Documenti di Didattica
Documenti di Professioni
Documenti di Cultura
Ragland (1991) Properties of Wood For Combustion Analysis
Caricato da
Juan Jose SossaTitolo originale
Copyright
Formati disponibili
Condividi questo documento
Condividi o incorpora il documento
Hai trovato utile questo documento?
Questo contenuto è inappropriato?
Segnala questo documentoCopyright:
Formati disponibili
Ragland (1991) Properties of Wood For Combustion Analysis
Caricato da
Juan Jose SossaCopyright:
Formati disponibili
Bioresource Technology 37 ( 1991 ) 161 - 168
Properties of Wood for Combustion Analysis
K. W. Ragland, D. J. Aerts
Department of Mechanical Engineering, University of Wisconsin-Madison, Madison, Wisconsin 53706, USA
&
A. J. Baker
USDA Forest Service, Forest Products Laboratory, Madison, Wisconsin 53705, USA*
(Received 4 June 1990; revised version received 1 October 1990; accepted 2 October 1990)
Abstract
A systematic compilation of 21 different property
values of wood and bark fuel was made to facilitate
the engineering analysis and modeling of combustion systems. Physical property values vary greatly
and properties such as density, porosity, and internal surface area are related to wood species
whereas bulk density, particle size, and shape distribution are related to fuel preparation methods.
Density of dry wood and bark varies from 300 to
550 kg m-.~; bulk density of prepared wood fuel
varies from about 160 to 230 kg m -3. Thermal
property values such as specific heat, thermal conductivity, and emissivity vary with moisture
content, temperature, and degree of thermal
degradation by one order of magnitude. The
carbon content of wood varies from about 47 to
_53% due to varying lignin and extractives content,
Mineral content of wood is less than 1%, but it can
be over 10 times that value in bark. The composition of mineral matter can vary between and within
each tree. Properties that need further investigation
are the temperature dependency of thermal conductivity and the thermal emissivity of char-ash.
How mineral matter is transformed into various
sizes of particulates is not well understood.
Key words: Wood, combustion, fuel properties,
modeling.
NOMENCLATURE
Ac
Pre-exponential factor for char
Aco
Pre-exponential factor for CO
*The Forest Products Laboratory is maintained in cooperation with the Universityof Wisconsin.
A Hc
Ao
Pre-exponential factor for hydrocarbons
Pre-exponential factor for pyrolysis
(h-')
A,
c
D
Ec
Eco
EHc
Ep
Gg
ho
k
kr
M
M'
p
R
Re
RHc
Rc
Rco
Rp
S
Sc
Surface area per unit volume (cm- 1)
Specific heat (J kg- i K - 1)
Diffusivity of oxygen (m 2 s - 1)
Activation energy for char (cal mol- ~)
Activation energy for CO (cal mol-J)
Activation energy for hydrocarbons (cal
mol- l)
Activation energy for pyrolysis (cal
mol- l)
Mass flux of gas (kg m -2 h -1)
Mass transfer coefficient (kg s-1 m - 2 )
Thermal conductivity (W m - ~K- 1)
Rate constant
Moisture mass fraction
Molecular weight (kg kgmol- 1)
Pressure (atm)
Universal gas constant (cal mol- 1 K- 1)
Reynolds number
Hydrocarbon reaction rate (kgrnol m-3
Char reaction rate (kg s- 1)
CO reaction rate (kgmol m-3)
Pyrolysis rate (kg s- 1)
Specific gravity
Schmidt number
Solid temperature (K)
Pg
P~
P
Void fraction
Char density (kg m - 3)
Density of gas phase (kg m-3)
Solid density (kg m - 3)
Viscosity (N s m - : )
[ ]
Species concentration (kgmol m-3)
6
Pc
161
Bioresource Technolog3' 0960-8524/91/S03.50 1991 Elsevier Science Publishers Ltd, England. Printed m
Great Britain
162
K. W. Ragland, D. J. Aerts, A. J. Baker
INTRODUCTION
Analysis and modeling of combustion in stoves,
furnaces, boilers and industrial processes require
adequate knowledge of wood properties. Detailed
computer modeling of combustion processes
requires accurate property values, and these properties are not generally available in one reference
source. The objectives of this paper are to review
existing property data on wood which are needed
for the analysis of combustion systems and to suggest properties that need further quantification.
Fuel properties for combustion analysis of
wood can be conveniently grouped into physical,
thermal, chemical, and mineral properties. Bark
properties should be distinguished from wood
properties. Thermal degradation products of
wood consist of moisture, volatiles, char and ash.
Volatiles are further subdivided into gases and
tars. Some properties vary with species, location
within the tree, and growth conditions. Other properties depend on the combustion environment.
Where the properties are highly variable, the
likely range of the property is given. Hence, this
paper is a general review of properties rather than
a tabulation of specific data.
Combustion systems using wood fuel may be
generally grouped into fixed bed, suspension
burning, and fluidized bed systems. The systems
range from residential to commercial and industrial to utility scale. The fuel property data
needed depend, of course, on the type of application and the detail of the model. In general, combustion models can be classified as macroscopic
or microscopic. Wood fuel properties for macroscopic analysis, such as ultimate analysis, heating
value, moisture content, particle size, bulk density,
and ash fusion temperature, have recently been
reviewed (Bushnell et al., 1989). Properties for
microscopic analysis include thermal, chemical
kinetic, and mineral data, and these properties
have not been collected in one source for wood.
Although there is a risk of oversimplifying complex processes, the authors hope that the information provided will assist the combustion modeler
in the difficult job of assembling property data.
The paper is not a comprehensive review of all
available wood fuel property data, but rather gives
a data set sufficient for modeling purposes.
PHYSICAL PROPERTIES
Physical properties for analysis of combustion
systems include density, bulk density, particle size,
internal and external surface area per unit volume,
porosity, and color.
The density of commercially important wood
species in the United States ranges from 300 to
550 kg m-3 on an ovendry basis (Forest Products
Laboratory, 1987). The range in density of bark is
similar (Koch, 1972). Tropical woods vary more
widely, with a dry density as high as 1040 kg m-3
For a particular species, the variation in dry
density is not more than about 10%. The density
of the wood cell wall is 1450 to 1550 kg m -3
(Weatherwax & Tarkow, 1968). With increasing
moisture content, both the density and volume
increase up to the fiber saturation point of about
30% moisture. Above this point, only the density
increases (Forest Products Laboratory, 1987).
Fresh, green wood has a moisture content of
35-60% on a wet basis. Dried wood used for fuel
typically has a moisture content of 5-20%.
