Documenti di Didattica
Documenti di Professioni
Documenti di Cultura
Electron Spin Resonance Spectroscopy: Objective
Caricato da
coolbro920 valutazioniIl 0% ha trovato utile questo documento (0 voti)
28 visualizzazioni9 pagineThis document describes an experiment to determine the g-factor of an electron using electron spin resonance spectroscopy with DPPH as the sample. The g-factor was calculated for resonance frequencies of 14 MHz, 16 MHz, and 18 MHz. The calculated g-factor values were 1.755, 2.191, and 2.35 respectively, which are consistent with the accepted value of 2.002319. Care must be taken to properly shield the ESR setup and not overheat the Helmholtz coils when performing this experiment.
Descrizione originale:
A detailed description about ESR
Titolo originale
ESR
Copyright
© © All Rights Reserved
Formati disponibili
PDF, TXT o leggi online da Scribd
Condividi questo documento
Condividi o incorpora il documento
Hai trovato utile questo documento?
Questo contenuto è inappropriato?
Segnala questo documentoThis document describes an experiment to determine the g-factor of an electron using electron spin resonance spectroscopy with DPPH as the sample. The g-factor was calculated for resonance frequencies of 14 MHz, 16 MHz, and 18 MHz. The calculated g-factor values were 1.755, 2.191, and 2.35 respectively, which are consistent with the accepted value of 2.002319. Care must be taken to properly shield the ESR setup and not overheat the Helmholtz coils when performing this experiment.
Copyright:
© All Rights Reserved
Formati disponibili
Scarica in formato PDF, TXT o leggi online su Scribd
0 valutazioniIl 0% ha trovato utile questo documento (0 voti)
28 visualizzazioni9 pagineElectron Spin Resonance Spectroscopy: Objective
Caricato da
coolbro92This document describes an experiment to determine the g-factor of an electron using electron spin resonance spectroscopy with DPPH as the sample. The g-factor was calculated for resonance frequencies of 14 MHz, 16 MHz, and 18 MHz. The calculated g-factor values were 1.755, 2.191, and 2.35 respectively, which are consistent with the accepted value of 2.002319. Care must be taken to properly shield the ESR setup and not overheat the Helmholtz coils when performing this experiment.
Copyright:
© All Rights Reserved
Formati disponibili
Scarica in formato PDF, TXT o leggi online su Scribd
Sei sulla pagina 1di 9
Electron Spin Resonance Spectroscopy
Soumik Ghosh (11MS007)
Satyajit Roy (11MS063)
Rishav Khare (11MS034)
Sayandip Dhara (11MS016)
DPS 4th year
Objective
To find out the 'g' value for electron using 2,2-Diphenyl-1-picrylhydrazyl (DPPH), (free radical,
95%) as a sample by ESR spectroscopy.
Theory
'Resonance' is a widely studied phenomenon both in classical as well as quantum physics.
The classical concept of a strong, effective coupling of interacting oscillators that occurs when
they share an identical frequency of oscillation. Electron Spin Resonance or ESR (also known as
Electron Paramagnetic resonance) is such a spectroscopic technique that makes use of unpaired
electron in the system under observation.
The phenomenon of magnetic resonance can be explained using classical picture of
magnetic moment and applied eld interaction. A particle with a magnetic moment placed in a
uniform magnetic field of intensity H0 will precess around H0 with an angular Larmor frequency
0 , where;
w0 = g(e/2me)H0
g, or the Land g-factor, depends on the state of the electron in the atom and is given by :
g = 3/2 + {S(S+1)-L(L+1)}/2J(J+1)
In case of anions in a crystal, the electronic behaviour is modified by the environment and
the g-factor may differ from the Land g-factor. This effective g-factor is also known as the
spectroscopic splitting factor. The theoretical value of the Land g-factor for an electron is very
close to 2 (The actual value of the g-factor changes for different particles used).
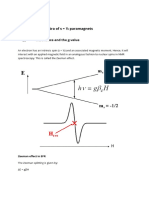
Now let us introduce an additional weak magnetic eld oriented in the xy plane and
rotating about the z axis (assuming Larmor precession along the z axis) with an angular frequency
1. As a result there will be an interaction between the precessing moment and the external field.
If the frequency 1 is different from 0, the angle between the field H1 and the magnetic moment
will continuously change so to average out their interaction will to zero. However, if 1 is equal
to 0 the angle between and H1 is maintained and net interaction is effective. If we look at the
system in a reference frame that is rotating about the z axis with the angular velocity 0 then the
spin will appear to make an angle =(90
0
-) with H1 and according to the previous argument will
start to precess (in the rotating frame) about H1.
