Documenti di Didattica
Documenti di Professioni
Documenti di Cultura
1
Caricato da
hanskd350 valutazioniIl 0% ha trovato utile questo documento (0 voti)
8 visualizzazioni2 paginePresent developments in SA production focus on a biologicalprocess based on the conversion of a renewable feedstock to SA by a biocatalyst. Biological SA production assimilates carbon dioxide(a possible environmental benefit) and is not directly dependent on crude oil. Wild strains of Actinobacillus succinogenes show promise due to its ability to naturally produce SA at a hightitre and its high acid tolerance.
Descrizione originale:
Copyright
© © All Rights Reserved
Formati disponibili
DOCX, PDF, TXT o leggi online da Scribd
Condividi questo documento
Condividi o incorpora il documento
Hai trovato utile questo documento?
Questo contenuto è inappropriato?
Segnala questo documentoPresent developments in SA production focus on a biologicalprocess based on the conversion of a renewable feedstock to SA by a biocatalyst. Biological SA production assimilates carbon dioxide(a possible environmental benefit) and is not directly dependent on crude oil. Wild strains of Actinobacillus succinogenes show promise due to its ability to naturally produce SA at a hightitre and its high acid tolerance.
Copyright:
© All Rights Reserved
Formati disponibili
Scarica in formato DOCX, PDF, TXT o leggi online su Scribd
0 valutazioniIl 0% ha trovato utile questo documento (0 voti)
8 visualizzazioni2 pagine1
Caricato da
hanskd35Present developments in SA production focus on a biologicalprocess based on the conversion of a renewable feedstock to SA by a biocatalyst. Biological SA production assimilates carbon dioxide(a possible environmental benefit) and is not directly dependent on crude oil. Wild strains of Actinobacillus succinogenes show promise due to its ability to naturally produce SA at a hightitre and its high acid tolerance.
Copyright:
© All Rights Reserved
Formati disponibili
Scarica in formato DOCX, PDF, TXT o leggi online su Scribd
Sei sulla pagina 1di 2
Title
Author
Year Published
Publication
For Introduction
Raw Materials
Products
Process
Continuous succinic acid production by Actinobacillus
succinogenes in a biofilm reactor: Steady-state metabolic flux
variation
M.F.A. Bradfield, W. Nicol
2014
Department of Chemical Engineering, University of Pretoria,
Lynnwood Road, Hatfield, 0002 Pretoria, South Africa
SA is conventionally produced from crude oil-based precursors
viaan expensive, complex process with potential environmental
costs[4]. With this in mind and the expected growth in the
market forSA, present developments in SA production focus on a
biologicalprocess based on the conversion of a renewable
feedstock to SA bya biocatalyst. Biological SA production
assimilates carbon dioxide(a possible environmental benefit) and
is not directly dependent oncrude oil. Presently, the most
favourable biocatalysts for SA produc-tion are wild strains of
Actinobacillus succinogenes [5], Mannheimiasucciniciproducens
[6], Anaerobiospirillum succiniciproducens [7] andrecombinant
strains of Escherichia coli [8], with A. succinogenesshowing
promise due to its ability to naturally produce SA at a hightitre
[1] and its high acid tolerance [9].
Actinobacillus Succinogenes
Current studies on SA production in batch and
continuousreactors reflect that A. succinogenes is a competitive
SA producer[1318]. These studies report obtained yields and byproduct distri-butions but do not attempt to reconcile the results
with the centralmetabolic network by analysing the metabolic
flux distribution.Variations in product distributions are reported in
these studiesalthough the redox implications of the variations are
not explored.Van Heerden and Nicol [19] performed a partial
metabolic analysiswhere it was shown that all continuous results
exclusively followthe pyruvate formate-lyase route. Constant
product distributionswere obtained at all the dilution rates but no
redox checks wereperformed.
Succinic acid (SA) or butanedioic acid has various
importantapplications in the pharmaceutical, food, biopolymers
and chemi-cals industry [1] and has been identified as a potential
large-scale,biomass-derived chemical by the US Department of
Energy [2,3].
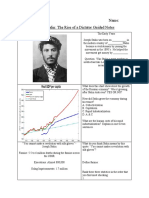
Continuous anaerobic fermentations were performed in a novel
external-recycle, biofilm reactor using d-glucose and CO2 as
carbon substrates. Succinic acid (SA) yields were found to be an
increasing function of glucose consumption with the succinic acid
to acetic acid ratio increasing from 2.4 g g1 at a glucose
consumption of 10 g L1, to 5.7 g g1 at a glucose consumption
of 50 g L1. The formic acid to acetic acid ratio decreased from
an equimolar value (0.77 g g1) at a glucose consumption of 10
g L1 to a value close to zero at 50 g L1. The highest SA yield
on glucose and highest SA titre obtained were 0.91 g g1 and
48.5 g L1 respectively. Metabolic flux analysis based on the
established C3 and C4 metabolic path-ways of Actinobacillus
succinogenes revealed that the increase in the succinate to
acetate ratio could not be attributed to the decrease in formic
acid and that an additional source of NADH was present. The
fraction of unaccounted NADH increased with glucose
consumption, suggesting that additional reducing power is
present in the medium or is provided by the activation of an
alternative metabolic pathway.