The particle size of wood fuels ranges from
whole trees to sander dust and includes processed
and pelletized fuels. Dry bulk density, which is
defined as mass of ovendry wood per unit of
green bulk volume, ranges from 157 kg m -3 for
hardwood shavings to 227 kg m -3 for hardwood
sawdust (Table 1) (Forest Products Laboratory,
1987). Approximately 80% of the sawdust is
smaller than 2.5 mm and 80% of the pulp chips
and hog fuel are less than 25 mm and 30 mm,
respectively, based on the particle width (Fig. 1)
(Seppanen, 1988). Of course, the size distributions vary with the equipment used to make the
chips. Representative shape factors (length to
thickness ratio) for sawdust, pulp chips, and hog
fuel, are 3.5, 5.8 and 35, respectively (Seppanen,
1988).
The porosity and internal surface area of wood
char are needed for detailed modeling of pulverized wood combustion. Upon completion of pyrolysis, the porosity of char is 0.8-0.9 for wood
heated under combustion conditions. The initial
char density is 10-20% that of the dry wood,
Table !. Representative dry bulk density of typical wood
fuels (Forest Products Laboratory, 1987)
Fuel type
Bulk density (kg m-t)
Hardwood sawdust and bark
Mixed pine-hardwood sawdust
Clean hardwood pulp chips
Hardwood whole-tree chips
Pine whole-tree chips
Hogged dry trims (hardwood)
Shavings(hardwood)
227
219
210
211
181
221
157
Properties of wood for combusilon analysis
',otI
t
//"
//
tI
16 3
wood-water bonds and can be represented by a
correction term (TenWolde et aL, 1988)
/ .f
c(wet)=[c(dry)+ 4.19M]/(1 + M)+ A
(kJ kg-' K - ' )
(2)
where
ti
//
,011
"]l
0
tJ
oJ!
/~
/i
/i
10
....
A =(0-02355 T - 1.32M-6.191)M,
T= temperature in K, and
M = fractional moisture content, dry basis.
Sawdust
Pulp Chips
---
Hog Fue,
20
30
40
Screen Opening (mm)
50
Fig. 1. Cumulative
size distribution
unscreened pulp chips and hog fuel.
for
60
sawdust,
depending on the heating rate. As the char reacts,
the density decreases further. The internal surface
area of wood char (Wark & Warner, 1981) is of
the order of 106 m 2 kg-1. The pore size distribution of wood char (Baileys & Blankenhorn, 1982)
ranges from 0.01 to 30 Mm. For packed bed combustion of pulp chips, the void fraction and
external surface area per unit total volume are
used in some models. For pulp chips, the void
fraction is 0.5-0.6, and the surface area per unit
total volume (Ragland, K. W., Aerts, D. J. & Baker,
A. J., unpublished) is about 1.5 cm- i.
When heated, wood changes color. At 250C,
wood is dark brown; above 300C, wood is black.
The brown color is associated with the onset of
pyrolysis. Above 300C, shrinkage also begins
(Baileys & Blankenhorn, 1982).
THERMAL PROPERTIES
Important thermal properties in the analysis of
wood combustion include the specific heat of
wood and char, the thermal conductivity of wood
and char, and the emissivity of char. Specific heat
depends on temperature and moisture content but
not on density or species. The specific heat of dry
wood is given by (TenWolde et al., 1988)
c(dry)= 0"1031 + 0.003867 T
Above the fiber saturation point, A = 0. Specific
heat can vary by as much as 100% depending on
the temperature and moisture content (Table 2).
The specific heat of wood char can reasonably
be assumed to be the same as that of graphite. The
specific heat of graphite varies from 0.715 kJ kgK-~ at 3 0 0 K t o 2 - 0 4 k J k g - ~ K-~ at 2000K. A
curve fit of the data from 700 to 2000 K, which
holds to within 5%, is given by (Stull, 1971)
c(char) = 1.39 + 0-00036T
The thermal conductivity of wood increases
with density, moisture content, and temperature.
Also, the thermal conductivity is approximately
1.8 times greater parallel to the grain than in
either the radial or tangential directions. For a
wide variety of wood species at room temperature, the average thermal conductivity perpendicular to the grain (TenWolde et aL, 1988)is given
by
k = S(0.1941 + 0.4064M) + 0"01864
( W m - ' K -~)
The specific heat of wet wood is greater than
would be expected from the simple law of mixtures as a result of the energy absorbed by the
(4)
where S is the specific gravity based on volume at
the current moisture content and weight when
ovendry. For example, if S--0-47 and M--0.10,
k--0.129 W m-1 K-~. The thermal conductivity
of wood increases with temperature at the rate of
approximately 0.2% per C above room temperature. Hence, thermal conductivity increases 10%
for every 50 K increase in temperature. The tern-
Table 2. Specific heat of wood for selected temperatures
and moisture contents
Specific heat (kJ kg- 1K- i)
Temperature
(K)
(kJkg -l K -1)
(1)
(kJ kg -~ K -~) (3)
280
300
320
340
360
c(dry)
c(5%)
1"2
1'3
1'3
1"4
1"5
1"3
1'4
1'5
1'6
1'7
c(12%)
1'5
1"7
1'8
1"9
2"0
c(20%)
1"7
1"9
2-0
2'2
2'3
164
K. W. Ragland, D. J. Aerts, A. J. B a k e r
perature correction is based on limited data, however and should be used with caution.
The thermal conductivity of char is reported to
be 0.052 W m-t K-I at room temperature for
maple, beech, and birch char (Wheast, 1976).
Intuitively, the thermal conductivity of char
depends on density, which depends on the heating
rate. Evans and Emmons (1977) report that the
thermal conductivity is given by
k-- 0"67S- 0.071
(Wm -L K -~)
(5)
where S is the specific gravity as noted above.
Obviously, this relation fails at S<0.106. The
variation of char conductivity with temperature is
not available.
The thermal radiation emissivity of carbon is
0"80 and is independent of temperature (Anon.,
1972a). Actual wood char may have a lower emissivity resulting from the ash layer on the surface of
the char. The character of the ash layer depends
on temperature, flow velocity, and oxygen concentration; therefore, char emissivity is difficult to
assess. Refractory oxides typically have an emissivity of about 0.4 at 1300 K, for example, and
wood ash emissivity may be similar. A char emissivity of 0"75 has been used for modeling
purposes (Evans & Emmons, 1977).
