test-1Documento6 pagineThermodynamics and Applications - Test-1sap2279Nessuna valutazione finora
- A Modern Course in Statistical PhysicsDa EverandA Modern Course in Statistical PhysicsValutazione: 3.5 su 5 stelle3.5/5 (2)
- Thermodynamics Worked ExamplesDocumento13 pagineThermodynamics Worked ExamplesSalah Salman100% (1)
- HYD ME 2 1 Thermo Set 1Documento10 pagineHYD ME 2 1 Thermo Set 1manikantar15Nessuna valutazione finora
- Process Intensification for Sustainable Energy ConversionDa EverandProcess Intensification for Sustainable Energy ConversionNessuna valutazione finora
- Thermal Physics Tutorial Solution 2012 (Students Copy)Documento8 pagineThermal Physics Tutorial Solution 2012 (Students Copy)Wee Chee LimNessuna valutazione finora
- Alcorcon PIPE Merged SolvedDocumento80 pagineAlcorcon PIPE Merged SolvedBernalynMalinaoNessuna valutazione finora
- Alcorcon Engineering Review Center: Power & Industrial Plant EngineeringDocumento13 pagineAlcorcon Engineering Review Center: Power & Industrial Plant EngineeringNeil SequioNessuna valutazione finora
- Air PropDocumento4 pagineAir Propanup_nairNessuna valutazione finora
- We M5Documento14 pagineWe M5knyogishNessuna valutazione finora
- Thermal MCQDocumento42 pagineThermal MCQRanjeet KumarNessuna valutazione finora
- Problem 5.1ADocumento53 pagineProblem 5.1ALuis PiscalNessuna valutazione finora
- Steam Engines Machinery's Reference Series, Number 70Da EverandSteam Engines Machinery's Reference Series, Number 70Nessuna valutazione finora
- RESPUESTAS 235.Documento4 pagineRESPUESTAS 235.blancanestor76Nessuna valutazione finora
- Air PropDocumento4 pagineAir PropWrya SaeedNessuna valutazione finora
- A Rigid Tank Contains 2 Kmol of N2 and 6 Kmol of CO2 Gases at 3000K and 15 MpaDocumento19 pagineA Rigid Tank Contains 2 Kmol of N2 and 6 Kmol of CO2 Gases at 3000K and 15 MpaJustin SantosNessuna valutazione finora
- Thermodynamics (Solved Prob)Documento22 pagineThermodynamics (Solved Prob)Jubert Perez100% (1)
- BB101 Engineering Science Chapter 6 Temperature and HeatDocumento7 pagineBB101 Engineering Science Chapter 6 Temperature and HeatAh Tiang100% (4)
- Physics 2101 Assignment Problem 1 Heat TransferDocumento9 paginePhysics 2101 Assignment Problem 1 Heat TransfersqrlzNessuna valutazione finora
- Solution Week 9Documento6 pagineSolution Week 9Ariadne ChuaNessuna valutazione finora
- HW 5 SolnDocumento7 pagineHW 5 SolnNik Hafiy HafiziNessuna valutazione finora
- AP Physics Problems - Thermodynamics and Heat TransferDocumento10 pagineAP Physics Problems - Thermodynamics and Heat TransferOPEN ARMSNessuna valutazione finora
- Chapter 03 The First Law of Thermodynamics (PP 59-81)Documento23 pagineChapter 03 The First Law of Thermodynamics (PP 59-81)Muhammad Ashfaq Ahmed100% (1)
- Multiple choice questions on mechanics, heat and thermodynamicsDocumento21 pagineMultiple choice questions on mechanics, heat and thermodynamicsGodwinNessuna valutazione finora
- First Law of Thermodynamics 1580126216698243765e2ed008943f7Documento3 pagineFirst Law of Thermodynamics 1580126216698243765e2ed008943f7Atul YadavNessuna valutazione finora
- Cooling Tower Thermodynamics Lab ReportDocumento10 pagineCooling Tower Thermodynamics Lab ReportMohammed IslamNessuna valutazione finora
- HWSolutions PDFDocumento42 pagineHWSolutions PDFJames AhnNessuna valutazione finora
- Mae 320 HW 04 SolDocumento7 pagineMae 320 HW 04 SolEvan DurstNessuna valutazione finora
- Unit Four Homework Solutions, September 23. 2010: Mechanical Engineering 370 ThermodynamicsDocumento3 pagineUnit Four Homework Solutions, September 23. 2010: Mechanical Engineering 370 ThermodynamicsRengganis Putri ParmudyaNessuna valutazione finora
