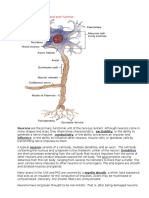
Documenti di Didattica
Documenti di Professioni
Documenti di Cultura
New Roles For Astrocytes - Regulation of Synaptic Transmission - Newman 2003
Caricato da
Lucas GallindoTitolo originale
Copyright
Formati disponibili
Condividi questo documento
Condividi o incorpora il documento
Hai trovato utile questo documento?
Questo contenuto è inappropriato?
Segnala questo documentoCopyright:
Formati disponibili
New Roles For Astrocytes - Regulation of Synaptic Transmission - Newman 2003
Caricato da
Lucas GallindoCopyright:
Formati disponibili
536
Review
TRENDS in Neurosciences Vol.26 No.10 October 2003
New roles for astrocytes:
Regulation of synaptic transmission
Eric A. Newman
Department of Neuroscience, University of Minnesota, 6-145 Jackson Hall, 321 Church Street SE, Minneapolis, MN 55455, USA
Although glia often envelop synapses, they have traditionally been viewed as passive participants in synaptic
function. Recent evidence has demonstrated, however,
that there is a dynamic two-way communication
between glia and neurons at the synapse. Neurotransmitters released from presynaptic neurons evoke Ca21
concentration increases in adjacent glia. Activated glia,
in turn, release transmitters, including glutamate and
ATP. These gliotransmitters feed back onto the presynaptic terminal either to enhance or to depress further
release of neurotransmitter. Transmitters released from
glia can also directly stimulate postsynaptic neurons,
producing either excitatory or inhibitory responses.
Based on these new findings, glia should be considered
an active partner at the synapse, dynamically regulating
synaptic transmission.
The presynaptic terminal and the postsynaptic neuron
have traditionally been viewed as the two functionally
important elements of the synapse. Yet a third cellular
component, the glial cell, is often associated with the
synaptic structure. In the cortex, synapses are ensheathed
by processes of astrocytes, the most common type of glial
cell in the brain [1,2]. Synapses in the cerebellum are
enveloped by Bergmann glia (specialized astrocytes) [3,4]
and synapses in the retina are contacted by Muller cells
(astrocyte-like radial glia) [5]. In the peripheral nervous
system, the neuromuscular junction is tightly ensheathed
by perisynaptic Schwann cells, which are specialized, nonmyelinating peripheral glia [6,7].
These perisynaptic glia have traditionally been thought
to play little role in synaptic function. Recent research has
revealed, however, that a lively, bidirectional conversation
is conducted between perisynaptic glia and the neuronal
elements at the synapse. Release of neurotransmitter from
the presynaptic terminal not only stimulates the postsynaptic neuron but also activates the perisynaptic glia. The
activated glial cell, in turn, releases gliotransmitters that
can directly stimulate the postsynaptic neuron and can
feed back onto the presynaptic terminal either to enhance
or to depress further release of neurotransmitter. (Transmitters released from glia will here be referred to as
gliotransmitters, to distinguish them from neurotransmitters released from neurons [8].) Thus, the perisynaptic
glial cell functions as an active partner in synaptic transmission. To understand synaptic function fully, the
Corresponding author: Eric A. Newman (ean@umn.edu).
synapse must be considered a tripartite, rather than a
bipartite, structure [9,10].
Many examples of bidirectional communication between
neurons and glia at the synapse have now been documented.
Experiments using culture and intact-tissue preparations
demonstrate modulation of synaptic transmission by glia.
Several of these studies will be highlighted in this review of
glial regulation of synaptic function.
Neuronal activation of glia
Astrocytes and other CNS glia express a wide variety of
neurotransmitter receptors [11,12]. Activation of these
receptors evokes a rich repertoire of responses in glia,
including increases in intracellular Ca2 concentration
([Ca2]) and release of gliotransmitters, including glutamate and ATP.
Glial responses to neurotransmitters were initially
characterized in vitro, using cultured astrocytes as a
model system. These studies demonstrated that glial
[Ca2] increases could be evoked by a variety of neurotransmitters, including glutamate, GABA, adrenaline,
ATP, serotonin, ACh and several peptides [11,12]. An
essential question remained unanswered by these early
studies, however: does the release of transmitters from
neurons in vivo evoke glial responses? Recent experiments
using brain slices have answered this question with a convincing yes, confirming findings from cell culture studies.
Release of several different neurotransmitters from
active neurons can stimulate glia in intact tissue preparations, evoking increases in glial [Ca2]. In hippocampal
slices, electrical stimulation of neurons evokes [Ca2]
increases in astrocytes. This response is mediated by
several different astrocytic receptors, including metabotropic glutamate receptors [13,14], GABAB receptors [15]
and muscarinic ACh receptors [16]. Similarly, in cerebellar
slices, electrical stimulation of parallel fibers evokes
[Ca2] increases in Bergmann glia [3]. In this instance,
the response is mediated by the release of nitric oxide (NO)
from neurons [17]. Moderate neuronal stimulation of
parallel fibers evokes [Ca2] increases in small regions
(microdomains) of individual Bergmann glia, suggesting
that subcellular compartments of glia can function as
independent units [3,4]. Neuronal activity can also evoke
[Ca2] increases in perisynaptic Schwann cells at the
neuromuscular junction, with stimulation rates as low as
10 Hz evoking glial responses [6,18].
The capacity of glia to respond to neuronal activity has
prompted suggestions that they might regulate local blood
http://tins.trends.com 0166-2236/$ - see front matter q 2003 Elsevier Ltd. All rights reserved. doi:10.1016/S0166-2236(03)00237-6
Review
TRENDS in Neurosciences Vol.26 No.10 October 2003
flow in response to changes in neuronal activity [19]. As
in other preparations, electrical stimulation of neurons in
cortical brain slices evokes [Ca2] increases in astrocytes
a response mediated by the release of glutamate from
neurons and activation of glial metabotropic receptors
[20]. Stimulation of neurons in this preparation also
induces dilation of blood vessels contacted by the glia. It
is likely that dilation is mediated by the glia, as direct
simulation of astrocytes induces vessel dilation. Ca2
chelators and inhibitors of cyclooxygenase block vessel
dilation, suggesting that glia might regulate blood flow by
a Ca2-dependent release of prostaglandins, which are
cyclooxygenase products [20].
An intriguing aspect of glial Ca2 responses is that they
sometimes trigger self-propagating waves. Activation of
glia in the retina [21,22] and in brain slices [23], by
mechanical stimulation or by application of neurotransmitters, evokes glial [Ca2] increases that propagate as
Ca2 waves through neighboring glia at distances of up to
several hundred mm. These intercellular Ca2 waves are
propagated by two mechanisms: by diffusion of inositol
(1,4,5)-trisphosphate [Ins(1,4,5)P3] through gap junctions
coupling the cells, and by release of ATP, which functions
as an extracellular messenger [22]. A crucial and, as yet,
(a)
Electrical
stimulus
537
unanswered question is whether transmitter release from
active neurons can trigger propagated intercellular glial
Ca2 waves. If propagated waves are evoked, they could
represent a mechanism by which glia modulate synaptic
transmission over long distances. Such propagated Ca2
waves might also contribute to the regulation of blood flow
in the brain.