CHEMICAL PROPERTIES
Important chemical properties for combustion are
the ultimate analysis, proximate analysis, analysis
of pyrolysis products, overall heating value, heat
of pyrolysis, heating value of the volatiles, and
heating value of the char.
The ultimate analysis (Table 3) provides weight
percentage of C, H, O, N, and S. The C content of
softwood species is 50-53%, and that of hardwood species 47-50%, due to the varying lignin
and extractives content. All wood species contain
about 6% H (Petura, 1979). Oxygen content
Table 3. Ultimate analysis of wood (dry, ash-free weight per
cent)
Element
C
H
O
N
S
Average of I I
hardwoods ~
A verage of 9
softwoods"
Oak
bark h
Pine
bark b
50"2
6.2
43.5
0" 1
--
52.7
6.3
40.8
0"2
0"0
52.6
5.7
41"5
0" 1
0" 1
54.9
5"8
39.0
0"2
(1"1
"Tillman et al. ( 1981 ).
1'Anon. (1972b).
ranges from 40% to 44%, S is less than 0.1% and
N ranges from 0"1-0.2%.
Proximate analysis gives the weight fraction of
moisture, and volatiles including tar, char, and
ash, according to ASTM standard test method
E870-82. The ground wood sample is heated in
air to 103C to constant weight to obtain dry
wood, to 950C in a capped crucible to obtain
char, and to 600C in air to obtain ash. The
volatile yield can be increased by increasing the
heating rate (i.e. increasing the heat transfer rate
or increasing the exposed surface area). Conversely, the char yield is increased by slow heating
of large particles. The standard procedure for
proximate analysis is more representative of combustion on a grate than in suspension. The ASTM
volatile yield for dry ash-free wood ranges from
70% to 90%, depending on the species. The
ASTM volatile yield for bark is typically closer to
70%.
The volatile yield of dry wood under combustion conditions may be further subdivided into
light hydrocarbons, tar, carbon monoxide, carbon
dioxide, hydrogen, and moisture yield (Table 4).
The yields depend on the temperature and heating rate of pyrolysis. The tar has an average composition (Adams, 1980) of C6H6.200.2, and the
light hydrocarbons are primarily methane.
The higher heating value of different wood
species on a moisture-free basis varies less than
15%. The higher heating value of softwoods
(Baker, 1989) is 20-22 MJ kg -n and of hardwoods, 19-21 MJ kg- t. The average higher heating value of seven Populus hybrids (Bowersox et
al., 1979) is 19.49 MJ kg-t. Typically, softwoods
have more extractives and more lignin than do
hardwoods, which accounts for the slightly higher
heating value.
The heating value of the volatiles may be
estimated by the following method. Pyrolysis is
Table 4. Distribution of pyrolysis products
for dry wood under combustion conditions ~
Product
Mass fraction
H20
CO
CO,
H,
Light hydrocarbons
Tar
Char
0"25
0"183
0"115
0"005
0.047
0.20
0.20
"Bowersox et aL (1979) and Forests Products
Laboratory (1987).
Properties of wood for combustion analysis
initially slightly endothermic and then exothermic,
so that overall pyrolysis is considered essentially
thermally neutral (Roberts, 1971 ). Using the data
in Table 4 and assuming that char is all carbon
with a higher heating value of 32.8 MJ kg-z, an
average higher heating value of the volatiles is
calculated to be 16.2 MJ kg-1. Subtracting the
heating value of CO and H2, the light hydrocarbons and tars have a higher heating value of
41-0 MJ kg-i.
Above 700C char contains little hydrogen.
Stimely and Blankenhorn (1985) reported the
hydrogen content of char as a function of temperature for four woods. For example, for hybrid
poplar, the percentage of hydrogen by weight is
H2= 7"39-0"01T
(6)
where T is the final carbonization temperature
(C). Hence for temperatures above 739C, which
is typically the case for most combustion applications, char does not contain hydrogen.
REACTION RATES
To analyze combustion systems, it is often useful
to model the rate at which drying, pyrolysis, and
char burn take place. Drying rates are outside the
scope of this paper. During pyrolysis the com-
165
ponents of wood, such as cellulose, hemicellulose,
and lignin, pyrolyze at different rates; however,
for combustion analysis, a single global rate
expression of the Arrhenius form often gives
acceptable results (Table 5). Roberts (1970)
reviewed the literature on the pyrolysis of wood
and suggested that an activation energy of 31 kcal
mol-t is appropriate for both small and large
particles (Table 6); a conclusion that was supported by Belleville (1984). The suggested preexponential constant (Roberts, 1970; Belleville,
1984) in the Arrhenius equation is 7 x 107 S-k. As
noted previously, the pyrolysis gases contain
hydrogen, carbon monoxide, light hydrocarbons
and tars. The hydrogen can be considered to react
instantaneously at combustion temperatures. The
global oxidation rate of CO to CO 2 is well known
and depends on the concentration of water vapor
(Table 5). Global kinetic reaction rates for hydrocarbon volatiles from wood combustion are not
available. Smoot and Smith (1985) have reviewed
the kinetics for coal volatiles, and their results
indicated that long-chain and cyclic hydrocarbons
react to form hydrogen and carbon monoxide
according to the following global reaction:
C n H m + 0 " 5 / / 0 2 --- 0"5mH 2 +
Light hydrocarbons and tars are lumped together
in this approach.
Table 5. Reaction rate equations
Rate equation
Reference
Ep/RTs)
~ ' M "~/
M " s) kr + 2hD)/((Mg/Ms)
"
'
' kr+2hD)
"
Rc=~~A,.Po:[
RHc = A HCTgp'3[HC]'5[O2] exp( - EHc/RTg )
Rco = Aco[CO] [O_,]25[H20]'5 exp( - Eco/RTg )
where
k~= Acexp(- Ec/RT~)
/~O= 1"46 PN: M'gG g S c - 273Re 0.41( 1 - 6 )0-2
Re=Gg/(Av(1-6)t~);
Sc~-~/p~
Rp = Ap(p~ - Pc) exp( -
Roberts (1970)
Smoot and Smith (1985)
Smoot and Smith (1985)
Smoot and Smith (1985)
Gamson ( 1951 )
Table 6. Reaction rate constant values for equations in Table 5
Parameter
Ap
Ep
Aco
Eco
A ,c
EHc
Ac
Ec
nCO
Value
2"52 x 10 ~l h - l
3-1 x 104 cal mol-~
4.0 x 105 (see reaction rate)
4.10 x 104 (cal mol-~)
2.07 x 104 (see reaction rate)
1.917 x 107 cal mol- 1
1.71 x 107 kg rn- -"h - ~
23.8 x 104 cal mol- ~
Reference
Roberts (1970)
Roberts (1970)
Smoot and Smith
Smoot and Smith
Smoot and Smith
Smoot and Smith
Bhagat (1980)
Bhagat (1980)
(1985)
(1985)
(1985)
( 1985 )
166
K. W. Ragland, D. J. Aerts, A. J. Baker
Char reacts with oxygen, carbon dioxide, water
and hydrogen. Char oxidation is the dominant
reaction in a combustion environment. Since this
is a surface reaction, the reaction rate depends on
kinetics and diffusion of oxygen to the char
surface (Tables 5 and 6). Minimal wood-char
kinetic data are available compared to coal-char
data, although this is important because char
burnout is much longer than volatile burnout. The
diffusion parameters depend on the gas conditions and are beyond the scope of this paper, as is
the use of intrinsic kinetic data instead of global
kinetics.
