Documenti di Didattica
Documenti di Professioni
Documenti di Cultura
Thermo Problems
Caricato da
Christer John UyCopyright
Formati disponibili
Condividi questo documento
Condividi o incorpora il documento
Hai trovato utile questo documento?
Questo contenuto è inappropriato?
Segnala questo documentoCopyright:
Formati disponibili
Thermo Problems
Caricato da
Christer John UyCopyright:
Formati disponibili
ME 24-221
THERMODYNAMICS I
Solutions to extra problems in chapter 8
November 9, 2000
Fall 2000
J. Murthy
8.5
Water is used as the working fluid in a Carnot cycle heat engine, where it changes
from saturated liquid to saturated vapor at 200C as heat is added. Heat is rejected
in a constant pressure process (also constant T) at 20 kPa. The heat engine powers
a Carnot cycle refrigerator that operates between -15C and +20C. Find the heat
added to the water per kg water. How much heat should be added to the water in
the heat engine so the refrigerator can remove 1 kJ from the cold space?
Solution:
Carnot cycle:
qH = TH (s2 s 1 ) = hfg = 473.15 (4.1014) = 1940 kJ/kg
TL = Tsat (20 kPa) = 60.06 oC
ref = QL / W = TL / (TH - TL ) = (273 - 15) / (20 - (-15))
= 258 / 35 = 7.37
W = QL / = 1 / 7.37 = 0.136 kJ
HE = 1 - 333/473 = 0.29
W = HE Q H H2O
Q H H2O = 0.136 / 0.296 = 0.46 kJ
8.12
A cylinder fitted with a piston contains ammonia at 50C, 20% quality with a
volume of 1 L. The ammonia expands slowly, and during this process heat is
transferred to maintain a constant temperature. The process continues until all the
liquid is gone. Determine the work and heat transfer for this process.
C.V. Ammonia in the cylinder.
Table B.2.1: T1 = 50C, x 1 = 0.20, V1 = 1 L
NH3
v1 = 0.001777 + 0.2 0.06159 = 0.014095
50 C
s 1 = 1.5121 + 0.2 3.2493 = 2.1620
m = V1/v1 = 0.001/0.014095 = 0.071 kg
1
v2 = vG = 0.06336,
Process: T = constant to x2 = 1.0,
s2 = sG = 4.7613
P = constant = 2.033 MPa
1W2 = PdV = Pm(v2 - v1) = 2033 0.071 (0.06336 - 0.014095) = 7.11 kJ
1Q 2 = TdS = Tm(s2 - s1) = 323.2 0.071(4.7613 - 2.1620) = 59.65 kJ
or
1Q2 = m(u2 - u1) + 1W2 = m(h2 - h1)
h1 = 421.48 + 0.2 1050.01 = 631.48,
h2 = 1471.49
1Q 2 = 0.071(1471.49 - 631.48) = 59.65 kJ
8.14
A cylinder fitted with a frictionless piston contains water. A constant hydraulic
pressure on the back face of the piston maintains a cylinder pressure of 10 MPa.
Initially, the water is at 700C, and the volume is 100 L. The water is now cooled
and condensed to saturated liquid. The heat released during this process is the Q
supply to a cyclic heat engine that in turn rejects heat to the ambient at 30C. If
the overall process is reversible, what is the net work output of the heat engine?
C.V.: H2O, 13, this is a control mass:
Continuity Eq.: m1 = m3 = m
P
2
m(u3-u1) = 1Q3 1W3;
Energy Eq.:
Process: P = C => 1W3 = P dV = Pm(v3-v1)
State 1: 700 C, 10 MPa, V1 = 100 L Table B.1.4
v1 = 0.04358 m3/kg => m = m1 = V1/v1 = 2.295 kg
h1 = 3870.5 kJ/kg,
s1 = 7.1687 kJ/kg K
State 3: P3 = P1 = 10 MPa, x3 = 0
T
3
1
2
Table B.1.2
s
h3 =hf = 1407.5 kJ/Kg, s3 = sf = 3.3595 kJ/Kg K
1Q 3 = m(u3-u1) + Pm(v3 - v1) = m(h3 - h1)
= -5652.6 kJ
Heat transfer to the heat engine:
-1Q 3
QH = -1Q3 = 5652.6 kJ
Take control volume as total water and heat engine.