Glutamate release from astrocytes
Glial [Ca2] increases evoked by neuronal activity might
be written off as a curious epiphenomenon of little
importance to brain function if it were not for the fact
that they initiate additional glial responses. Perhaps the
most significant of these Ca2-dependent responses is the
release of glutamate from glia.
In both culture [24 27] and brain-slice [15,14,28,29]
preparations, increases in [Ca2] result in a release of
glutamate from astrocytes. The mechanism by which
glutamate is released remains uncertain, although several
lines of evidence indicate that glutamate can be released
by a Ca2-dependent exocytotic process [28,30,31]. As
described in the following paragraphs, glutamate release
from astrocytes is an important means by which glia
Mechanical
stimulus
Recording
pipette
(3)
(2)
(1)
(b)
(i)
(ii)
(iii)
50 pA
50 ms
(c)
Prestimulus
Poststimulus
90 pA
500 ms
TRENDS in Neurosciences
Fig. 1. Glial regulation of synaptic transmission in co-cultures of astrocytes and neurons. (a) The glial neuronal culture preparation. A presynaptic neuron is stimulated
with electrical pulses (1) while synaptic currents are recorded from a postsynaptic neuron (2). Astrocytes beneath the neurons (yellow) are activated with a mechanical
stimulus (3). (b) Stimulation of the presynaptic neuron evokes an excitatory postsynaptic current (EPSC) in the postsynaptic neuron (i). Simultaneous stimulation of an adjacent astrocyte reduces the amplitude of the EPSC (ii). Following termination of glial stimulation, synaptic transmission recovers (iii). Glial depression of synaptic transmission was blocked by metabotropic-glutamate-receptor antagonists. Reproduced, with permission, from Ref. [25]. (c) Spontaneous miniature EPSCs (mEPSCs) recorded
from a neuron (prestimulus). The frequency of mEPSCs increases following stimulation of a neighboring astrocyte (poststimulus). Glial enhancement of mEPSCs was
blocked by NMDA antagonists. Reproduced, with permission, from Ref. [26], q (1998) the Society for Neuroscience.
http://tins.trends.com
Review
538
TRENDS in Neurosciences Vol.26 No.10 October 2003
(a)
Ast
Pyr
20 s
200 mV
86 mV
Rest
Bicuculline
50 pA
Stim
0.5 s
(b)
PGE2
PGE2
PGE2
60 s
NBQX + AP5
(c)
modulate the release of neurotransmitters from presynaptic terminals.
Modulation of synaptic transmission in culture
Glial regulation of synaptic transmission has been best
characterized in co-cultures of astrocytes and neurons,
where [Ca2] increases can be evoked in astrocytes by
several stimuli, including electrical and mechanical stimulation and application of peptides and prostaglandin E2
(PGE2) [24 26,32,33]. Activation of astrocytes reduces the
amplitude of excitatory and inhibitory postsynaptic currents (EPSCs and IPSCs, respectively) evoked by electrical
stimulation of presynaptic neurons [25] (Figure 1a,b). This
depression of synaptic transmission is mediated by glutamate release from astrocytes and is blocked by metabotropic-glutamate-receptor antagonists.
Spontaneous synaptic events recorded from postsynaptic neurons are also modulated by astrocytes. The frequency of both miniature EPSCs (mEPSCs) and miniature
IPSCs (mIPSCs) rises following astrocyte activation [26]
(Figure 1c). This response, opposite in sign to glial modulation of evoked synaptic potentials, is mediated by glutamate release from astrocytes and stimulation of presynaptic
NMDA receptors. Glutamate release from astrocytes can
also directly stimulate postsynaptic neurons, evoking
depolarizing slow inward currents by activating neuronal
AMPA and NMDA receptors [24,32,33]. In summary,
experiments utilizing co-cultures of astrocytes and neurons demonstrate that glia are able to modulate synaptic
transmission by release of glutamate.
(i)
(ii)
10 s
TRENDS in Neurosciences
Fig. 2. Glial regulation of synaptic transmission in intact tissue preparations.
(a) Spontaneous miniature inhibitory postsynaptic currents (mIPSCs) recorded
from a pyramidal neuron (Pyr) in a hippocampal slice (Rest). Following stimulation
of an astrocyte (Ast), the frequency of mIPSCs increases (Stim). The mIPSCs
are blocked by the GABAA-receptor antagonist bicuculline (bottom trace),
whereas glial enhancement of mIPSC frequency was blocked by AMPA- and
NMDA-receptor antagonists (not shown). Reproduced, with permission, from
Ref. [15], q (1998) Nature (http://www.nature.com), Macmillan Magazines Limited.
(b) Stimulation of astrocytes with prostaglandin E2 (PGE2) in a hippocampal slice
evokes increases in Ca2 concentration ([Ca2]) in an astrocyte (gray traces) and in
an adjacent neuron (black traces). Addition of the glutamate antagonists 6-nitro-7sulfamoyl-benz( f)quinoxaline-2,3-dione (NBQX) and 2-amino-5-phosphonopentanoate (AP5) blocks the neuronal response but not the glial response,
http://tins.trends.com
Modulation in brain slices
Recent experiments employing brain slices and other
preparations in which the morphological relationship
between neurons and glia is preserved also reveal glial
regulation of synaptic transmission. In the hippocampus,
repetitive firing of inhibitory interneurons leads to potentiation of synaptic transmission between the interneurons
and pyramidal cells. This potentiation is mediated by
glutamate release from neighboring astrocytes [15].
Potentiation arises when GABA, released from the
interneurons, activates astrocytic GABAB receptors, evoking a Ca2-dependent glutamate release from the glia. The
released glutamate activates interneuron AMPA and
NMDA receptors, potentiating transmitter release. Direct
electrical stimulation of hippocampal astrocytes leads to
increased mIPSC frequency (Figure 2a) and raises the
success rate of synaptic responses recorded from pyramidal neurons, most likely by Ca2-dependent glutamate
release from the glia [15].
demonstrating that glia stimulate the neuron by releasing glutamate. Reproduced,
with permission, from Ref. [28], q (1998) Nature (http://www.nature.com),
Macmillan Magazines Limited. (c) Glial modulation of light-evoked spike activity
recorded from ganglion cells in the retina. The top traces in (i) and (ii) show a
running average of neuronal spike frequency and the bottom traces show glial
Ca2 levels. (i) Neuron spike frequency increases when a stimulus (arrow) evokes
a glial [Ca2] increase. (ii) Spike frequency of a different neuron decreases when a
glial [Ca2] increase is evoked. Both excitatory and inhibitory modulation are
mediated by presynaptic mechanisms. The bar at the bottom shows the repetitive
light stimulus that evoked the spiking, with periods of light ON (open segments)
and OFF (closed segments) indicated. Reproduced, with permission, from Ref. [35],
q (1998) the Society for Neuroscience.
Review
539
TRENDS in Neurosciences Vol.26 No.10 October 2003
excitatory and inhibitory glial modulation is mediated by
presynaptic mechanisms (E.A. Newman, unpublished).