MINERAL PROPERTIES
The mineral content of clean wood of temperate
tree species is 0.1% to 0-6% and that of bark 3%
to 5%. Mineral matter in wood consists mostly of
salts of calcium, potassium, and magnesium, but
salts of many other elements are also present in
lesser amounts (Table 7). Some salts are formed
with the organic acid groups of the cell wall components, whereas others occur as carbonates,
phosphates, sulfates, silicates and oxalates. The
mineral content of wood and bark is highly
variable between and within species and can vary
with soil and growth rate. Whole-tree analyses of
minerals and trace elements in aspen, pine bark,
and oak bark (Table 8) are different from that of
pure wood. Silica is rarely present in more than
trace amounts in the wood of temperate tree
species (Pettersen, 1984). The silica content of
clean bark is higher than that of wood. Analysis of
bark from 18 softwood species and 24 hardwood
species indicated an average silica content of
Table
7.
Mineral analysis of wood
Element
Mineral content (ppm)
Yellow Poplar"
White Oak"
Loblolly Pine b
1182
517
265
258
108
48
35
14
3
8
1976
1 128
294
216
75
59
70
32
2
62
1141
116
326
230
40
121
72
---
Ca
K
Mg
Na
P
Mn
Fe
AI
Cu
Zn
" K o c h (1985).
h K o c h (1972).
--
Table 8. M i n e r a l a n a l y s i s of w h o l e tree a n d b a r k
Element
Ca
K
Mg
P
S
Fe
Zn
Mn
AI
B
Cu
Na
Pb
Cd
Si
Mineral cotltent (ppm)
Aspen
(whole tree)"
Pine bark ~
Oak bark'
4 160
1424
493
323
107
30
36
17
10
6
13
17
1'4
0'30
--
5 282
1 333
1 137
--609
--2 149
--280
--5 278
24418
88
3 816
--1 224
--29
--3 500
--2 745
Total ash: "-- 1.45%; h _- 2.9%; ' - - 5-3%.
0.05% and 0.11%, respectively (Harder &
Einspahr, 1980). The silca content of bark, as
used for fuel, however, is higher because of soil
contamination by wind and harvesting methods.
Ash is formed from mineral matter during
combustion and gasification. The ash yield of
wood grown in the temperate zones is 0.1-1.0%,
whereas wood grown in the tropics contains up to
5% ash (Fengel & Wegener, 1984). Bark contains
3-8% ash. Wood ash typically includes 40-70%
calcium oxide and 10-30% potassium oxide.
During combustion, the mineral ions oxidize
and volatilize or form particulates. The char
surface is hotter than the gas or the interior of the
particle, and ash particles tend to form on the char
surface. As the char burns away, the ash particles
are released. Knowledge of mineral matter
behavior is needed for analysis of ash deposition,
erosion and corrosion in combustion systems, as
well as for understanding stack gas opacity.
The ash-fusion temperature, which is measured
in the laboratory by ASTM method D 1857, is
used as an indicator of deposition tendency in
actual combustion systems (Table 9). The ashfusion temperature is determined by mixing the
ash sample from proximate analysis with a solution of dextrin to form a stiff paste that is pressed
into small cone-shaped molds. The oven temperature at which the cone first deforms is reported as
the onset of ash fusion. In addition to the initial
deformation temperature, the softening temperature, hemispherical temperature, and fluid temperature are reported. Both oxidizing (air) and
167
Properties of wood for combustion ana(vsis
Table 9. Ash-fusion temperatures for wood and bark
Source of ash
Wood "
Initial deformation
Fluid
Oak bark ~'
Initial deformation
Softening
Fluid
Pine bark ~'
Initial deformation
Softening
Fluid
Reducing temperature
Oxidizing temperature
1 450-1 5 1 5
1 500-1 550
--
(~)
(%)
--
1 477
1 493
1 504
1 471
1 499
1 51(1
1 193
1 226
1 266
1 210
1 249
1 288
"Baumeister et al. (1978).
bAnon.(1972b).
reducing (60% CO, 40% CO2) atmospheres are
used. Wood has a higher ash-fusion temperature
than do many coals (Anon., 1972b) and hence can
sometimes be less prone to ash deposition than
coal. However, detailed testing is required to
investigate fouling, slagging and agglomeration.
C O N C L U D I N G REMARKS
Property values for the analysis of combustion of
wood fuels have been collected and are reviewed
in this paper. Selected properties are given for
unheated wood and bark and for the thermal
degradation products of volatiles, char and ash. In
some geographical locations, bark is the main
wood fuel; generally, less basic data on bark are
available when compared to available data on
wood.
Physical properties of density, size, and surface
area per unit volume are presented for various
types of wood fuels. The thermal properties of
specific heat and thermal conductivity of wood
and char are well known; however, the temperature dependency of the char thermal conductivity
is not known. The thermal emissivity of char-ash
is not well known. Overall heating values and elemental analyses are readily available. Pyrolysis
product yields are highly dependent on heating
conditions. Depending on the actual conditions,
the standard proximate analysis may or may not
be applicable. Suggestions are made above for
approximating the volatile reaction rates, but
further investigation in this area is needed. Global
char reaction rates also need further observation.
Mineral matter in wood is well characterized.
Little is known, however, on how the mineral
matter is transformed into paniculate ash of
various sizes.
REFERENCES
Anon. (1972a). Thermophysical Properties of Matter, vol. 8."
Thermal Radiative Properties of Non-metallic Solids.
Plenum Press, New York.
Anon. (1972b). Steam~Its Generation and Use. The Babcock
and Wilcox Co., New York, pp. 5-23.