Process: Rev.,
H.E.
Snet = 0 ; TL = 30oC
2nd Law:
Snet = m(s3 - s1) - Qcv/TL ;
Qcv = To m(s3 - s1) = -2650.6 kJ
=> QL = -Qcv = 2650.6 kJ
Wnet = WHE = QH - QL = 3002 kJ
QL
Tamb
WHE
8.22
A heavily-insulated cylinder fitted with a frictionless piston contains ammonia
6C, 90% quality, at which point the volume is 200 L. The external force on th
piston is now increased slowly, compressing the ammonia until its temperature
reaches 50C. How much work is done on the ammonia during this process?
Solution:
C.V. ammonia in cylinder, insulated so assume adiabatic Q = 0.
Cont.Eq.:
m2 = m1 = m ;
Entropy Eq.:
Energy Eq.:
m(u2 u1) = 1Q2 1W2
m(s2 s1) = dQ/T + 1S2 gen
State 1: T1 = 6oC, x1 = 0.9, V1 = 200 L = 0.2 m3
Table B.2.1 saturated vapor, P 1 = Pg = 534 kPa
v1 = vf + x1vfg = 0.21166 m3/kg,
u1 = uf + x1ufg = 207.414 + 0.91115.3 = 1211.2 kJ/kg
s1 = sf + x1sfg = 0.81166 + 0.94.4425 = 4.810 kJ/kg-K,
m1 = V1/v1 = 0.2 / 0.21166 = 0.945 kg
Process: 12 Adiabatic 1Q2 = 0 & Reversible 1S2 gen = 0 => s1 = s2
State 2: T2 = 50oC, s2 = s1 = 4.810 kJ/kg-K
superheated vapor, interpolate in Table B.2.2
v2 = 0.0684 m3/kg,
=>
P2 = 1919 kPa,
h2 = 1479.5 kJ/kg
u2 = h2 - P2v2 = 1479.5 - 19190.0684 = 1348.2 kJ/kg
T
2
2
1
1
v
Energy equation gives the work as
1W2
= m(u1 - u2) = 0.945 ( 1211.2 - 1348.2) = -129.4 kJ
8.27
An insulated cylinder/piston contains R-134a at 1 MPa, 50C, with a volume of
100 L. The R-134a expands, moving the piston until the pressure in the cylinder
has dropped to 100 kPa. It is claimed that the R-134a does 190 kJ of work against
the piston during the process. Is that possible?
C.V. R-134a in cylinder. Insulated so assume Q = 0.
State 1: Table B.5.2,
s1 = 1.7494,
v1 = 0.02185, u1 = 431.24 - 10000.02185 = 409.4,
m = V1/v1 = 0.1/0.02185 = 4.577 kg
m(u2 - u1) = 1Q2 - 1W2 = 0/ - 190 u2 = 367.89 kJ/kg
Energy Eq.:
State 2: P2 , u2 Table B.5.2:
T2 = -19.25C ; s2 = 1.7689 kJ/kg K
dQ/T + 1S2,gen = 1S2,gen = 0.0893 kJ/K
m(s2 - s1) =
Entropy Eq.:
This is possible since 1S2,gen > 0/
P s=C
1
T
1
2
2
v
8.29
A mass and atmosphere loaded piston/cylinder contains 2 kg of water at 5 MPa,
100C. Heat is added from a reservoir at 700C to the water until it reaches
700C. Find the work, heat transfer, and total entropy production for the system
and surroundings.
C.V. Water.