Equipment
Conclusion of
Journal
This study provides evidence that the metabolic flux distribu-tion
of A. succinogenes in a continuous biofilm reactor is a function
ofglucose consumption. More specifically, CSAincreased
continuouslywith increasing glucose consumption while the
concentrationsof the other metabolites, acetate and formate,
flattened out anddecreased to zero, respectively. Furthermore,
succinic acid pro-duction (i.e. flux to the C4metabolic pathway)
was favoured athigh values of glucose consumption, when the
cells are probablyin a maintenance or non-growth state and
therefore express dif-ferent metabolic pathways, allowing greater
yields. Values greaterthan the theoretical maximum values were
obtained for YAASA(5.70 g g1) and YGLSA(0.91 g g1). In
addition, YAAFAwas consis-tently less than 0.77 g g1and
approached zero as CFAdecreased,which suggests that either the
PDH or the FDH pathway, or a combi-nation thereof, becomes
increasingly active with increasing glucoseconsumption. These
pathways, and biomass synthesis, allow for anincrease in
YAASAdue to NADH generation, but could not account forthe total
increase, which is probably explainable by the activationof
alternative metabolic pathways that generate additional NADHor
by the presence of additional reducing power in the medium
Potrebbero piacerti anche
- The Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeDa EverandThe Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeValutazione: 4 su 5 stelle4/5 (5794)
- The Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreDa EverandThe Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreValutazione: 4 su 5 stelle4/5 (1090)
- Never Split the Difference: Negotiating As If Your Life Depended On ItDa EverandNever Split the Difference: Negotiating As If Your Life Depended On ItValutazione: 4.5 su 5 stelle4.5/5 (838)
- Hidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceDa EverandHidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceValutazione: 4 su 5 stelle4/5 (895)
- Grit: The Power of Passion and PerseveranceDa EverandGrit: The Power of Passion and PerseveranceValutazione: 4 su 5 stelle4/5 (588)
- Shoe Dog: A Memoir by the Creator of NikeDa EverandShoe Dog: A Memoir by the Creator of NikeValutazione: 4.5 su 5 stelle4.5/5 (537)
- The Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersDa EverandThe Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersValutazione: 4.5 su 5 stelle4.5/5 (344)
- Elon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureDa EverandElon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureValutazione: 4.5 su 5 stelle4.5/5 (474)
- Her Body and Other Parties: StoriesDa EverandHer Body and Other Parties: StoriesValutazione: 4 su 5 stelle4/5 (821)
- The Sympathizer: A Novel (Pulitzer Prize for Fiction)Da EverandThe Sympathizer: A Novel (Pulitzer Prize for Fiction)Valutazione: 4.5 su 5 stelle4.5/5 (120)
- The Emperor of All Maladies: A Biography of CancerDa EverandThe Emperor of All Maladies: A Biography of CancerValutazione: 4.5 su 5 stelle4.5/5 (271)
- The Little Book of Hygge: Danish Secrets to Happy LivingDa EverandThe Little Book of Hygge: Danish Secrets to Happy LivingValutazione: 3.5 su 5 stelle3.5/5 (399)
- The World Is Flat 3.0: A Brief History of the Twenty-first CenturyDa EverandThe World Is Flat 3.0: A Brief History of the Twenty-first CenturyValutazione: 3.5 su 5 stelle3.5/5 (2259)
- The Yellow House: A Memoir (2019 National Book Award Winner)Da EverandThe Yellow House: A Memoir (2019 National Book Award Winner)Valutazione: 4 su 5 stelle4/5 (98)
- Devil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaDa EverandDevil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaValutazione: 4.5 su 5 stelle4.5/5 (266)
- A Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryDa EverandA Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryValutazione: 3.5 su 5 stelle3.5/5 (231)
- Team of Rivals: The Political Genius of Abraham LincolnDa EverandTeam of Rivals: The Political Genius of Abraham LincolnValutazione: 4.5 su 5 stelle4.5/5 (234)