Adams, T. N. (1980). A simple fuel bed model for predicting
paniculate emissions from a wood-waste boiler. Combustion and Flame, 39,225-39.
Baileys, R. T. & Biankenhorn, P. R. (1982). Calorific and
porosity development in carbonized wood. Wood Science,
15, 19-28.
Baker, A. J. (1989). In Mark-Bikales-Overberger-Menges
Encyclopedia of Polymer Science and Engineering, vol. 17,
2nd edn, Wiley, pp. 843-87.
Baumeister, T. (ed.) (1978). Mark's Standard Handbook for
Mechanical Engineers, 8th edn, McGraw-Hill, New York,
pp. 7-13.
Belleville. P. (1984)/ Pyrolysis of large wood samples.
Applied Energy, 16,223-37.
Bhagat, P. M. (1980). Wood charcoal combustion. Combustion and Flame, 37,275-91.
Bowersox, T. W., Blankenhorn, P. R. & Murphey, W. K.
(1979). Heat of combustion, ash content, nutrient content
and chemical content of Populus hybrids. Wood Science,
11,257-62.
Bushnell, D. J., Haluzok, C. & Dadkhah-Nikoo, A. (1989).
Biomass fuel characterization: testing and evaluating the
combustion characteristics of selected biomass fuels.
Bonneville Power Administration, Corvallis, OR.
Corder, S. E. & Athenon, G. H. (1971 ). Improved performance of wigwam burners. Forest Prod. J., 21 (9), 45.
Evans, D. D. & Emmons. H. W. (1977). Combustion of wood
charcoal. Fire Research, 1, 57-66.
Fengel, D. & Wegener, G. (1984). Wood: Chemistry, Ultrasmwture and Reactions. de Gruyter, Berlin, pp. 217-20.
Forest Products Laboratory (1987). Wood Handbook: Wood
as an Engineering Material Agric. Handbook 72(rev).
168
K. W. Ragland, D. J. Aerts, A. J. Baker
Washington, DC: US Department of Agriculture, pp.
4-15,3-12.
Gamson, B. W. (1951). Heat and mass transfer: fluid-solid
systems. Chemical Engin. Prog., 47, 19-28.
Harder, M. L. & Einspahr, D. W. (1980). Levels of some
essential metals in bark. TAPPI, 3, 110-2.
Koch, E (ed.) (1972). Utilization of the Southern Pines. Agric.
Handbook 420, vol. 1, Washington, DC: US Department
of Agriculture, pp. 501,220.
Koch, E (ed.)(1985). Utilization of Hardwoods Growing on
Southern Pine Sites. Agric. Handbook 605, vol. 1,
Washington, DC: US Department of'Agriculture, pp. 443,
445.
Pettersen, R. C. (1984). The chemical composition of wood.
In Chemistry of Solid Wood, ed. R. M. Rowe, Advances in
Chemistry Series 207, American Chemical Society,
Washington, DC, p. 74.
Petura, R. C. (1979). Thermodynamic Data for Waste Incineration. American Society of Mechanical Engineers,
New York, pp. 107-15.
Roberts, A. E (1970). A review of kinetics data for pyrolysis
of wood and related substances. Combustion and Flame,
14,261-72.
Roberts, A. E (1971). Heat of reaction during pyrolysis of
wood. Combustion and Flame, 17, 79-86.
Seppanen, V. P. (1988). Particle size of fuel chips made by
landing chippers. Technical Research Center of Finland,
Research Note 863, Espo, Finland.
Smoot, L. D. & Smith, P.J. (1985). Coal Combustion and
Gasification. Plenum Press, New York, p. 71.
Stimely, G. L. & Blankenhorn, P. R. (1985). Effects of species,
specimen size and heating rate on char yield and fuel properties. Wood and Fiber Science, 7,476-89.
Stull, D. R. (1971). JANAF thermochemical tables, NSRDSNBS 37, US Government Printing Office.
TenWolde, A., McNatt, J. D. & Krahn, L. (1988). Thermal
properties of wood and wood panel products for use in
buildings. DOE/USDA-21697/1, Oak Ridge National
Laboratory, Oak Ridge, TN.
Tillman, D. A., Rossi, A. J. & Kitto, W. D. (1981). Wood
Combustion: Principles, Processes and Economics.
Academic Press, Orlando, p. 43.
Wark, K. & Warner, C. F. ( 1981 ). Air Pollution: Its Origin and
Control. Harper & Row, New York, p. 258.
Weatherwax, R. C. & Tarkow, H. (1968). Density of wood
substance -- importance of penetration and adsorption
compression of the displacement fluid. Forest Products J.,
18 (7), 44-6.
Wheast, R. C. (1976). Handbook of Chemistry and Physics,
CRC Press, Cleveland, Ohio, p. E-5.