Process: P = const. so
1 W2
= P(V2 - V1)
U2 - U1 = 1Q2 - 1W2 or 1Q2 = H2 - H1 = m(h2 - h1)
P
1
T
2
v
1
v
1Q 2
= 2(3900.1 - 422.72) = 6954.76 kJ
1 W2
= 1Q2 - m(u2 - u1) = 874.6 kJ
B.1.4: h1 = 422.72, u1 = 417.52
s1 = 1.303, v1 = 0.00104
B.1.3: h2 = 3900.1, u2 = 3457.6
s2 = 7.5122, v2 = 0.08849
dQ/T + 1S2 gen = 1Q2/Tres + 1S2 gen
m(s2 - s1) =
1S 2 gen
= m(s2-s1) -1Q2/Tres = 2(7.5122 - 1.303) - 6954/973 = 5.27 kJ/K
8.41
A large slab of concrete, 5 8 0.3 m, is used as a thermal storage mass in a
solar-heated house. If the slab cools overnight from 23C to 18C in an 18C
house, what is the net entropy change associated with this process?
C.V.: Control mass concrete.
V = 5 8 0.3 = 12 m3
m = V = 2300 12 = 27600 kg
1Q 2 = mCT = 27600 0.65(-5) = -89700 kJ
SSYST = mC ln
T2
T1
= 27600 0.65 ln
291.2
= -305.4 kJ/K
296.2
SSURR = -1Q2/T0 = +89700/291.2 = +308.0 kJ/K
SNET = -305.4 + 308.0 = +2.6 kJ/K
8.44
A hollow steel sphere with a 0.5-m inside diameter and a 2-mm thick wall contains
water at 2 MPa, 250C. The system (steel plus water) cools to the ambient
temperature, 30C. Calculate the net entropy change of the system and
surroundings for this process.
C.V.: Steel + water. This is a control mass.
mSTEEL = (V)STEEL = 8050 (/6)[(0.504)3 - (0.5)3] = 12.746 kg
USTEEL = (mC)STEEL(T2 - T1) = 12.746 0.48(30 - 250) = -1346 kJ
VH2O = (/6)(0.5)3, m = V/v = 6.54510-2/0.11144 = 0.587 kg
v2 = v1 = 0.11144 = 0.001004 + x2 32.889 => x 2 = 3.35810-3
u2 = 125.78 + 3.35810-3 2290.8 = 133.5
s2 = 0.4639 + 3.35810-3 8.0164 = 0.4638
UH
1Q 2
2O
= mH2O(u2 - u1)H2O = 0.587(133.5 - 2679.6) = -1494.6
= -1346 + (-1494.6) = -2840.6
STOT = SSTEEL + SH2O = 12.746 0.48 ln (303.15 / 523.15)
+ 0.587(0.4638 - 6.545) = -6.908 kJ/K
SSURR = -1Q2/T0 = +2840.6/303.2 = +9.370 kJ/K
SNET = -6.908 + 9.370 = +2.462 kJ/K
8.47
Consider a Carnot-cycle heat pump having 1 kg of nitrogen gas in a
cylinder/piston arrangement. This heat pump operates between reservoirs at 300
K and 400 K. At the beginning of the low-temperature heat addition, the pressure
is 1 MPa. During this processs the volume triples. Analyze each of the four
processes in the cycle and determine
a.
The pressure, volume, and temperature at each point
b.
The work and heat transfer for each process
T
4
T1 = T2 = 300 K, T3 = T4 = 400 K,
P1 = 1 MPa, V2 = 3 V1
a) P2V2 = P1V1 => P2 = P1/3 = 0.3333 MPa
mRT1 1 0.2968 300
=
= 0.08904 m3
V1 =
P1
1000
N2
2
V2 = 0.26712 m3
k
400
P3 = P2(T3/T2)k-1 = 0.3333 3.5 = 0.9123 MPa
300
V 3 = V2
P2
P3
T3
T2
= 0.26712
0.3333 400
= 0.1302 m3
0.9123 300
k
400
P4 = P1(T3/T1)k-1 = 1 3.5 = 2.73707 MPa
300
V 4 = V1
P1
P4
T4
T1
= 0.08904
1
400
= 0.04337 m3
2.737 300
b) 1W2 = 1Q2 = mRT1 ln (P1/P2)
= 1 0.2968 300 ln(1/0.333) = 97.82 kJ
3W4 = 3Q 4 = mRT3 ln(P3/P4)
= 1 0.2968 400 ln(0.9123/2.737) = -130.43 kJ
2W3 = -mCV0(T3 - T2) = -1
0.7448(400 - 300) = -74.48 kJ
4W1 = -mCV0(T1 - T4) = -1
0.7448(300 - 400) = +74.48 kJ
2Q 3 =
0, 4Q1 = 0
8.52
A rigid storage tank of 1.5 m3 contains 1 kg argon at 30C. Heat is then
transferred to the argon from a furnace operating at 1300C until the specific
entropy of the argon has increased by 0.343 kJ/kg K. Find the total heat transfer
and the entropy generated in the process.