In addition to the excitatory and inhibitory glial modulation of synaptic transmission, glia can influence neuronal activity in the retina by a third mechanism: direct
inhibition of postsynaptic ganglion cells [36]. When Muller
cells, the specialized radial glia of the retina, are activated,
they release ATP into the inner synaptic layer of the
retina. The released ATP is rapidly converted in the
extracellular space to adenosine, which activates ganglion
cell A1 receptors and inhibits the neurons by increasing K
conductance.
Release of ATP from glia could have widespread modulatory effects in the CNS. Activation of astrocytes as well as
Muller cells results in ATP release [22,37,38] (although the
mechanism responsible for ATP release from glia is a
matter of debate). Released ATP could excite neurons
directly through activation of P2X receptors. Alternatively,
released ATP, once converted to adenosine, could inhibit
neurons by activating A1 receptors, as it does in the retina
[36]. Adenosine could also act presynaptically via A1 and
A2 receptors to either depress or potentiate synaptic
transmission. In addition, released ATP can activate additional glia via glial purinergic receptors [11,12,21,22].
Additional experiments in brain slices of the hippocampus [14,28] and thalamus [34] demonstrate that activated astrocytes can directly stimulate neurons by release
of glutamate. Application of glutamate agonists and
PGE2 in addition to neuronal stimulation evokes a
Ca2-dependent glutamate release from astrocytes.
Released glutamate activates neighboring neurons and
results in increases in neuronal Ca2 levels (Figure 2b).
The evoked neuronal responses are blocked by antagonists
of AMPA and NMDA receptors.
Modulation in the retina
Glia can also modulate synaptic activity driven by natural,
physiological stimuli. This has been demonstrated in the
mammalian retina, where glial activation modulates
ganglion cell spike activity that is driven by light stimulation [35]. (Ganglion cells are the output neurons of the
retina that project to the brain.) For some ganglion cells,
light-evoked spiking is enhanced when neighboring glia
are activated; in other ganglion cells, spiking is inhibited
(Figure 2c). The precise mechanism of glial modulation of
ganglion cell activity is not known, but recent experiments
monitoring ganglion cell EPSCs demonstrate that both
(a)
(1)
0.5
EPC amplitude (mV)
Motor
nerve
Injection
pipette
(3)
(2)
0.4
0.3
0.2
0.1
Recording
pipette
Muscle
fiber
0.0
0
(b) (i)
120
EPC amplitude (% control)
Poke Inject
PSC GTPS
(c)
10
20
30
Time (min)
40
50
(ii)
120
100
100
80
80
60
60
40
40
20
20
10 Hz
0
0
10 Hz
4
6
Time (min)
0
8
6
4
Time (min)
TRENDS in Neurosciences
Fig. 3. Glial depression of synaptic transmission at the neuromuscular junction. (a) The neuromuscular junction preparation. A motor nerve axon (1) synapses onto a
muscle fiber. The synaptic junction (2) is ensheathed by a perisynaptic Schwann cell (red, 3). Stimulation of the motor nerve evokes an endplate current (EPC), which is
recorded from the muscle fiber. EPC amplitudes are graphed in (b) and (c). (b) Synaptic transmission is depressed when the motor nerve is stimulated by a train of pulses
(i). The activity-dependent depression is blocked following injection of the GDP analog GDPbS into the perisynaptic Schwann cell (ii). (c) Injection of the GTP analog GTPgS
into a perisynaptic Schwann cell induces depression at the synapse ensheathed by the glial cell. The arrow (Poke PSC) indicates when the injection pipette penetrates the
perisynaptic Schwann cell. (b) and (c) reproduced, with permission, from Ref. [7].
http://tins.trends.com
540
Review
TRENDS in Neurosciences Vol.26 No.10 October 2003
junction preparation closely reproduces conditions in vivo.
In addition, levels of motor nerve stimulation that reliably
evoke glial depression of synaptic transmission (10 Hz) are
well within the range that occurs in vivo [18,40].
Modulation at the neuromuscular junction
Glial modulation of synaptic transmission is not limited to
the CNS. A dramatic example of such modulation occurs at
the neuromuscular junction, where perisynaptic Schwann
cells are responsible for half of the synaptic depression
seen during repetitive stimulation of the motor nerve
[6,18,39,40]. Release of ACh and ATP from the presynaptic
terminal activates the perisynaptic Schwann cell, evoking
a Ca2-independent release of a gliotransmitter, thought
to be glutamate, from the glial cell [6,18,39,41]. The
released gliotransmitter, through production of NO, feeds
back onto the presynaptic terminal, decreasing ACh release
[39]. Perisynaptic Schwann cell depression of synaptic
transmission is mediated by activation of a G-protein
pathway in the glial cell. Synaptic depression is blocked
by the GDP analog GDPbS and is stimulated by the GTP
analog GTPgS injected into the glial cell [7] (Figure 3).
Glial regulation of synaptic transmission at the neuromuscular junction is complex, as perisynaptic Schwann
cells possess a second, Ca2-dependent mechanism by
which they can enhance transmitter release [42]. This
excitatory action of the glial cell is weaker than is the
inhibitory action and is observed only when inhibition
is blocked.
Perisynaptic Schwann cell modulation of synaptic transmission at the neuromuscular junction provides perhaps
the most convincing evidence to date that glia regulate
synapses in vivo. Morphologically, the neuromuscular
Other forms of synaptic modulation
As reviewed in preceding sections, release of gliotransmitters from glia can modulate the release of neurotransmitters from the presynaptic terminal and can stimulate
postsynaptic neurons. This mechanism of synaptic regulation can be termed direct modulation. Glia can also
regulate synaptic transmission by indirect mechanisms.
The best characterized of the indirect regulatory mechanisms is the uptake of glutamate by glia via excitatory
amino acid transporters [43,44]. Synaptic transmission at
glutamatergic synapses is terminated by removal of glutamate from the synaptic cleft. Glia (primarily astrocytes in
the brain and Muller cells in the retina) account for the
bulk of glutamate uptake at the synapse [45 47]. Glial
uptake of glutamate can significantly affect synaptic
transmission. In the retina, for instance, the amplitude
and duration of ganglion cell EPSCs are dramatically
increased when the Muller glial glutamate transporter is
blocked [48]. Glial glutamate transport can also modulate
short-term synaptic plasticity [49].