- AI Intelligence (Sam Charrington and Abeba Birhane)Documento2 pagineAI Intelligence (Sam Charrington and Abeba Birhane)Ethel Shammah Waoulda Acleta90% (10)
- On Fire: The (Burning) Case for a Green New DealDa EverandOn Fire: The (Burning) Case for a Green New DealValutazione: 4 su 5 stelle4/5 (73)
- The Unwinding: An Inner History of the New AmericaDa EverandThe Unwinding: An Inner History of the New AmericaValutazione: 4 su 5 stelle4/5 (45)
- AR 700-84 (Issue and Sale of Personal Clothing)Documento96 pagineAR 700-84 (Issue and Sale of Personal Clothing)ncfranklinNessuna valutazione finora
- Listo Si Kap!Documento32 pagineListo Si Kap!Bluboy100% (3)
- Names of PartnerDocumento7 pagineNames of PartnerDana-Zaza BajicNessuna valutazione finora
- Book Item 97952Documento18 pagineBook Item 97952Shairah May MendezNessuna valutazione finora
- Judgments of Adminstrative LawDocumento22 pagineJudgments of Adminstrative Lawpunit gaurNessuna valutazione finora
- The Absent Presence of Progressive Rock in The British Music Press 1968 1974 PDFDocumento33 pagineThe Absent Presence of Progressive Rock in The British Music Press 1968 1974 PDFwago_itNessuna valutazione finora
- Stalin Guided NotesDocumento2 pagineStalin Guided Notesapi-536892868Nessuna valutazione finora
- Assets Misappropriation in The Malaysian Public AnDocumento5 pagineAssets Misappropriation in The Malaysian Public AnRamadona SimbolonNessuna valutazione finora
- Mock Act 1 - Student - 2023Documento8 pagineMock Act 1 - Student - 2023Big bundahNessuna valutazione finora
- Travel Reservation August 18 For FREDI ISWANTODocumento2 pagineTravel Reservation August 18 For FREDI ISWANTOKasmi MinukNessuna valutazione finora
- AssignmentDocumento25 pagineAssignmentPrashan Shaalin FernandoNessuna valutazione finora
- Bond by A Person Obtaining Letters of Administration With Two SuretiesDocumento2 pagineBond by A Person Obtaining Letters of Administration With Two Suretiessamanta pandeyNessuna valutazione finora
- BRUNOISEDocumento1 paginaBRUNOISEMelchor Dagandan Toylo JfyNessuna valutazione finora
- Alpha Beta Gamma Delta Epsilon Zeta Eta Theta Iota Kappa Lambda MuDocumento3 pagineAlpha Beta Gamma Delta Epsilon Zeta Eta Theta Iota Kappa Lambda MulanNessuna valutazione finora
- The Mental Health Act 2018 Tested by High Court of UgandaDocumento7 pagineThe Mental Health Act 2018 Tested by High Court of UgandaLDC Online ResourcesNessuna valutazione finora
- Đề Cương CK - QuestionsDocumento2 pagineĐề Cương CK - QuestionsDiệu Phương LêNessuna valutazione finora
- About ArevaDocumento86 pagineAbout ArevaAbhinav TyagiNessuna valutazione finora
- Appleby, Telling The Truth About History, Introduction and Chapter 4Documento24 pagineAppleby, Telling The Truth About History, Introduction and Chapter 4Steven LubarNessuna valutazione finora
- Tucker Travis-Reel Site 2013-1Documento2 pagineTucker Travis-Reel Site 2013-1api-243050578Nessuna valutazione finora
- Code of Civil Procedure: Recording of Evidence by Court/Court CommissionerDocumento12 pagineCode of Civil Procedure: Recording of Evidence by Court/Court CommissionercahirenrajaNessuna valutazione finora
- Ingles Junio 17 CanariasDocumento5 pagineIngles Junio 17 CanariaskinkirevolutionNessuna valutazione finora
- Ais CH5Documento30 pagineAis CH5MosabAbuKhater100% (1)
- People Vs SolangonDocumento7 paginePeople Vs SolangonChris YumNessuna valutazione finora
- Anderson v. Eighth Judicial District Court - OpinionDocumento8 pagineAnderson v. Eighth Judicial District Court - OpinioniX i0Nessuna valutazione finora
- A Documentary Report of Work Immersion Undertaken at The City Mayor'S OfficeDocumento17 pagineA Documentary Report of Work Immersion Undertaken at The City Mayor'S OfficeHamdan AbisonNessuna valutazione finora
- Tiktok PresentationDocumento17 pagineTiktok Presentationapi-681531475Nessuna valutazione finora
- Personal Data Breach Notifications in The GDPRDocumento17 paginePersonal Data Breach Notifications in The GDPRgschandel3356Nessuna valutazione finora
- Balkan Languages - Victor FriedmanDocumento12 pagineBalkan Languages - Victor FriedmanBasiol Chulev100% (1)
- Company Analysis of Axis BankDocumento66 pagineCompany Analysis of Axis BankKirti Bhite100% (1)