Solution:
C.V. Argon. Control mass. R = 0.20813,
m = 1 kg
Energy Eq.:
m (u2 - u1 ) = m Cv (T2 - T1) = 1Q2
Process: V = constant => v2 = v1
State 1:
P1 = mRT/V = 42.063 kPa
State 2:
s2 = s1 + 0.343,
s2 - s1 = Cp ln (T2 / T1 ) - R ln (T2 / T1 ) = Cv ln (T2 / T1 )
ln (T2 / T1 ) = (s2 - s1)/ Cv = 0.343/0.312 = 1.0986
Pv = RT
=>
(P2 / P1) (v2 / v1) = T2 / T1 = P2 / P1
T2 = 2.7 T1 = 818.3, P2 = 2.7 P1 = 113.57
T
2
1
v=C
2
P1
v
1Q 2
= 1 0.3122 (818.3 - 303.15) = 160.8 kJ
m(s2 s1) = 1Q2/Tres + 1S2 gen tot
1S 2 gen tot =
1 0.31 - 160.8 / (1300 + 273) = 0.208 kJ/K
8.66
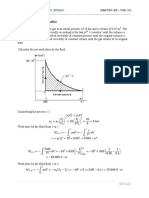
A cylinder/piston contains air at ambient conditions, 100 kPa and 20C with a
volume of 0.3 m3. The air is compressed to 800 kPa in a reversible polytropic
process with exponent, n = 1.2, after which it is expanded back to 100 kPa in a
reversible adiabatic process.
a.
Show the two processes in Pv and Ts diagrams.
b.
Determine the final temperature and the net work.
c.
What is the potential refrigeration capacity (in kilojoules) of the air at the
final state?
a)
T
2
m = P1V1/RT1
2
1
1
3
1w 2 = Pdv =
1
P2v2-P1v1
1-n
100 0.3
= 0.3565 kg
0.287 293.2
n- 1
b) T2 = T1(P2/P1) n
V
2
R(T2-T1)
1-n
8000.167
= 293.2
= 414.9 K
100
0.287(414.9-293.2)
= -174.6 kJ/kg
1-1.20
k-1
1000.286
= 228.9 K
T3 = T2(P3/P2) k = 414.9
800
2w 3 = CV0(T2-T3) = 0.717(414.9 - 228.9) = +133.3 kJ/kg
wNET = 0.3565(-174.6 + 133.3) = -14.7 kJ
c) Refrigeration: warm to T 0 at const P
3Q 1 = mCP0(T1 - T3) = 0.3565 1.004 (293.2 - 228.9) = 23.0 kJ
Potrebbero piacerti anche
- Flow in Pipes CompleteDocumento21 pagineFlow in Pipes CompleteSYaz WAni67% (6)
- Advanced Heat and Mass Transfer Textbook ReviewDocumento2 pagineAdvanced Heat and Mass Transfer Textbook ReviewSlim Kanoun0% (1)
- Thermodynamic processes and energy transfers in open and closed systemsDocumento12 pagineThermodynamic processes and energy transfers in open and closed systemsravitejabvNessuna valutazione finora
- CH 7 PowerCyclesDocumento12 pagineCH 7 PowerCyclescarleston thurgoodNessuna valutazione finora
- George Haller - Transport Barriers and Coherent Structures in Flow Data - Advective, Diffusive, Stochastic and Active Methods-Cambridge University Press (2023)Documento427 pagineGeorge Haller - Transport Barriers and Coherent Structures in Flow Data - Advective, Diffusive, Stochastic and Active Methods-Cambridge University Press (2023)Cesar RosalesNessuna valutazione finora
- HWSolutions PDFDocumento42 pagineHWSolutions PDFJames AhnNessuna valutazione finora
- Thermodynamics Worked ExamplesDocumento13 pagineThermodynamics Worked ExamplesSalah Salman100% (1)
- HW 5 SolnDocumento7 pagineHW 5 SolnNik Hafiy HafiziNessuna valutazione finora
- Thermodynamics Advanced HomeWork 1Documento11 pagineThermodynamics Advanced HomeWork 1فیضان قادرNessuna valutazione finora
- Solution Week 9Documento6 pagineSolution Week 9Ariadne ChuaNessuna valutazione finora
- Applied Thermodynamics Exam 2018 Wirh SolutionsDocumento9 pagineApplied Thermodynamics Exam 2018 Wirh SolutionsFarouk BassaNessuna valutazione finora
- CH 08Documento28 pagineCH 08hirenpatel_universalNessuna valutazione finora
- TH-005-Examples of Chapter Five PDFDocumento7 pagineTH-005-Examples of Chapter Five PDFLinda LCNessuna valutazione finora