Glia also modulate synaptic transmission by releasing
chemical cofactors. The best-documented example of such
modulation is activation of the NMDA receptor, which
Glia
Glutamate uptake
Presynaptic
neuron
H+
K+
10
Postsynaptic
neuron
Glu
7
ATP
4
5
Glu
Glu
Glu
ATP
Ca2+
IP3
Glia
6
Glia
IP3
Ca2+ store
ATP
Ca2+
2
Ca2+ store
TRENDS in Neurosciences
Fig. 4. Summary of proposed mechanisms of glial regulation of synaptic transmission. Release of glutamate (Glu) from the presynaptic terminal activates glial receptors
(1), evoking an increase in Ca2 levels (2) and the release of glutamate from glia. Glutamate activation of presynaptic receptors (3) regulates transmitter release, while activation of postsynaptic receptors (4) directly depolarizes neurons. Stimulation of glia also elicits the release of ATP, which inhibits postsynaptic neurons by activating A1
receptors (5). Activation of glia might also evoke an intercellular Ca2 wave, which propagates between glia by diffusion of inositol (1,4,5)-trisphosphate (IP3) through gap
junctions (6) and by release of ATP (7), and results in the modulation of distant synapses (8). Glia also modulate synaptic transmission by uptake of glutamate (9) and by
regulating extracellular K and H levels (10). Other glial neuronal interactions are not illustrated. These include glial activation by neurotransmitters other than glutamate
and glial regulation of NMDA-receptor-containing synapses by release of D -serine.
http://tins.trends.com
Review
TRENDS in Neurosciences Vol.26 No.10 October 2003
requires the presence of glutamate as well as a cofactor
that binds to the glycine-binding site of the receptor.
Recent evidence indicates that D -serine, not glycine, is the
endogenous agonist that activates the glycine-binding site
[50,51]. Glia (astrocytes in the brain and Muller cells in the
retina) are the sole source of D -serine in the CNS and most
likely modulate synaptic transmission at NMDA synapses
by releasing D -serine [52,53].
A third indirect mechanism of modulation is via glial
regulation of extracellular ion levels. Neuronal activity
leads to substantial variations in the concentrations of
K and H in the extracellular space [54 57]. These
variations can alter synaptic transmission; increases in
K levels depolarize synaptic terminals [58], whereas
H block presynaptic Ca2 channels [59,60] and NMDA
receptors [61]. Glia play an essential role in regulating
extracellular K and H concentrations [55,62,63], thus
influencing the effect of these ions on synaptic transmission.
Glia can also modulate synaptic transmission by directly
controlling synaptogenesis. Retinal ganglion cells, when
cultured in the absence of glia, display little synaptic
activity. Addition of astrocytes to these cultures substantially increases the frequency of mEPSCs, decreases the
failure rate of evoked transmission, and increases the
number of morphologically identified synapses [64,65].
Glial enhancement of synapse formation is mediated, at
least in part, by the release from astrocytes of cholesterol
complexed to apolipoprotein-E-containing lipoproteins [66].
Concluding remarks
There is increasing evidence that glia play a dynamic role
in regulating synaptic transmission. Experiments conducted in both culture and intact-tissue preparations
demonstrate that transmitters released from neurons can
stimulate glia, leading to the release of glutama4te, ATP
and other neuroactive substances from the glia. These
gliotransmitters can feed back onto the presynaptic terminal to either enhance or depress the further release of
neurotransmitter. Gliotransmitters released from glia can
directly stimulate postsynaptic neurons as well, evoking
either excitatory or inhibitory responses in these cells.
These glial neuronal interactions are summarized in
Figure 4.
Box 1. Key questions for future research
What mechanisms are responsible for the release of
gliotransmitters, such as glutamate and ATP, from glia?
Does the activity of neurons in vivo evoke increases in Ca2
concentration in glia at synapses? What patterns of
neuronal activity induce glial activation?
Do these glial increases in Ca2 concentration trigger Ca2
waves that propagate between glial cells?
Do activated glia release gliotransmitters at synapses
in vivo? Which gliotransmitters modulate the release of
neurotransmitters from presynaptic terminals? Which
gliotransmitters directly stimulate postsynaptic neurons?
What role do glia play in information processing, learning
and memory in the brain?
http://tins.trends.com
541
Despite recent advances, many important questions
remain to be answered concerning glial regulation of
synaptic transmission (Key questions for future research).
Are glia activated by neurons in vivo, as in situ experiments suggest? Does the release of gliotransmitters from
glia in vivo modulate the release of neurotransmitters at
synapses and directly stimulate postsynaptic neurons, as
it does in culture and in intact-tissue preparations? What
role do glia play in information processing, learning and
memory? These questions must be answered before we can
fully understand the function of glia at the synapse.
Acknowledgements
My research is supported by NIH grant EY04077 I thank Paul Ceelen for
preparation of illustrations and Janice Gepner, Paul Mermelstein and
Monica R. Metea for comments on the manuscript.
References
1 Ventura, R. and Harris, K.M. (1999) Three-dimensional relationships between hippocampal synapses and astrocytes. J. Neurosci. 19,
6897 6906
2 Schikorski, T. and Stevens, C.F. (1999) Quantitative fine-structural
analysis of olfactory cortical synapses. Proc. Natl. Acad. Sci. U. S. A.
96, 4107 4112
3 Grosche, J. et al. (1999) Microdomains for neuron glia interaction:
parallel fiber signaling to Bergmann glial cells. Nat. Neurosci. 2,
139 143
4 Grosche, J. et al. (2002) Bergmann glial cells form distinct morphological structures to interact with cerebellar neurons. J. Neurosci. Res.
68, 138 149
5 Newman, E.A. (2001) Chapter 6: Glia of the retina. In Retina (Vol. 3),
3rd edn, (Ryan, S.J., ed.), pp. 89 103, Mosby
6 Reist, N.E. and Smith, S.J. (1992) Neurally evoked calcium transients
in terminal Schwann cells at the neuromuscular junction. Proc. Natl.
Acad. Sci. U. S. A. 89, 7625 7629
7 Robitaille, R. (1998) Modulation of synaptic efficacy and synaptic
depression by glial cells at the frog neuromuscular junction. Neuron
21, 847 855
8 Volterra, A. and Bezzi, P. (2002) Release of transmitters from glial cells.
In The Tripartite Synapse (Volterra, A. et al., eds), pp. 164 182, Oxford
University Press
9 Araque, A. et al. (1999) Tripartite synapses: glia, the unacknowledged
partner. Trends Neurosci. 22, 208 215
10 Volterra, A. et al. (2002) The Tripartite Synapse. Glia in Synaptic
Transmission, Oxford University Press
11 Finkbeiner, S.M. (1993) Glial calcium. Glia 9, 83 104
12 Porter, J.T. and McCarthy, K.D. (1997) Astrocytic neurotransmitter
receptors in situ and in vivo. Prog. Neurobiol. 51, 439 455
13 Porter, J.T. and McCarthy, K.D. (1996) Hippocampal astrocytes in situ
respond to glutamate released from synaptic terminals. J. Neurosci.
16, 5073 5081
14 Pasti, L. et al. (1997) Intracellular calcium oscillations in astrocytes:
a highly plastic, bidirectional form of communication between neurons
and astrocytes in situ. J. Neurosci. 17, 7817 7830
15 Kang, J. et al. (1998) Astrocyte-mediated potentiation of inhibitory
synaptic transmission. Nat. Neurosci. 1, 683 692
16 Araque, A. et al. (2002) Synaptically released acetylcholine evokes
Ca2 elevations in astrocytes in hippocampal slices. J. Neurosci. 22,
2443 2450
17 Matyash, V. et al. (2001) Nitric oxide signals parallel fiber activity to
Bergmann glial cells in the mouse cerebellar slice. Mol. Cell. Neurosci.