- CHE Thermodynamics Make-up Exam Q2 (40chDocumento3 pagineCHE Thermodynamics Make-up Exam Q2 (40chkellykapperNessuna valutazione finora
- 108 Soln 1Documento7 pagine108 Soln 1ria setianingrumNessuna valutazione finora
- Class Tutorial 2 Steam Cooling Process Heat CalculationDocumento1 paginaClass Tutorial 2 Steam Cooling Process Heat CalculationmarkNessuna valutazione finora
- CH 3Documento63 pagineCH 3Hoi An Sze100% (7)
- Uitwerking We Co 13Documento5 pagineUitwerking We Co 13henk waterbeenNessuna valutazione finora
- Xii Physics CH 11 Numerical sheetDocumento3 pagineXii Physics CH 11 Numerical sheethirahera436Nessuna valutazione finora
- Lecture 4Documento8 pagineLecture 4Refai SayyadNessuna valutazione finora
- Second Law of Thermodynamics Practice Questions - Without AnsDocumento4 pagineSecond Law of Thermodynamics Practice Questions - Without AnsKirti KumarNessuna valutazione finora
- HW4VM235SU2016Documento2 pagineHW4VM235SU2016tony960129Nessuna valutazione finora
- Worked Example For Cooling TowerDocumento2 pagineWorked Example For Cooling TowerErnesto Sánchez GómezNessuna valutazione finora
- Tutorial Sheet 02 Answers 2014Documento24 pagineTutorial Sheet 02 Answers 2014checkmeout803100% (1)
- B.T. Kumaon Institute of Technology, Dwarahat End Semester (Back) Examination, 2020-2021Documento2 pagineB.T. Kumaon Institute of Technology, Dwarahat End Semester (Back) Examination, 2020-2021verma.ashok031Nessuna valutazione finora
- Calculating heat energy radiated from a furnace flameDocumento5 pagineCalculating heat energy radiated from a furnace flameIvy Joy Ubina100% (1)
- HEATING STEAM PROBLEMDocumento31 pagineHEATING STEAM PROBLEMSirish Chand Putla0% (1)
- BSGS Sample Problems 2 - BB CollabDocumento21 pagineBSGS Sample Problems 2 - BB CollabNeo GarceraNessuna valutazione finora
- Phys 181 CH 2122 Sample ProblemsDocumento4 paginePhys 181 CH 2122 Sample Problemsluis atenciaNessuna valutazione finora
- Ch19 SSMDocumento24 pagineCh19 SSMme12m113Nessuna valutazione finora
- HW 10Documento2 pagineHW 10hangman001Nessuna valutazione finora
- Module 2 - Heat and ThermodynamicsDocumento12 pagineModule 2 - Heat and ThermodynamicsANGELO NINO ALVARADONessuna valutazione finora
- Antivirus DeveloperDocumento21 pagineAntivirus DevelopersarathNessuna valutazione finora
- WWW Campusgate Co in 2011 10 Time and Work HTMLDocumento16 pagineWWW Campusgate Co in 2011 10 Time and Work HTMLsarathNessuna valutazione finora
- WWW Campusgate Co in 2011 10 Time and Work HTMLDocumento16 pagineWWW Campusgate Co in 2011 10 Time and Work HTMLsarathNessuna valutazione finora
- Expt Lab IandcsDocumento3 pagineExpt Lab IandcssarathNessuna valutazione finora
- 24 .Area Fundemental ConceptsDocumento9 pagine24 .Area Fundemental Conceptsz1y2Nessuna valutazione finora
- JNTUDocumento75 pagineJNTUbrijkishor2017Nessuna valutazione finora
- PartnershipDocumento3 paginePartnershipz1y2Nessuna valutazione finora
- INstrumentation and Control Systems Unit4Documento1 paginaINstrumentation and Control Systems Unit4sarathNessuna valutazione finora
- Mechanism and Types of ClutchesDocumento41 pagineMechanism and Types of Clutchessarath100% (1)
- Low Cost HousingDocumento35 pagineLow Cost HousingsarathNessuna valutazione finora
- Fluid Mechanics Mech Gate Ies NotesDocumento21 pagineFluid Mechanics Mech Gate Ies NotesSourabh Nanda50% (2)
- 4.1 Understanding Thermal EquilibriumDocumento47 pagine4.1 Understanding Thermal EquilibriumcgharyatiNessuna valutazione finora