- Heat Exchanger Design OptimizationDocumento27 pagineHeat Exchanger Design OptimizationUsama AkramNessuna valutazione finora
- MM301 5 Flow Analysis 3Documento15 pagineMM301 5 Flow Analysis 3efeln1Nessuna valutazione finora
- ThermodynamicsDocumento79 pagineThermodynamicstixy2013Nessuna valutazione finora
- Heat Exchanger - Effectiveness - NTU MethodDocumento15 pagineHeat Exchanger - Effectiveness - NTU MethodhendiyrNessuna valutazione finora
- Cooling and Heating Load Calculations - Heat Transfer Through Buildings - Fabric Heat Gain/LossDocumento28 pagineCooling and Heating Load Calculations - Heat Transfer Through Buildings - Fabric Heat Gain/LosscaptainhassNessuna valutazione finora
- Turbines, pumps and compressors explainedDocumento5 pagineTurbines, pumps and compressors explainedJuan HeroNessuna valutazione finora
- 125452033-Thermodynamics D.J.Dunn PDFDocumento185 pagine125452033-Thermodynamics D.J.Dunn PDFkalite123Nessuna valutazione finora
- ThermodynamicsDocumento10 pagineThermodynamicsمحمد لقمانNessuna valutazione finora
- Chapter 12 - ThermodynamicsDocumento69 pagineChapter 12 - Thermodynamicszahid_polyNessuna valutazione finora
- Chapter 11 Refrigeration CyclesDocumento20 pagineChapter 11 Refrigeration Cycleskalite123Nessuna valutazione finora
- Refrigeration Test RigDocumento7 pagineRefrigeration Test Rigshibin874141100% (1)
- Ch. 2Documento11 pagineCh. 2مصطفى العباديNessuna valutazione finora
- Centrifugalcompressorpower Si UnitsDocumento4 pagineCentrifugalcompressorpower Si UnitsJoshi DhvanitNessuna valutazione finora
- Solution Manual-Munson 4th - MergedDocumento10 pagineSolution Manual-Munson 4th - MergedRafay SaeedNessuna valutazione finora
- GT ThermodynamicsDocumento22 pagineGT Thermodynamicsforzama100% (1)
- Me2202 Engineering Thermodynamics - Uq - Nov Dec 2010Documento4 pagineMe2202 Engineering Thermodynamics - Uq - Nov Dec 2010BIBIN CHIDAMBARANATHANNessuna valutazione finora
- COMPRESSION RATIO GUIDEDocumento14 pagineCOMPRESSION RATIO GUIDEDirkMyburghNessuna valutazione finora
- Heat Exchanger Tutorial No 2 PDFDocumento2 pagineHeat Exchanger Tutorial No 2 PDFNitin BuZzNessuna valutazione finora
- Steam Table SI Units PDFDocumento256 pagineSteam Table SI Units PDFAlex SeguidaNessuna valutazione finora
- Thermodynamics Multiple Choice ReviewDocumento26 pagineThermodynamics Multiple Choice ReviewWillina Marie Chong MableNessuna valutazione finora
- Atk2 1 2014Documento34 pagineAtk2 1 2014Zakiyah Kamto IrfinNessuna valutazione finora
- Tutorial - 6 - EntropyDocumento7 pagineTutorial - 6 - EntropyanotherdeobiNessuna valutazione finora
- HS Diagram PDFDocumento1 paginaHS Diagram PDFmars1Nessuna valutazione finora
- BoilBal SpreadsheetDocumento16 pagineBoilBal SpreadsheetZulkarnain NasrullahNessuna valutazione finora
- Lecture 7 - Combustion of Solid FuelsDocumento11 pagineLecture 7 - Combustion of Solid FuelsShailani HossainNessuna valutazione finora
- Stoichiometric CombustionDocumento3 pagineStoichiometric CombustionKhairil MunawirNessuna valutazione finora
- Thermodynamics Homework 3 Efficiencies and Flow RatesDocumento5 pagineThermodynamics Homework 3 Efficiencies and Flow Ratescalebgriffin31Nessuna valutazione finora