18, 664 670
18 Jahromi, B.S. et al. (1992) Transmitter release increases intracellular
calcium in perisynaptic Schwann cells in situ. Neuron 8, 1069 1077
19 Paulson, O.B. and Newman, E.A. (1987) Does the release of potassium
from astrocyte endfeet regulate cerebral blood flow? Science 237,
896 898
20 Zonta, M. et al. (2003) Neuron-to-astrocyte signaling is central to the
dynamic control of brain microcirculation. Nat. Neurosci. 6, 43 50
542
Review
TRENDS in Neurosciences Vol.26 No.10 October 2003
21 Newman, E.A. and Zahs, K.R. (1997) Calcium waves in retinal glial
cells. Science 275, 844 847
22 Newman, E.A. (2001) Propagation of intercellular calcium waves in
retinal astrocytes and Muller cells. J. Neurosci. 21, 2215 2223
23 Schipke, C.G. et al. (2002) Astrocyte Ca2 waves trigger responses in
microglial cells in brain slices. FASEB J. 16, 255 257
24 Parpura, V. et al. (1994) Glutamate-mediated astrocyte neuron
signalling. Nature 369, 744 747
25 Araque, A. et al. (1998) Glutamate-dependent astrocyte modulation
of synaptic transmission between cultured hippocampal neurons.
Eur. J. Neurosci. 10, 2129 2142
26 Araque, A. et al. (1998) Calcium elevation in astrocytes causes an
NMDA receptor-dependent increase in the frequency of miniature
synaptic currents in cultured hippocampal neurons. J. Neurosci. 18,
6822 6829
27 Innocenti, B. et al. (2000) Imaging extracellular waves of glutamate
during calcium signaling in cultured astrocytes. J. Neurosci. 20,
1800 1808
28 Bezzi, P. et al. (1998) Prostaglandins stimulate calcium-dependent
glutamate release in astrocytes. Nature 391, 281 285
29 Pasti, L. et al. (2001) Cytosolic calcium oscillations in astrocytes may
regulate exocytotic release of glutamate. J. Neurosci. 21, 477 484
30 Araque, A. et al. (2000) SNARE protein-dependent glutamate release
from astrocytes. J. Neurosci. 20, 666 673
31 Mazzanti, M. et al. (2001) Glutamate on demand: astrocytes as a ready
source. Neuroscientist 7, 396 405
32 Hassinger, T.D. et al. (1995) Evidence for glutamate-mediated activation of hippocampal neurons by glial calcium waves. J. Neurobiol.
28, 159 170
33 Sanzgiri, R.P. et al. (1999) Prostaglandin E2 stimulates glutamate
receptor-dependent astrocyte neuromodulation in cultured hippocampal cells. J. Neurobiol. 41, 221 229
34 Parri, H.R. et al. (2001) Spontaneous astrocytic Ca2 oscillations in situ
drive NMDAR-mediated neuronal excitation. Nat. Neurosci. 4, 803812
35 Newman, E.A. and Zahs, K.R. (1998) Modulation of neuronal activity
by glial cells in the retina. J. Neurosci. 18, 4022 4028
36 Newman, E.A. (2003) Glial cell inhibition of neurons by release of ATP.
J. Neurosci. 23, 1659 1666
37 Cotrina, M.L. et al. (1998) Connexins regulate calcium signaling by
controlling ATP release. Proc. Natl. Acad. Sci. U. S. A. 95, 1573515740
38 Wang, Z. et al. (2000) Direct observation of calcium-independent
intercellular ATP signaling in astrocytes. Anal. Chem. 72, 2001 2007
39 Thomas, S. and Robitaille, R. (2001) Differential frequency-dependent
regulation of transmitter release by endogenous nitric oxide at the
amphibian neuromuscular synapse. J. Neurosci. 21, 1087 1095
40 Rochon, D. et al. (2001) Synapse glia interactions at the mammalian
neuromuscular junction. J. Neurosci. 21, 3819 3829
41 Pinard, A. et al. (2002) NO-dependence of glutamate-mediated
synaptic depression at the frog neuromuscular junction. In 2002
Abstract Viewer and Itinerary Planner, Program No. 838.6, Society for
Neuroscience, Online
42 Castonguay, A. and Robitaille, R. (2001) Differential regulation of
transmitter release by presynaptic and glial Ca2 internal stores at the
neuromuscular synapse. J. Neurosci. 21, 1911 1922
43 Bergles, D.E. et al. (1999) Clearance of glutamate inside the synapse
and beyond. Curr. Opin. Neurobiol. 9, 293 298
44 Anderson, C.M. and Swanson, R.A. (2000) Astrocyte glutamate
transport: review of properties, regulation, and physiological functions. Glia 32, 1 14
45 Rothstein, J.D. et al. (1996) Knockout of glutamate transporters
46
47
48
49
50
51
52
53
54
55
56
57
58
59
60
61
62
63
64
65
66
reveals a major role for astroglial transport in excitotoxicity and
clearance of glutamate. Neuron 16, 675 686
Bergles, D.E. and Jahr, C.E. (1998) Glial contribution to glutamate
uptake at Schaffer collateral commissural synapses in the hippocampus. J. Neurosci. 18, 7709 7716
Oliet, S.H.R. et al. (2001) Control of glutamate clearance and synaptic
efficacy by glial coverage of neurons. Science 292, 923 926
Higgs, M.H. and Lukasiewicz, P.D. (1999) Glutamate uptake
limits synaptic excitation of retinal ganglion cells. J. Neurosci. 19,
3691 3700
Turecek, R. and Trussell, L.O. (2000) Control of synaptic depression by
glutamate transporters. J. Neurosci. 20, 2054 2063
Wolosker, H. et al. (1999) Serine racemase: a glial enzyme synthesizing
D -serine to regulate glutamate-N -methyl-D -aspartate neurotransmission. Proc. Natl. Acad. Sci. U. S. A. 96, 13409 13414
Mothet, J-P. et al. (2000) D -serine is an endogenous ligand for the
glycine site of the N -methyl-D -aspartate receptor. Proc. Natl. Acad. Sci.
U. S. A. 97, 4926 4931
Schell, M.J. et al. (1995) D -serine, an endogenous synaptic modulator:
localization to astrocytes and glutamate-stimulated release. Proc.
Natl. Acad. Sci. U. S. A. 92, 3948 3952
Stevens, E.R. et al. (2003) D -serine and serine racemase are present in
the vertebrate retina and contribute to the functional expression of
NMDA receptors. Proc. Natl. Acad. Sci. U. S. A. 100, 6789 6794
Kelly, J.P. and Van Essen, D.C. (1974) Cell structure and function in
the visual cortex of the cat. J. Physiol. 238, 515 547
Newman, E.A. (1995) Chapter 47: Glial cell regulation of extracellular
potassium. In Neuroglia (Kettenmann, H. and Ransom, B.R., eds),
pp. 717 731, Oxford University Press
Karwoski, C.J. et al. (1985) Light-evoked increases in extracellular
K in the plexiform layers of amphibian retinas. J. Gen. Physiol. 86,
189 213
Chesler, M. and Kaila, K. (1992) Modulation of pH by neuronal activity.