This corresponds to a "nutation" and a consequent change of the angle which implies a change
in the potential energy of the particle in the magnetic field. The change in is the classical analogy
to a transition between sublevels with different m. We see that such transitions may take place
only if the rotating field has an angular frequency 1 = 0. Now suppose that the intrinsic spin
angular momentum of the electron S couples with the orbital angular momentum of electron L
to give a resultant angular momentum J. If no external field is applied on the system, the states
with different values of ml and ms are degenerate. But, on application of external magnetic field
(say H0), the J+1 magnetic sublevels are split by equal energy difference 4E, such that,
4E = g0H0
Where 0 is the Bohr's Magneton.
Now let us consider a situation where the particle is subjected to a perturbation by an alternating
magnetic field with a frequency 1 such that the quantum h1 is exactly the same as the difference
between the levels 4E, and the direction of the alternating field is perpendicular to the direction
of the static magnetic field. Then there will be induced transitions between neighboring sublevels
according to the selection rules 4m = 1 for magnetic dipolar radiation. The condition for
resonance is, therefore given by:
4E = g0H0 = hv0 = hv1
Then the g-factor can be easily calculated by:
g = hv0/0H0
The sample used to carry out the experiment is 2,2-Diphenyl-1-picrylhydrazyl (DPPH) (free radical,
95%). The chemical structure of the sample is given below.
Working Formula
Magnetic field at the center of the Helmholtz Coil is given by :
H = 32nI/(10a125) Gauss
where, `n' is the number of turns in the Helmholtz coil.(n=500)
a is the radius of the Helmholtz coil.(a =7.7 cm)
I is the current (in amp) flowing through the Helmholtz coils.
Now the peak to peak field is given by:
HPP = 22H gauss
From this the magnetic field for resonance is given by:
H0 = Q.HPP/P = 2232nQI /10125aP Gauss
Now for resonance the condition we obtained earlier is :
4E= g0H0 = h0
or, g = h0/0H0
where, 0 is the resonance frequency.
Experimental Results and Analysis
For Resonance frequency 0 = 14MHz
P = 132 V
I (mA) 2Q
102 84
112 76
122 68
131 64
143 56
152 52
164 48
174 48
184 44
195 40
204 40
Corresponding Graph
For Resonance frequency 0 = 16MHz
P = 132 V
I (mA) 2Q
101 84
112 76
125 68
139 60
151 56
163 52
170 48
188 48
199 44
210 40
For Resonance frequency 0 = 18MHz
P = 132 V
I (mA) 2Q
102 84
112 76
123 68
135 64
148 56
160 52
174 48
186 44
196 44
Calculation of 'g' factor
The Planck's constant h = 6.625x10
-27
erg-s
The value of 0 = 0.927x10
-20
erg/Gauss
1. For Resonance frequency 0 = 14MHz
From the slope of the graph,
QI = 4.56315 V-Amp
The resonance frequency is given by,
H0 = (2232nQI)/(10125aP) =5.70 Gauss
The Value of 'g' is
g = h0/0H0 =
6.62510
27
1410
6
0.92710
20
5.70
= 1.755
Thus calculated value of 'g' is =1.755
2. For Resonance frequency 0 = 16MHz
From the slope of the graph,
QI = 4.17 V-Amp
The resonance frequency is given by,
H0 = (2232nQI)/(10125aP) =5.217
The Value of 'g' is
g = h0/0H0 =
6.62510
27
1610
6
0.92710
20
5.217
= 2.191
Thus calculated value of 'g' is = 2.191
3. For Resonance frequency 0 = 18MHz
From the slope of the graph,
QI = 4.3768 V-Amp
The resonance frequency is given by,
H0 = (2232nQI)/(10125aP) =5.47
The Value of 'g' is
g = h0/0H0 =
6.62510
27
1810
6
0.92710
20
5.47
= 2.35
Thus calculated value of 'g' is = 2.35
Result
Thus the value of 'g' factor obtained for three frequency values 14 MHz ,16MHz and
18 MHz are 1.755,2.191 and 2.35 respectively.
Discussion
Thus we have carried out the experiment and the obtained value of 'g' is quite
satisfactory. The value of 'g' determined using QED is 2.002319 which is quite in
range of our experimentally obtained values. While carrying out the experiment,
four things we should keep in mind first :
1. The ESR setup must be shielded from external magnetic field or electric
field sources that interfere with the magnetic field used in the experiment.