- HEAT ENGINES E-LEARNING LECTURE #5 MODIFICATIONS ON BRAYTON CYCLEDocumento42 pagineHEAT ENGINES E-LEARNING LECTURE #5 MODIFICATIONS ON BRAYTON CYCLEMohamed KhaledNessuna valutazione finora
- Piston PumpDocumento7 paginePiston Pumpmohanadymt100% (1)
- Steam Table PDFDocumento16 pagineSteam Table PDFMustafa MohamedNessuna valutazione finora
- Heat Exchanger ReportDocumento8 pagineHeat Exchanger Reportarslan shahidNessuna valutazione finora
- Void Fractions in Two-Phase FlowsDocumento0 pagineVoid Fractions in Two-Phase FlowsMohsen AghaeiNessuna valutazione finora
- 8-2. Heat Ex ChangersDocumento6 pagine8-2. Heat Ex ChangersBambang HermawanNessuna valutazione finora
- Lecture No.3 The Ideal Reheat Rankine CycleDocumento12 pagineLecture No.3 The Ideal Reheat Rankine Cycledanielalejandro133Nessuna valutazione finora
- Assignment Answer Scheme PDFDocumento17 pagineAssignment Answer Scheme PDFHizami Mohammad Noor100% (2)
- All The Lecture Notes of ME56Documento31 pagineAll The Lecture Notes of ME56Kent NabzNessuna valutazione finora
- Tutorial Sheet 2Documento4 pagineTutorial Sheet 2oginoweijNessuna valutazione finora
- Chapter 4 External ConvectionDocumento68 pagineChapter 4 External Convectionpvr2k1Nessuna valutazione finora
- PV RT: Equations of StateDocumento11 paginePV RT: Equations of StateJeff HardyNessuna valutazione finora
- Practical Chemical Thermodynamics for GeoscientistsDa EverandPractical Chemical Thermodynamics for GeoscientistsNessuna valutazione finora
- High-Pressure Fluid Phase Equilibria: Phenomenology and ComputationDa EverandHigh-Pressure Fluid Phase Equilibria: Phenomenology and ComputationNessuna valutazione finora
- Thermal engineering A Clear and Concise ReferenceDa EverandThermal engineering A Clear and Concise ReferenceNessuna valutazione finora
- High Pressure Phase Behaviour of Multicomponent Fluid MixturesDa EverandHigh Pressure Phase Behaviour of Multicomponent Fluid MixturesNessuna valutazione finora
- Experimental Thermodynamics: Experimental Thermodynamics of Non-Reacting FluidsDa EverandExperimental Thermodynamics: Experimental Thermodynamics of Non-Reacting FluidsB. Le NeindreNessuna valutazione finora
- Extra Sol8 PDFDocumento8 pagineExtra Sol8 PDFJimmy JohnsNessuna valutazione finora
- Electrical PropertiesDocumento26 pagineElectrical PropertiesChrister John UyNessuna valutazione finora
- CrystallizationDocumento60 pagineCrystallizationChrister John UyNessuna valutazione finora
- Calibration of Acid BuretteDocumento7 pagineCalibration of Acid BuretteChrister John Uy100% (2)
- Principles of Engineering EconomyDocumento1 paginaPrinciples of Engineering EconomyChrister John UyNessuna valutazione finora
- Abstract UyDocumento1 paginaAbstract UyChrister John UyNessuna valutazione finora
- CrystallizationDocumento60 pagineCrystallizationChrister John UyNessuna valutazione finora
- PolymersDocumento32 paginePolymersChrister John UyNessuna valutazione finora
- Introduction To Materials ScienceDocumento38 pagineIntroduction To Materials ScienceChrister John UyNessuna valutazione finora
- CeramicsDocumento32 pagineCeramicsChrister John UyNessuna valutazione finora
- Gaseous FuelDocumento12 pagineGaseous FuelChrister John UyNessuna valutazione finora