Potrebbero piacerti anche
- Model 4Documento12 pagineModel 4Oluwaseun AdetayoNessuna valutazione finora
- Thermal Properties of WoodDocumento11 pagineThermal Properties of Woodsnowari3szNessuna valutazione finora
- BioRes 13 1 0157 Cabalova KLVBCD Effect Thermal Treatm Improvement Properties Oak Wood 12780Documento14 pagineBioRes 13 1 0157 Cabalova KLVBCD Effect Thermal Treatm Improvement Properties Oak Wood 12780yonNessuna valutazione finora
- MB Numerical Modeling of A Deep Fixed Bed Combustor Kenneth Energy and Fuels 10-2-1996Documento8 pagineMB Numerical Modeling of A Deep Fixed Bed Combustor Kenneth Energy and Fuels 10-2-1996Mohammad Shahed TusharNessuna valutazione finora
- Elastic and Strength Properties of HeatDocumento18 pagineElastic and Strength Properties of Heatkulin_banNessuna valutazione finora
- Changes in Chemistry, Color, Dimensional Stability and Fungal Resistance of Pinus Radiata D. Don Wood With Oil Heat-TreatmentDocumento9 pagineChanges in Chemistry, Color, Dimensional Stability and Fungal Resistance of Pinus Radiata D. Don Wood With Oil Heat-TreatmentArjun AzisNessuna valutazione finora
- 5eures WoodPropertiesPDF PDFDocumento8 pagine5eures WoodPropertiesPDF PDFFahmi ImanuddinNessuna valutazione finora
- Torrefaction of Wood Part 2. Analysis of ProductsDocumento6 pagineTorrefaction of Wood Part 2. Analysis of Products김형진Nessuna valutazione finora
- Propiedades de Madera EncinoDocumento15 paginePropiedades de Madera EncinoArturo del BosqueNessuna valutazione finora
- Effects of Thermal Treatment On ChemicalDocumento6 pagineEffects of Thermal Treatment On ChemicalnurizzaatiNessuna valutazione finora
- Properties - of - Wood - and - Wood - Based - Materials SUBJECTED To THERMAL TREATMENTS UNDER Various TrethmansDocumento14 pagineProperties - of - Wood - and - Wood - Based - Materials SUBJECTED To THERMAL TREATMENTS UNDER Various TrethmansHasan Žera TalićNessuna valutazione finora
- 6441 28571 1 PBDocumento15 pagine6441 28571 1 PBOĞUZHAN YÜKSELNessuna valutazione finora
- BioRes 03-3-0789 Bodirlau TS ChemMod Beach ThermalStabDocumento12 pagineBioRes 03-3-0789 Bodirlau TS ChemMod Beach ThermalStabalin425Nessuna valutazione finora
- Processes 09 01598 v2Documento15 pagineProcesses 09 01598 v2Milos MMNessuna valutazione finora
- Bioresources.: The Effects of Natural Weathering On The Properties of Heat-Treated Alder WoodDocumento18 pagineBioresources.: The Effects of Natural Weathering On The Properties of Heat-Treated Alder WoodyonNessuna valutazione finora
- 10.1007@s00226 010 0361 3Documento13 pagine10.1007@s00226 010 0361 3ari goldNessuna valutazione finora
- TTTTDocumento4 pagineTTTTRuweydaNessuna valutazione finora
- Torrefaction of Wood - Par 2 PDFDocumento6 pagineTorrefaction of Wood - Par 2 PDFVictor Fassina BroccoNessuna valutazione finora
- 2F-1 Kinetic Modelling of Biomass Gasification and CombustionDocumento20 pagine2F-1 Kinetic Modelling of Biomass Gasification and Combustionizzet1969Nessuna valutazione finora
- Infrared Spectroscopy of The Surface of Thermally-Modified Teak Juvenile WoodDocumento10 pagineInfrared Spectroscopy of The Surface of Thermally-Modified Teak Juvenile Woodmargarita PetroNessuna valutazione finora
- Afe2015no66p137 144Documento9 pagineAfe2015no66p137 144Fernando SilvaNessuna valutazione finora
- Wood Ash. A New Source of Pozzolanic Material. Naik and KrausDocumento25 pagineWood Ash. A New Source of Pozzolanic Material. Naik and KrausMARCOS ANTONIO GOLATONessuna valutazione finora
- Guntekin and SahinDocumento9 pagineGuntekin and SahinYuri KomissarovNessuna valutazione finora
- Determination of Chemical Changes in Heat-Treated Wood Using ATR-FTIR and FT Raman SpectrometryDocumento6 pagineDetermination of Chemical Changes in Heat-Treated Wood Using ATR-FTIR and FT Raman SpectrometryKatherin Meza BenitezNessuna valutazione finora
- Numerical Modeling of A Deep, Fixed Bed Combustor: MW To 400 MW Rankine Cycle Power PlantsDocumento5 pagineNumerical Modeling of A Deep, Fixed Bed Combustor: MW To 400 MW Rankine Cycle Power PlantsLuis Angel SantamariaNessuna valutazione finora
- Esteves2006 Article InfluenceOfSteamHeatingOnThePrDocumento15 pagineEsteves2006 Article InfluenceOfSteamHeatingOnThePrFernandoNessuna valutazione finora
- Forest Harvesting and The Carbon Debt in Boreal East-Central CanadaDocumento17 pagineForest Harvesting and The Carbon Debt in Boreal East-Central CanadaBjart HoltsmarkNessuna valutazione finora
- Wood Ash DetailsDocumento19 pagineWood Ash DetailsG S DeshpandeNessuna valutazione finora
- Fire Performance Characteristics of Acetylated Ash Fraxinus Excelsior L. WoodDocumento5 pagineFire Performance Characteristics of Acetylated Ash Fraxinus Excelsior L. Woodvishnupriya r nairNessuna valutazione finora
- Evaluation of Biomass Properties For The Production of Solid BiofuelsDocumento7 pagineEvaluation of Biomass Properties For The Production of Solid BiofuelsAltamirando AltamirandoNessuna valutazione finora
- Mechanical Characterisation of Densi Fied Hardwood With Regard To Structural ApplicationsDocumento19 pagineMechanical Characterisation of Densi Fied Hardwood With Regard To Structural ApplicationsaantceNessuna valutazione finora
- Temperature Variation in A Sawdust Oven Using Different Wood SpeciesDocumento6 pagineTemperature Variation in A Sawdust Oven Using Different Wood SpeciesgirmagNessuna valutazione finora
- The Influence of Drying Temperature On The Hygroscopicity of Rubberwood (Hevea Brasiliensis)Documento11 pagineThe Influence of Drying Temperature On The Hygroscopicity of Rubberwood (Hevea Brasiliensis)Kumar RaghvendraNessuna valutazione finora
- Carbon Sink For A Century: Brief CommunicationsDocumento1 paginaCarbon Sink For A Century: Brief CommunicationsNilton FariasNessuna valutazione finora
- Garcia Et Al (2012) Wood Science and TechnologyDocumento12 pagineGarcia Et Al (2012) Wood Science and TechnologyRafael MedeirosNessuna valutazione finora