- Module 03 Vapor Compression RefrigerationDocumento10 pagineModule 03 Vapor Compression RefrigerationUkas RiyupiNessuna valutazione finora
- Project energy analysis and HVAC load calculationDocumento4 pagineProject energy analysis and HVAC load calculationgurramviswaNessuna valutazione finora
- Chapter 7 Temperature MeasurementDocumento11 pagineChapter 7 Temperature MeasurementHoàng DoãnNessuna valutazione finora
- STD 8 Geography Chapter 3Documento5 pagineSTD 8 Geography Chapter 3Royston NorbertNessuna valutazione finora
- Performance Evaluation of Air ConditionersDocumento11 paginePerformance Evaluation of Air Conditionershybri8Nessuna valutazione finora
- 11-An - Industrial Dehumidifier SizingDocumento4 pagine11-An - Industrial Dehumidifier Sizingthilangac100% (1)
- Refrigeration COP and Heat CalculationsDocumento19 pagineRefrigeration COP and Heat CalculationskelvinNessuna valutazione finora
- Effects of Meteorology on Air PollutionDocumento14 pagineEffects of Meteorology on Air Pollutionaruna mahadevan67% (9)
- Práctica A RefrigeraciónDocumento4 paginePráctica A RefrigeraciónAndres Ante VargasNessuna valutazione finora
- Thermal ComfortDocumento4 pagineThermal ComfortVidya HittiNessuna valutazione finora
- Fcu-Rf-01 Heat Load CalculationDocumento11 pagineFcu-Rf-01 Heat Load CalculationKhalifa RiswanNessuna valutazione finora
- Rumus AntoineDocumento7 pagineRumus AntoineGaluh ChynintyaNessuna valutazione finora
- HVAC Design Using E20-II SoftwareDocumento88 pagineHVAC Design Using E20-II SoftwareBryan Ng Horng HengNessuna valutazione finora
- Boles Lecture Notes Thermodynamics Chapter 10Documento15 pagineBoles Lecture Notes Thermodynamics Chapter 10prince assiriNessuna valutazione finora
- Air Conditioning Unit With Remote Air Cooled Condenser: Working ConditionsDocumento2 pagineAir Conditioning Unit With Remote Air Cooled Condenser: Working ConditionsJulianto zhengNessuna valutazione finora
- Emd5m6a1 Lab Report Thermo 2Documento21 pagineEmd5m6a1 Lab Report Thermo 2Zulfaris NasirNessuna valutazione finora
- Technical YCWMDocumento8 pagineTechnical YCWMnairam2003Nessuna valutazione finora
- Guias Tecnicos Aplicacoes Avac en 0Documento25 pagineGuias Tecnicos Aplicacoes Avac en 0cachiletNessuna valutazione finora
- Presentation of Mary School of Clarin, IncDocumento5 paginePresentation of Mary School of Clarin, IncjuicymayNessuna valutazione finora
- Hvac & AhuDocumento23 pagineHvac & AhuTanaya Vankudre100% (1)
- Determining Dew Point and Minimum Application TemperatureDocumento1 paginaDetermining Dew Point and Minimum Application TemperatureMircea BobarNessuna valutazione finora
- Air-Conditioning Lexicon - A To Z Index - StulzDocumento7 pagineAir-Conditioning Lexicon - A To Z Index - StulzAnkesh RajNessuna valutazione finora
- Cooling Capacity Calculation PrintingDocumento1 paginaCooling Capacity Calculation PrintingJay PatelNessuna valutazione finora
- Measuring Forehead Temperatures in SecondsDocumento10 pagineMeasuring Forehead Temperatures in SecondsGheorghe ManolacheNessuna valutazione finora
- RAC Lab AssignmentDocumento6 pagineRAC Lab AssignmentAhmed AliNessuna valutazione finora
- Process of Condensation: Group 7Documento12 pagineProcess of Condensation: Group 7Andrei CalmaNessuna valutazione finora
- Thermometer Temperature CalculationsDocumento27 pagineThermometer Temperature CalculationsFrances GalangNessuna valutazione finora
- Week 1 Lecture NoteDocumento4 pagineWeek 1 Lecture Notealibaba011Nessuna valutazione finora