- Diffusion in SolidDocumento47 pagineDiffusion in SolidChrister John UyNessuna valutazione finora
- LimitsDocumento17 pagineLimitsChrister John UyNessuna valutazione finora
- Atoms BrownDocumento10 pagineAtoms BrownChrister John UyNessuna valutazione finora
- Diffusion in Solids PDFDocumento47 pagineDiffusion in Solids PDFChrister John UyNessuna valutazione finora
- Gaseous FuelDocumento12 pagineGaseous FuelChrister John UyNessuna valutazione finora
- Heat ExchangersDocumento31 pagineHeat ExchangersChrister John UyNessuna valutazione finora
- Recrystallization & Melting Point Determination Purification of Crystalline Organic CompoundDocumento10 pagineRecrystallization & Melting Point Determination Purification of Crystalline Organic CompoundChrister John UyNessuna valutazione finora
- Second SOC of Guimaras Province (20181205) Smaller Opt PDFDocumento226 pagineSecond SOC of Guimaras Province (20181205) Smaller Opt PDFChrister John UyNessuna valutazione finora
- Ideal Solns, Colig PropsDocumento19 pagineIdeal Solns, Colig PropsChrister John Uy50% (2)
- Wetlands: Prepared By: Benedict J. Salazar Christer John M. UyDocumento30 pagineWetlands: Prepared By: Benedict J. Salazar Christer John M. UyChrister John UyNessuna valutazione finora
- Seawater Quality Assessment and Identification of Pollution Sources Along the Central Coastal Area of Gabes Gulf (SE Tunisia)_ Evidence of Industrial Impact and Implications for Marine Environment ProtectionDocumento8 pagineSeawater Quality Assessment and Identification of Pollution Sources Along the Central Coastal Area of Gabes Gulf (SE Tunisia)_ Evidence of Industrial Impact and Implications for Marine Environment ProtectionChrister John UyNessuna valutazione finora
- Analytical ChemistryDocumento1 paginaAnalytical ChemistryChrister John UyNessuna valutazione finora
- 2018 BSChE 2PSG Annex II - Sample Curriculum Map-FinalDocumento3 pagine2018 BSChE 2PSG Annex II - Sample Curriculum Map-FinalChrister John UyNessuna valutazione finora
- Op-Ed: Dolphin For Dinner? Poor Countries Increasing Dolphin Consumption Elizabeth Batt, January 8, 2012 SummaryDocumento3 pagineOp-Ed: Dolphin For Dinner? Poor Countries Increasing Dolphin Consumption Elizabeth Batt, January 8, 2012 SummaryChrister John UyNessuna valutazione finora
- D3300 SG (En) 02Documento144 pagineD3300 SG (En) 02Christer John UyNessuna valutazione finora
- Revised Withholding Tax TablesDocumento1 paginaRevised Withholding Tax TablesJonasAblangNessuna valutazione finora
- Reviewer 1Documento8 pagineReviewer 1Christer John UyNessuna valutazione finora
- Alumni Tracer Form (BTTHS)Documento2 pagineAlumni Tracer Form (BTTHS)Christer John UyNessuna valutazione finora
- Intro Propulsion Lect 7Documento30 pagineIntro Propulsion Lect 7Arun YogaNessuna valutazione finora
- Design of Wall Girt: Input DataDocumento3 pagineDesign of Wall Girt: Input DataAnonymous V6up3d0QGNessuna valutazione finora
- Understanding Loss Modulus and Tan DeltaDocumento7 pagineUnderstanding Loss Modulus and Tan DeltaJohn BenedictNessuna valutazione finora
- Elementary Statics and DynamicsDocumento15 pagineElementary Statics and DynamicsPooja Bk0% (1)