- Woody Biomass Characterization For 100 Megawatt-Hours (MW)Documento6 pagineWoody Biomass Characterization For 100 Megawatt-Hours (MW)Sek PyroNessuna valutazione finora
- Volatile Organic Compounds Emitted From Hardwood Drying As A Function of Processing ParametersDocumento8 pagineVolatile Organic Compounds Emitted From Hardwood Drying As A Function of Processing ParametersJose VeraNessuna valutazione finora
- Torrefaction of Reed Canary Grass, Wheat Straw and Willow To Enhance Solid Fuel Qualities and Combustion PropertiesDocumento13 pagineTorrefaction of Reed Canary Grass, Wheat Straw and Willow To Enhance Solid Fuel Qualities and Combustion Properties김형진Nessuna valutazione finora
- Density Biomass UkraineDocumento6 pagineDensity Biomass UkraineasertveNessuna valutazione finora
- Open Fires: Experiments and TheoryDocumento34 pagineOpen Fires: Experiments and TheoryIlya Bechara MastaNessuna valutazione finora
- Heat Value of Four Hardwood Species From Sudan: Tarig O. Khider and Osman T. ElsakiDocumento5 pagineHeat Value of Four Hardwood Species From Sudan: Tarig O. Khider and Osman T. ElsakiTarig KhiderNessuna valutazione finora
- Deposition of Inorganic Material in Biomass CombustionDocumento12 pagineDeposition of Inorganic Material in Biomass CombustionDiane HeistandNessuna valutazione finora
- Wood Thermal PropertiesDocumento6 pagineWood Thermal PropertiesKevonSingh1Nessuna valutazione finora
- Rapid Pyrolysis of Wood Block by Microwave HeatingDocumento13 pagineRapid Pyrolysis of Wood Block by Microwave HeatingMadhanNessuna valutazione finora
- ISO 834 Fire Exposure of Bamboo Medium Size FurnaceDocumento19 pagineISO 834 Fire Exposure of Bamboo Medium Size Furnacevishnupriya r nairNessuna valutazione finora
- Charring of Wood Based MaterialsDocumento11 pagineCharring of Wood Based MaterialsgunavathyNessuna valutazione finora
- Estimating Carbon Stocks in Sub Tropical 3Documento5 pagineEstimating Carbon Stocks in Sub Tropical 3Rizwan officialNessuna valutazione finora
- A2-Ajb-11 (9) 2204-2209 - 2012Documento23 pagineA2-Ajb-11 (9) 2204-2209 - 2012niglibNessuna valutazione finora
- The Role of Carbon Biotemplate Density in Mechanical Properties of Biomorphic SiCDocumento8 pagineThe Role of Carbon Biotemplate Density in Mechanical Properties of Biomorphic SiCAntonio CamaranoNessuna valutazione finora
- Hardwood HRRDocumento10 pagineHardwood HRRLegwanOPOLENessuna valutazione finora
- 1990 Analysis of The Fluidized Bed Drying of Wood ParticlesDocumento11 pagine1990 Analysis of The Fluidized Bed Drying of Wood ParticlesssshhawnNessuna valutazione finora
- A Review of Thermal Properties of Timber and Char at Elevated TemperaturesDocumento16 pagineA Review of Thermal Properties of Timber and Char at Elevated TemperaturesLannie FrancisNessuna valutazione finora
- Point SatuuDocumento10 paginePoint Satuukhaerunnisaaaz13Nessuna valutazione finora
- Polymers 12 00048Documento8 paginePolymers 12 00048fexara9242Nessuna valutazione finora
- Release of Trace Elements From Wood Ash by Nitric AcidDocumento15 pagineRelease of Trace Elements From Wood Ash by Nitric AcidManav SaxenaNessuna valutazione finora
- Unit 3 Types of Fuels and Their CharacteristicsDocumento13 pagineUnit 3 Types of Fuels and Their Characteristicsbakhtyar21Nessuna valutazione finora
- Burning of WoodDocumento7 pagineBurning of WoodadylightNessuna valutazione finora
- BioRes 14 2 3289 Aytin KC Effect ThermoWood Heat Treatm Props S Torminalis 13449Documento12 pagineBioRes 14 2 3289 Aytin KC Effect ThermoWood Heat Treatm Props S Torminalis 13449yonNessuna valutazione finora
- Chemistry in the Utilization of Wood: Pergamon Series of Monographs on Furniture and TimberDa EverandChemistry in the Utilization of Wood: Pergamon Series of Monographs on Furniture and TimberNessuna valutazione finora
- Mathematical Biosciences 22, 113-120 (1974) : of Physics, Purdue Fort WayneDocumento8 pagineMathematical Biosciences 22, 113-120 (1974) : of Physics, Purdue Fort WayneJuan Jose SossaNessuna valutazione finora
- Lisbon Yellow 4 in 1 Hop-On Hop-Off Bus and Tram MapDocumento1 paginaLisbon Yellow 4 in 1 Hop-On Hop-Off Bus and Tram MapJuan Jose SossaNessuna valutazione finora
- Worlds Apart: A Story of Three Possible Warmer WorldsDocumento1 paginaWorlds Apart: A Story of Three Possible Warmer WorldsJuan Jose SossaNessuna valutazione finora
- A Push For Green' Crackers: Steam-Cracker TrendsDocumento7 pagineA Push For Green' Crackers: Steam-Cracker TrendsJuan Jose SossaNessuna valutazione finora
- Recovery For The Paraxylene Industry or Temporary Reprieve?: Chemicals Research & AnalysisDocumento9 pagineRecovery For The Paraxylene Industry or Temporary Reprieve?: Chemicals Research & AnalysisJuan Jose SossaNessuna valutazione finora
- SBC Energy Institute - Hydrogen-Based Energy Conversion - AbstractDocumento17 pagineSBC Energy Institute - Hydrogen-Based Energy Conversion - AbstractJuan Jose SossaNessuna valutazione finora
- Moller (2012) Effects of Anaerobic Digestion On Digestate Nutrient Availability and Crop Growth - A ReviewDocumento17 pagineMoller (2012) Effects of Anaerobic Digestion On Digestate Nutrient Availability and Crop Growth - A ReviewJuan Jose SossaNessuna valutazione finora
- Effects of Operational Shocks On Key Microbial Populations For Biogas Production in UASB (Upflow Anaerobic Sludge Blanket) ReactorsDocumento9 pagineEffects of Operational Shocks On Key Microbial Populations For Biogas Production in UASB (Upflow Anaerobic Sludge Blanket) ReactorsJuan Jose SossaNessuna valutazione finora
- Guide For Students: Chemical Engineering Design ProjectDocumento10 pagineGuide For Students: Chemical Engineering Design ProjectJuan Jose SossaNessuna valutazione finora