Trends Neurosci. 15, 396 402
Raushe, G. et al. (1990) Effects of changes in extracellular potassium,
magnesium and calcium concentration on synaptic transmission in
area CA1 and the dentate gyrus of rat hippocampal slices. Pflugers
Arch. 415, 588 593
Barnes, S. and Bui, Q. (1991) Modulation of calcium-activated chloride
current via pH-induced changes of calcium channel properties in cone
photoreceptors. J. Neurosci. 11, 4015 4023
Prodhom, B. et al. (1989) Interactions of protons with single open
L-type calcium channels. Location of protonation site and dependence
of proton-induced current fluctuations on concentration and species of
permeant ion. J. Gen. Physiol. 94, 23 42
Traynelis, S.F. and Cull-Candy, S.G. (1990) Proton inhibition of
N -methyl-D -aspartate receptors in cerebellar neurons. Nature 345,
347 350
Chen, J.C.T. and Chesler, M. (1992) pH transients evoked by excitatory
synaptic transmission are increased by inhibition of extracellular
carbonic anhydrase. Proc. Natl. Acad. Sci. U. S. A. 89, 7786 7790
Newman, E.A. (1996) Acid efflux from retinal glial cells generated by
sodium-bicarbonate cotransport. J. Neurosci. 16, 159 168
Pfrieger, F.W. and Barres, B.A. (1997) Synaptic efficacy enhanced by
glial cells in vitro. Science 277, 1684 1688
Ullian, E.M. et al. (2001) Control of synapse number by glia. Science
291, 657 661
Mauch, D.H. et al. (2001) CNS synaptogenesis promoted by gliaderived cholesterol. Science 294, 1354 1357
News & Features on BioMedNet
Start your day withBioMedNets own daily science news, features, research update articles and special reports. Every two weeks,
enjoy BioMedNet Magazine, which contains free articles from Trends, Current Opinion, Cell and Current Biology. Plus, subscribe to
Conference Reporter to get daily reports direct from major life science meetings.
http://news.bmn.com
http://tins.trends.com
Potrebbero piacerti anche
- Seeley's Chapter 8 Nervous SystemDocumento100 pagineSeeley's Chapter 8 Nervous SystemChristine Tapawan88% (8)
- Central Nervous System OVL ANA 2103Documento76 pagineCentral Nervous System OVL ANA 2103Baguma MichaelNessuna valutazione finora
- MCB 62 Midterm 1 Study GuideDocumento6 pagineMCB 62 Midterm 1 Study GuideLauren MikeNessuna valutazione finora
- Nervous System Anatomy and Physiology LectureDocumento105 pagineNervous System Anatomy and Physiology LectureButch Dumdum75% (12)
- 4 Physiology Supplement Handout Based On Ganong For TOPNOTCH 21 by Jaffar PinedaDocumento46 pagine4 Physiology Supplement Handout Based On Ganong For TOPNOTCH 21 by Jaffar PinedaAthena Borja100% (3)
- Biological Relevance of Neurotransmitter ReleaseDocumento12 pagineBiological Relevance of Neurotransmitter ReleaseazzaassNessuna valutazione finora
- Badimon Et Al - 2020 - Negative Feedback Control of Neuronal Activity by MicrogliaDocumento41 pagineBadimon Et Al - 2020 - Negative Feedback Control of Neuronal Activity by Microgliakeon.arbabi.altNessuna valutazione finora
- Activity-Dependent Structural and Functional Plasticity of Astrocyte-Neuron InteractionsDocumento20 pagineActivity-Dependent Structural and Functional Plasticity of Astrocyte-Neuron InteractionsAmar JitjNessuna valutazione finora
- Molecular Mechanisms of Neurotransmitter ReleaseDocumento21 pagineMolecular Mechanisms of Neurotransmitter ReleaseNusrat JahanNessuna valutazione finora
- SynapseDocumento45 pagineSynapseroyjason4518Nessuna valutazione finora
- GABA Depolarizes Immature NeuronsDocumento13 pagineGABA Depolarizes Immature NeuronsDaniel ChaguenNessuna valutazione finora
- Stress Gates An Astrocytic Energy Reservoir To Impair Synaptic PlasticityDocumento18 pagineStress Gates An Astrocytic Energy Reservoir To Impair Synaptic PlasticityCony GSNessuna valutazione finora
- Astrocytes in Endocannabinoid Signalling: ReviewDocumento7 pagineAstrocytes in Endocannabinoid Signalling: ReviewHelington CostaNessuna valutazione finora
- Norepinephrine Modulates The Motility of Resting and Activated Microglia Via Different Adrenergic ReceptorsDocumento13 pagineNorepinephrine Modulates The Motility of Resting and Activated Microglia Via Different Adrenergic ReceptorsCarolAlvaradoNessuna valutazione finora
- Aims and Significance: Protti Sesqui R & D - 1Documento5 pagineAims and Significance: Protti Sesqui R & D - 1Jackson ChenNessuna valutazione finora
- AS Bases Celulares DE LAS Oscilaciones Neuronales: UmmaryDocumento8 pagineAS Bases Celulares DE LAS Oscilaciones Neuronales: Ummarydavid alejandro pinzon sanchezNessuna valutazione finora
- Gating + HomeostaticDocumento7 pagineGating + HomeostaticGhea Mae VillasNessuna valutazione finora
- Efficient Generation of Reciprocal Signals by Inhibition: K.khodakhah@einstein - Yu.eduDocumento42 pagineEfficient Generation of Reciprocal Signals by Inhibition: K.khodakhah@einstein - Yu.eduDulce AguirreNessuna valutazione finora
- Ni Hms 437509Documento19 pagineNi Hms 437509diijah678Nessuna valutazione finora
- M HypothalamusDocumento15 pagineM HypothalamusSrilakshmi SridharanNessuna valutazione finora
- G Proteins: Nicotinic Cholinergic ReceptorsDocumento2 pagineG Proteins: Nicotinic Cholinergic ReceptorsKatrina Vea CalaroNessuna valutazione finora
- Neuromuscular JunctionDocumento16 pagineNeuromuscular JunctionSamadshahirNessuna valutazione finora
- NeurotransmittersDocumento7 pagineNeurotransmittersAnn MutisyaNessuna valutazione finora
- Cytokines As Emerging Regulators of Central Nervou - 231113 - 111110Documento12 pagineCytokines As Emerging Regulators of Central Nervou - 231113 - 111110Maurício DallowayNessuna valutazione finora
- Physiology, Neuromuscular Junction of Skeletal MuscleDocumento8 paginePhysiology, Neuromuscular Junction of Skeletal Musclemethmal subasingheNessuna valutazione finora
- Lin 1993Documento13 pagineLin 1993Stress LessNessuna valutazione finora
- Seizure PDFDocumento3 pagineSeizure PDFjustinedaleespedidoNessuna valutazione finora
- TMP 9 DA2Documento11 pagineTMP 9 DA2FrontiersNessuna valutazione finora