- Chapter 2 Basics For A Single-Degree-of-Freedom RotorDocumento29 pagineChapter 2 Basics For A Single-Degree-of-Freedom RotorMohammed SaeedNessuna valutazione finora
- Electric Charges & Fields: Chapter-1 class-XIIDocumento20 pagineElectric Charges & Fields: Chapter-1 class-XIIMohit SahuNessuna valutazione finora
- Fundamentals of Metal WorkingDocumento59 pagineFundamentals of Metal WorkingRaghu SNessuna valutazione finora
- Forms of EnergyDocumento15 pagineForms of EnergyTrajce StojanovNessuna valutazione finora
- Katrine PHD ThesisDocumento207 pagineKatrine PHD ThesiszewhiteNessuna valutazione finora
- Numerical Analysis of Composite Steel-Concrete Columns of Arbitrary Cross SectionDocumento10 pagineNumerical Analysis of Composite Steel-Concrete Columns of Arbitrary Cross SectionNasar KhanNessuna valutazione finora
- Assignment 3 2022Documento2 pagineAssignment 3 2022DICKSON SIMUTAMINessuna valutazione finora
- Formulacion de Capacidad PortanteDocumento11 pagineFormulacion de Capacidad PortanteJhon KenyiNessuna valutazione finora
- Revision Plan-II (DPP # 2) - Physics - EnglishDocumento12 pagineRevision Plan-II (DPP # 2) - Physics - EnglishBhriguKansra50% (2)
- Motores FullingmotorDocumento50 pagineMotores Fullingmotorservitecdj100% (1)
- Physics II Problems PDFDocumento1 paginaPhysics II Problems PDFBOSS BOSSNessuna valutazione finora
- (Asce) GT 1943-5606 0000921Documento11 pagine(Asce) GT 1943-5606 0000921gayathry100% (1)
- Grout Pressures Around A Tunnel Lining Influence oDocumento9 pagineGrout Pressures Around A Tunnel Lining Influence oTanumayaMitraNessuna valutazione finora
- Bosch ESP Systems 5 Years of Experience 2000-01-1633Documento11 pagineBosch ESP Systems 5 Years of Experience 2000-01-1633Syed GibranNessuna valutazione finora
- Aircraft Stability and ControlDocumento2 pagineAircraft Stability and ControlSaravanan AtthiappanNessuna valutazione finora
- Electric field of a uniformly charged diskDocumento7 pagineElectric field of a uniformly charged diskJay ZillaNessuna valutazione finora
- P P/ V/ X X / L P F F - F: 30.4 Decay of Turbulence Downstream From A Stilling BasinDocumento2 pagineP P/ V/ X X / L P F F - F: 30.4 Decay of Turbulence Downstream From A Stilling Basinamanj2009Nessuna valutazione finora
- Gunpowder RocketsDocumento4 pagineGunpowder RocketsMartin EcheverriaNessuna valutazione finora
- PT Specialist training friction loss calculationDocumento28 paginePT Specialist training friction loss calculationVladimir SimeunovicNessuna valutazione finora
- Chapter Ii - Fluid StaticsDocumento72 pagineChapter Ii - Fluid StaticszetseatberhanuNessuna valutazione finora
- Practical Approximate Analysis of Beams and FramesDocumento4 paginePractical Approximate Analysis of Beams and Framessami_bangash_1100% (1)
- FSAE Aero Initial Performance Predictions - MonashDocumento10 pagineFSAE Aero Initial Performance Predictions - MonashPedro BarataNessuna valutazione finora
- MD Prelim Compiles QuestionsDocumento2 pagineMD Prelim Compiles Questionsrjmanahan1223Nessuna valutazione finora
- Class 11 CRPDocumento2 pagineClass 11 CRPprayag527Nessuna valutazione finora
- Article Vilebrequin F8L413Documento12 pagineArticle Vilebrequin F8L413Maarten BaanNessuna valutazione finora