- Perrigault (2012) Towards Thermal Design Optimization of Tubular Digesters in Cold Climates - A Heat Transfer ModelDocumento10 paginePerrigault (2012) Towards Thermal Design Optimization of Tubular Digesters in Cold Climates - A Heat Transfer ModelJuan Jose SossaNessuna valutazione finora
- Bornhoft (2013) Steady-State Analysis of The Anaerobic Digestion Model No 1Documento15 pagineBornhoft (2013) Steady-State Analysis of The Anaerobic Digestion Model No 1Juan Jose SossaNessuna valutazione finora
- Rosen (2006) Implementing ADM1 For Plantwide Benchmark Simulations in Matlab-SimulinkDocumento8 pagineRosen (2006) Implementing ADM1 For Plantwide Benchmark Simulations in Matlab-SimulinkJuan Jose SossaNessuna valutazione finora
- Jeong (2005) Analysis and Application of ADM1 For Anaerobic Methane ProductionDocumento9 pagineJeong (2005) Analysis and Application of ADM1 For Anaerobic Methane ProductionJuan Jose SossaNessuna valutazione finora
- Colussi (2012) Modelling of An Anaerobic Process Producing Biogas From Winery WastesDocumento6 pagineColussi (2012) Modelling of An Anaerobic Process Producing Biogas From Winery WastesJuan Jose SossaNessuna valutazione finora
- Walczak (2011) Dynamics of Methanogenic Archaeal Communities Based On The Rrna Analysis and Their Relation To Methanogenic ActivityDocumento8 pagineWalczak (2011) Dynamics of Methanogenic Archaeal Communities Based On The Rrna Analysis and Their Relation To Methanogenic ActivityJuan Jose SossaNessuna valutazione finora
- Gaida (2011) MATLAB Toolbox For Biogas Plant Modelling and OptimizationDocumento4 pagineGaida (2011) MATLAB Toolbox For Biogas Plant Modelling and OptimizationJuan Jose SossaNessuna valutazione finora
- Jeppsson (2007) Benchmark Simulation Model No 2 - General Protocol and Exploratory Case StudiesDocumento12 pagineJeppsson (2007) Benchmark Simulation Model No 2 - General Protocol and Exploratory Case StudiesJuan Jose SossaNessuna valutazione finora
- Batstone (2006) Industrial Applications of The IWA Anaerobic Digestion Model No. 1 (ADM1)Documento8 pagineBatstone (2006) Industrial Applications of The IWA Anaerobic Digestion Model No. 1 (ADM1)Juan Jose SossaNessuna valutazione finora
- Innerwear Industry Pitch PresentationDocumento19 pagineInnerwear Industry Pitch PresentationRupeshKumarNessuna valutazione finora
- Bomba MixerDocumento2 pagineBomba MixerBinelli79Nessuna valutazione finora
- Determinants - DPP 01 (Of Lec 02) - Lakshya JEE 2.0 2024Documento3 pagineDeterminants - DPP 01 (Of Lec 02) - Lakshya JEE 2.0 2024Apurv ChitranshNessuna valutazione finora
- Fulltext 4 PDFDocumento4 pagineFulltext 4 PDFSubhadip Banerjee0% (1)
- S TR GEN ID (Component Marking) (Rev 3 2009) - AN Marked UpDocumento6 pagineS TR GEN ID (Component Marking) (Rev 3 2009) - AN Marked UpsnclgsraoNessuna valutazione finora
- Scanner and Xcal Comperative Analysis v2Documento22 pagineScanner and Xcal Comperative Analysis v2Ziya2009Nessuna valutazione finora
- Naming Hydrocarbons Worksheet1 Key 12 26 08 PDFDocumento2 pagineNaming Hydrocarbons Worksheet1 Key 12 26 08 PDFMarilyn Castro Laquindanum100% (1)
- Fines Reduction Project at Wendling Bowser QuarryDocumento2 pagineFines Reduction Project at Wendling Bowser QuarryMarcos Antonio ParoliniNessuna valutazione finora
- CapstoneDocumento23 pagineCapstoneA - CAYAGA, Kirby, C 12 - HermonNessuna valutazione finora
- Guia de CondensadoresDocumento193 pagineGuia de CondensadoresPaola Segura CorreaNessuna valutazione finora
- Dawn of Solar PV CookingDocumento5 pagineDawn of Solar PV CookingAbhinav AgrawalNessuna valutazione finora
- Bulacan Agricultural State College: Lesson Plan in Science 4 Life Cycle of Humans, Animals and PlantsDocumento6 pagineBulacan Agricultural State College: Lesson Plan in Science 4 Life Cycle of Humans, Animals and PlantsHarmonica PellazarNessuna valutazione finora
- 3 Tasks and ANS - Gamma Maths PDFDocumento6 pagine3 Tasks and ANS - Gamma Maths PDFNora ZhangNessuna valutazione finora
- EXP4 The Diels Alder ReactionsDocumento3 pagineEXP4 The Diels Alder ReactionsLaura GuidoNessuna valutazione finora
- Inverse of One-To-One FunctionDocumento4 pagineInverse of One-To-One FunctionKathFaye EdaNessuna valutazione finora
- H107en 201906 r4 Elcor Elcorplus 20200903 Red1Documento228 pagineH107en 201906 r4 Elcor Elcorplus 20200903 Red1mokbelNessuna valutazione finora
- Dual Op Amp and Voltage Reference Ap4310/ADocumento12 pagineDual Op Amp and Voltage Reference Ap4310/AМихаил ЯненкоNessuna valutazione finora
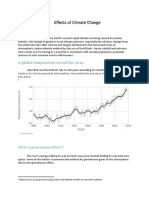
- Effects of Climate ChangeDocumento3 pagineEffects of Climate Changejiofjij100% (1)
- JKJKJDocumento3 pagineJKJKJjosecarlosvjNessuna valutazione finora
- Poly 103Documento20 paginePoly 103Sharifah Zulaikha BenYahyaNessuna valutazione finora
- Purpose in LifeDocumento1 paginaPurpose in Lifeaashish95100% (1)
- Cambridge IGCSE Paper 2Documento4 pagineCambridge IGCSE Paper 2irajooo epik zizterNessuna valutazione finora
- Updated G10 Class Routine Effective From 12 January 2023Documento1 paginaUpdated G10 Class Routine Effective From 12 January 2023NiloyNessuna valutazione finora
- Gemh 108Documento20 pagineGemh 108YuvrajNessuna valutazione finora
- Sika - Bitumen: Bitumen Emulsion Waterproof & Protective CoatingDocumento3 pagineSika - Bitumen: Bitumen Emulsion Waterproof & Protective Coatingdinu69inNessuna valutazione finora
- Toshiba: ® A20SeriesDocumento12 pagineToshiba: ® A20SeriesYangNessuna valutazione finora
- Parts Catalogue of Foton: (TC2A504-034K)Documento132 pagineParts Catalogue of Foton: (TC2A504-034K)МаксимNessuna valutazione finora
- SCM (Subway Project Report)Documento13 pagineSCM (Subway Project Report)Beast aNessuna valutazione finora
- Oil Whirl and Whip Instabilities - Within Journal BearingsDocumento27 pagineOil Whirl and Whip Instabilities - Within Journal BearingsTalha AamirNessuna valutazione finora
- 1762 Ob8 PDFDocumento16 pagine1762 Ob8 PDFRodríguez EdwardNessuna valutazione finora