- AcetylcholineDocumento50 pagineAcetylcholineEliza GhinitaNessuna valutazione finora
- Dresbach Et AlDocumento17 pagineDresbach Et Alalma ramirezNessuna valutazione finora
- Chapter 1Documento21 pagineChapter 1Chrisantus OkakaNessuna valutazione finora
- Gaba Actions and Ionic Plasticity in Epilepsy: SciencedirectDocumento8 pagineGaba Actions and Ionic Plasticity in Epilepsy: SciencedirectMuhammad Bayu Zohari HutagalungNessuna valutazione finora
- Norepinephrine Modulates Single Hypothalamic Arcuate Neurons ViaDocumento12 pagineNorepinephrine Modulates Single Hypothalamic Arcuate Neurons ViaMariann DuzzNessuna valutazione finora
- 5 GabaDocumento11 pagine5 GabaJose GiovannyNessuna valutazione finora
- Synaptic TransmissionDocumento17 pagineSynaptic TransmissionbnmjgcNessuna valutazione finora
- Long-Term Potentiation of Intrinsic Excitability at The Mossy Fiber - Granule Cell Synapse of Rat CerebellumDocumento9 pagineLong-Term Potentiation of Intrinsic Excitability at The Mossy Fiber - Granule Cell Synapse of Rat CerebellumJar JarNessuna valutazione finora
- Introduction To CNS PharmacologyDocumento49 pagineIntroduction To CNS Pharmacologymatchees-gone rogueNessuna valutazione finora
- Computational Models of Neuron-Astrocyte Interaction in EpilepsyDocumento10 pagineComputational Models of Neuron-Astrocyte Interaction in EpilepsyEmma FineNessuna valutazione finora
- Neuro PlasticityDocumento67 pagineNeuro Plasticitydrkadiyala2100% (1)
- As Troc YtesDocumento16 pagineAs Troc YtesMelanie Christine Soriano PascuaNessuna valutazione finora
- Novel Mechanisms of Action of Three Antiepileptic Drugs, Vigabatrin, Tiagabine, and TopiramateDocumento8 pagineNovel Mechanisms of Action of Three Antiepileptic Drugs, Vigabatrin, Tiagabine, and Topiramatedpf050Nessuna valutazione finora
- Velisek 2017 Puritan S View of The Menage A TroisDocumento3 pagineVelisek 2017 Puritan S View of The Menage A TroissilenceindigoNessuna valutazione finora
- Neurotransmission, Measuring Chemical Events In: Advanced ArticleDocumento12 pagineNeurotransmission, Measuring Chemical Events In: Advanced ArticleazzaassNessuna valutazione finora
- AstrocytesDocumento19 pagineAstrocytesMaria PardoNessuna valutazione finora
- BDNF en PCKDocumento8 pagineBDNF en PCKJesusNessuna valutazione finora
- Nociceptive Stimuli Activate The Hypothalamus-HabeDocumento24 pagineNociceptive Stimuli Activate The Hypothalamus-HabeVissente TapiaNessuna valutazione finora
- Cells: The Potential of Targeting Brain Pathology With Ascl1/Mash1Documento7 pagineCells: The Potential of Targeting Brain Pathology With Ascl1/Mash1Paige MunroeNessuna valutazione finora
- Introduction To Neural Plasticity MechanismDocumento9 pagineIntroduction To Neural Plasticity MechanismAlain CésarNessuna valutazione finora
- Central Pattern GeneratorsDocumento4 pagineCentral Pattern GeneratorsCHANGEZ KHAN SARDARNessuna valutazione finora
- HPHY 225 Assignment - FinalDocumento5 pagineHPHY 225 Assignment - FinalSylvester MathiasNessuna valutazione finora
- Varela Parodi 2011Documento4 pagineVarela Parodi 2011Jorge ParodiNessuna valutazione finora
- PIIS2211124723007945Documento23 paginePIIS2211124723007945Murillo MoraesNessuna valutazione finora
- Pathophysiology of EpilepsyDocumento16 paginePathophysiology of EpilepsyYexalen Zazil100% (1)
- Astrocito y SueñoDocumento17 pagineAstrocito y SueñoLEISLY TATIANA OVIEDO GOMEZNessuna valutazione finora
- c30 2014 PannaschDocumento13 paginec30 2014 PannaschkarlosicaNessuna valutazione finora
- KULIAH BLOK SYARAF PERILAKU-1-ELEKTROFISIOLOGI OTAK & EEG - DR SonnyDocumento104 pagineKULIAH BLOK SYARAF PERILAKU-1-ELEKTROFISIOLOGI OTAK & EEG - DR SonnyAndhani putri.kNessuna valutazione finora
- Pocket - Companion - To - Guyton - and - Hall - Textbook - of - Medical - Physiology - ExportedDocumento7 paginePocket - Companion - To - Guyton - and - Hall - Textbook - of - Medical - Physiology - Exportedhassnfiras04Nessuna valutazione finora
- Synaptic Physiology 1Documento16 pagineSynaptic Physiology 1snowyppuppyNessuna valutazione finora
- Applsci 09 04719Documento31 pagineApplsci 09 04719RANUM WANUDYA YUNASNessuna valutazione finora
- Christensen 2016Documento8 pagineChristensen 2016Lina DarkNessuna valutazione finora
- The Circadian System: A Regulatory Feedback Network of Periphery and BrainDocumento12 pagineThe Circadian System: A Regulatory Feedback Network of Periphery and BrainOscarMavilaHerediaNessuna valutazione finora
- Perception Deco PDFDocumento8 paginePerception Deco PDFxmlbioxNessuna valutazione finora
- Margarita Zachariou Et Al - Sensory Gating and Its Modulation by Cannabinoids: Electrophysiological, Computational and Mathematical AnalysisDocumento12 pagineMargarita Zachariou Et Al - Sensory Gating and Its Modulation by Cannabinoids: Electrophysiological, Computational and Mathematical AnalysisJmasnNessuna valutazione finora
- Neurotransmission: Proceedings of the Sixth International Congress of PharmacologyDa EverandNeurotransmission: Proceedings of the Sixth International Congress of PharmacologyLiisa AhteeNessuna valutazione finora
- Neuronal Networks in Brain Function, CNS Disorders, and TherapeuticsDa EverandNeuronal Networks in Brain Function, CNS Disorders, and TherapeuticsCarl FaingoldNessuna valutazione finora
- Fractional Fourier TransformDocumento6 pagineFractional Fourier TransformLucas GallindoNessuna valutazione finora
- Finding Hidden ComponentsDocumento21 pagineFinding Hidden ComponentsLucas GallindoNessuna valutazione finora
- Discrete-Time Fourier TransformDocumento5 pagineDiscrete-Time Fourier TransformLucas GallindoNessuna valutazione finora
- Hadamard TransformDocumento4 pagineHadamard TransformLucas GallindoNessuna valutazione finora
- Disintegration TheoremDocumento3 pagineDisintegration TheoremLucas GallindoNessuna valutazione finora
- Nyquist-Shannon Sampling TheoremDocumento10 pagineNyquist-Shannon Sampling TheoremLucas GallindoNessuna valutazione finora