- Practical Guides to Testing and Commissioning of Mechanical, Electrical and Plumbing (Mep) InstallationsDa EverandPractical Guides to Testing and Commissioning of Mechanical, Electrical and Plumbing (Mep) InstallationsValutazione: 3.5 su 5 stelle3.5/5 (3)
- Hyperspace: A Scientific Odyssey Through Parallel Universes, Time Warps, and the 10th DimensionDa EverandHyperspace: A Scientific Odyssey Through Parallel Universes, Time Warps, and the 10th DimensionValutazione: 4.5 su 5 stelle4.5/5 (3)
- Piping and Pipeline Calculations Manual: Construction, Design Fabrication and ExaminationDa EverandPiping and Pipeline Calculations Manual: Construction, Design Fabrication and ExaminationValutazione: 4 su 5 stelle4/5 (18)
- Einstein's Fridge: How the Difference Between Hot and Cold Explains the UniverseDa EverandEinstein's Fridge: How the Difference Between Hot and Cold Explains the UniverseValutazione: 4.5 su 5 stelle4.5/5 (50)
- The Laws of Thermodynamics: A Very Short IntroductionDa EverandThe Laws of Thermodynamics: A Very Short IntroductionValutazione: 4.5 su 5 stelle4.5/5 (10)
- Pressure Vessels: Design, Formulas, Codes, and Interview Questions & Answers ExplainedDa EverandPressure Vessels: Design, Formulas, Codes, and Interview Questions & Answers ExplainedValutazione: 5 su 5 stelle5/5 (1)
- Introduction to Applied Thermodynamics: The Commonwealth and International Library: Mechanical Engineering DivisionDa EverandIntroduction to Applied Thermodynamics: The Commonwealth and International Library: Mechanical Engineering DivisionValutazione: 2.5 su 5 stelle2.5/5 (3)
- Handbook of Mechanical and Materials EngineeringDa EverandHandbook of Mechanical and Materials EngineeringValutazione: 5 su 5 stelle5/5 (4)
- Quantum Mechanics 4: Spin, Lasers, Pauli Exclusion & Barrier PenetrationDa EverandQuantum Mechanics 4: Spin, Lasers, Pauli Exclusion & Barrier PenetrationValutazione: 1 su 5 stelle1/5 (1)
- Oil and Gas Pipelines: Integrity and Safety HandbookDa EverandOil and Gas Pipelines: Integrity and Safety HandbookNessuna valutazione finora
- Rolling Bearing Tribology: Tribology and Failure Modes of Rolling Element BearingsDa EverandRolling Bearing Tribology: Tribology and Failure Modes of Rolling Element BearingsNessuna valutazione finora
- 1,001 Questions & Answers for the CWI Exam: Welding Metallurgy and Visual Inspection Study GuideDa Everand1,001 Questions & Answers for the CWI Exam: Welding Metallurgy and Visual Inspection Study GuideValutazione: 3.5 su 5 stelle3.5/5 (7)
- Waves and Beaches: The Powerful Dynamics of Sea and CoastDa EverandWaves and Beaches: The Powerful Dynamics of Sea and CoastValutazione: 4 su 5 stelle4/5 (1)
- Formulas and Calculations for Drilling, Production, and Workover: All the Formulas You Need to Solve Drilling and Production ProblemsDa EverandFormulas and Calculations for Drilling, Production, and Workover: All the Formulas You Need to Solve Drilling and Production ProblemsNessuna valutazione finora
- Albert Einstein's Theory Of Relativity Explained SimplyDa EverandAlbert Einstein's Theory Of Relativity Explained SimplyNessuna valutazione finora
- Operational Amplifier Circuits: Analysis and DesignDa EverandOperational Amplifier Circuits: Analysis and DesignValutazione: 4.5 su 5 stelle4.5/5 (2)
- Machinery Failure Analysis Handbook: Sustain Your Operations and Maximize UptimeDa EverandMachinery Failure Analysis Handbook: Sustain Your Operations and Maximize UptimeValutazione: 3.5 su 5 stelle3.5/5 (4)
- Basic Alarm Electronics: Toolbox Guides for Security TechniciansDa EverandBasic Alarm Electronics: Toolbox Guides for Security TechniciansJohn SangerValutazione: 4 su 5 stelle4/5 (1)