- Discrete Hartley TransformDocumento5 pagineDiscrete Hartley TransformLucas GallindoNessuna valutazione finora
- Dot ProductDocumento6 pagineDot ProductLucas GallindoNessuna valutazione finora
- Natural Therapies Overview Report FinalDocumento188 pagineNatural Therapies Overview Report FinalLucas GallindoNessuna valutazione finora
- Cavalieri's PrincipleDocumento4 pagineCavalieri's PrincipleLucas GallindoNessuna valutazione finora
- PragPub The Second Iteration - Teaching Kids ProgrammingDocumento30 paginePragPub The Second Iteration - Teaching Kids ProgrammingLucas Gallindo100% (1)
- Symmetry of Second DerivativesDocumento4 pagineSymmetry of Second DerivativesLucas GallindoNessuna valutazione finora
- Fast Fourier TransformDocumento10 pagineFast Fourier TransformLucas GallindoNessuna valutazione finora
- The Costs and Benefits of World-Class UniversitiesDocumento5 pagineThe Costs and Benefits of World-Class UniversitiesLucas GallindoNessuna valutazione finora
- Convolution TheoremDocumento4 pagineConvolution TheoremLucas GallindoNessuna valutazione finora
- Fourier TransformDocumento23 pagineFourier TransformLucas Gallindo100% (1)
- Fubini's TheoremDocumento6 pagineFubini's TheoremLucas GallindoNessuna valutazione finora
- Laplace TransformDocumento13 pagineLaplace TransformLucas GallindoNessuna valutazione finora
- Discrete Fourier TransformDocumento12 pagineDiscrete Fourier TransformLucas GallindoNessuna valutazione finora
- The Statistical Analysis of World Class UniversitiesDocumento21 pagineThe Statistical Analysis of World Class UniversitiesLucas GallindoNessuna valutazione finora
- Creating World-Class Universities in Developing CountriesDocumento16 pagineCreating World-Class Universities in Developing CountriesLucas GallindoNessuna valutazione finora
- Harmonic AnalysisDocumento4 pagineHarmonic AnalysisLucas GallindoNessuna valutazione finora
- Pilates Method For Women's HealthDocumento12 paginePilates Method For Women's HealthLucas GallindoNessuna valutazione finora
- Fun With The Bell CurveDocumento2 pagineFun With The Bell CurveLucas GallindoNessuna valutazione finora
- Information Aggregation, With Application To Monotone Ordering, Advocacy, and ConvivialityDocumento173 pagineInformation Aggregation, With Application To Monotone Ordering, Advocacy, and ConvivialityLucas GallindoNessuna valutazione finora
- Division by ThreeDocumento35 pagineDivision by ThreeLucas GallindoNessuna valutazione finora
- 1207.0580improving Neural Networks by Preventing Co-Adaptation of Feature DetectorsDocumento18 pagine1207.0580improving Neural Networks by Preventing Co-Adaptation of Feature DetectorsLucas GallindoNessuna valutazione finora
- Samsung Galaxy S5 Disassemble GuideDocumento9 pagineSamsung Galaxy S5 Disassemble GuideLucas GallindoNessuna valutazione finora
- Brain Facts Book PDFDocumento96 pagineBrain Facts Book PDFjlangseth100% (1)
- Accepted Manuscript: and BiologyDocumento24 pagineAccepted Manuscript: and BiologylukeNessuna valutazione finora
- Nervous Tissue-LabDocumento32 pagineNervous Tissue-LabAlexis PriceNessuna valutazione finora
- Tisu Neural Neurofisiologi Neuron Neuroglia Terminologi SarafDocumento141 pagineTisu Neural Neurofisiologi Neuron Neuroglia Terminologi SarafRainne LeeNessuna valutazione finora
- Questions Examens PhysioDocumento32 pagineQuestions Examens PhysioZeineb KammounNessuna valutazione finora
- Histology of Nervous SystemDocumento59 pagineHistology of Nervous SystemMelissa Aina Mohd YusofNessuna valutazione finora
- Atlas NeuroanatomyDocumento874 pagineAtlas NeuroanatomyBilge Yilmaz100% (3)
- Anatomy and Physiology of BrainDocumento21 pagineAnatomy and Physiology of BrainNavjot BrarNessuna valutazione finora
- 86 Research Papers Supporting The Vaccine Autism LinkDocumento69 pagine86 Research Papers Supporting The Vaccine Autism LinkDanielPerez100% (1)
- Anatomy & Physiology 1 Chapter 9 The Nervous System Flashcards - QuizletDocumento7 pagineAnatomy & Physiology 1 Chapter 9 The Nervous System Flashcards - Quizletmalenya1100% (1)
- K3. 2014 11 10 Histology of Nervous SystemDocumento84 pagineK3. 2014 11 10 Histology of Nervous SystemRadian Dipta PNessuna valutazione finora
- Chapt12 LectureDocumento197 pagineChapt12 LecturePye Antwan DelvaNessuna valutazione finora
- To Unlock The Brain's Mysteries, Purée It - New York Times Magazine - MediumDocumento10 pagineTo Unlock The Brain's Mysteries, Purée It - New York Times Magazine - MediumSebastian FloresNessuna valutazione finora
- Ch02 PowerPoints FastInternet PDFDocumento33 pagineCh02 PowerPoints FastInternet PDFskdjfNessuna valutazione finora
- Neuro Study GuideDocumento11 pagineNeuro Study GuideEllen Hennings100% (1)
- Glial Reservoir Computing: Liverpool Hope University Liverpool Hope UniversityDocumento6 pagineGlial Reservoir Computing: Liverpool Hope University Liverpool Hope UniversityMonia LandolsiNessuna valutazione finora
- Anatomy and Physiology Chapter 19 Neurological SystemDocumento38 pagineAnatomy and Physiology Chapter 19 Neurological SystemBrantNessuna valutazione finora
- Peripheral Nervous SystemDocumento50 paginePeripheral Nervous SystemaveryNessuna valutazione finora
- Sinelnikov Vol IIIDocumento395 pagineSinelnikov Vol IIIFlorescu Diana NicoletaNessuna valutazione finora
- Progress in Neurodegeneration-The Role of MetalsDocumento358 pagineProgress in Neurodegeneration-The Role of MetalsMaria Rosa100% (2)
- Introduction To NeuroanatomyDocumento49 pagineIntroduction To NeuroanatomyPratik Dhakal100% (1)
- 1 Part I Central Nervous System 4zaDocumento32 pagine1 Part I Central Nervous System 4zaapi-302883249Nessuna valutazione finora
- Histology of Synapse and Neuroglia by Dr. RoomiDocumento18 pagineHistology of Synapse and Neuroglia by Dr. RoomiMudassar Roomi100% (1)
- Cognitive NeuroscienceDocumento42 pagineCognitive NeuroscienceDiya Saher100% (1)
- 30 Scientific Studies That Demonstrate Vaccines Can Cause AutismDocumento23 pagine30 Scientific Studies That Demonstrate Vaccines Can Cause Autismazrulelias100% (6)