2. High currents (~200 mA) should not be allowed to pass through the
helmholtz coil for a long time. This may cause unnecessary heat and
thereby damage can occur.
3. If the peaks do not coincide on the x-scale, the sinusoidal wave form of
the mains voltage should be checked which may be distorted due to overloading
of the main line due to other heavy gadgets working on the same
line.
4. While finding the resonance frequency, the resolution of the X channel
should be kept very low so that no peak can be observed. This will help
us to detect the beat patterns very easily.
Potrebbero piacerti anche
- Advances in Structure Research by Diffraction Methods: Fortschritte der Strukturforschung mit BeugungsmethodenDa EverandAdvances in Structure Research by Diffraction Methods: Fortschritte der Strukturforschung mit BeugungsmethodenR. BrillNessuna valutazione finora
- ESR1Documento4 pagineESR1salman suhailNessuna valutazione finora
- Interactions between Electromagnetic Fields and Matter: Vieweg Tracts in Pure and Applied PhysicsDa EverandInteractions between Electromagnetic Fields and Matter: Vieweg Tracts in Pure and Applied PhysicsNessuna valutazione finora
- IsotropicDocumento7 pagineIsotropicSiddiqui M. M.Nessuna valutazione finora
- Feynman Lectures Simplified 2C: Electromagnetism: in Relativity & in Dense MatterDa EverandFeynman Lectures Simplified 2C: Electromagnetism: in Relativity & in Dense MatterNessuna valutazione finora
- Untitled PDFDocumento5 pagineUntitled PDFriyaparnaNessuna valutazione finora
- Resonance Enhancement in Laser-Produced Plasmas: Concepts and ApplicationsDa EverandResonance Enhancement in Laser-Produced Plasmas: Concepts and ApplicationsNessuna valutazione finora
- Electron Spin Resonance Spectroscopy Calulating Land' Eg FactorDocumento5 pagineElectron Spin Resonance Spectroscopy Calulating Land' Eg FactorgjdapromiseNessuna valutazione finora
- EsrDocumento7 pagineEsrNishan RanabhatNessuna valutazione finora
- D.D.U. Gorakhpur University: (Physics Department)Documento15 pagineD.D.U. Gorakhpur University: (Physics Department)Narendra ChaurasiaNessuna valutazione finora
- ESR Lab ReportDocumento4 pagineESR Lab ReportVargheseAbinNessuna valutazione finora
- Cycle-III Expt 9 ESR - Lande G FactorDocumento6 pagineCycle-III Expt 9 ESR - Lande G FactorSwapnil BabeleNessuna valutazione finora
- Electron Spin Resonance Spectroscopy: Calculating The Land e G-Factor For A Free ElectronDocumento5 pagineElectron Spin Resonance Spectroscopy: Calculating The Land e G-Factor For A Free ElectronRahul DevarakondaNessuna valutazione finora
- Groener QuantumPaperDocumento5 pagineGroener QuantumPaperJulian David Henao EscobarNessuna valutazione finora
- EsrDocumento7 pagineEsrBean GemboelzNessuna valutazione finora
- A System To Convert Gravitational Energy Directly Into Electrical EnergyDocumento17 pagineA System To Convert Gravitational Energy Directly Into Electrical EnergyFran De AquinoNessuna valutazione finora
- Grav Radiation ProjectDocumento21 pagineGrav Radiation ProjectmossmanjNessuna valutazione finora
- Electron Spin Resonance ExperimentDocumento6 pagineElectron Spin Resonance ExperimentArooj Mukarram100% (1)
- G.V. Stupakov and M. S. Zolotorev - Ponderomotive Laser Acceleration and Focusing in Vacuum For Generation of Attosecond Electron BunchesDocumento4 pagineG.V. Stupakov and M. S. Zolotorev - Ponderomotive Laser Acceleration and Focusing in Vacuum For Generation of Attosecond Electron BunchesPocxaNessuna valutazione finora
- Kin Ect I Ccu An Tum Gravity TheoryDocumento11 pagineKin Ect I Ccu An Tum Gravity TheoryLuis Alberto MiglioreroNessuna valutazione finora
- Scaling of The Hot Electron Temperature and Laser Absorption in Fast IgnitionDocumento20 pagineScaling of The Hot Electron Temperature and Laser Absorption in Fast IgnitionKevin M GeorgeNessuna valutazione finora
- Dirac Equation ElectronDocumento12 pagineDirac Equation ElectronFranciscoNessuna valutazione finora
- Atomic & Molecular Spectroscopy Lab: Instruction ManualDocumento20 pagineAtomic & Molecular Spectroscopy Lab: Instruction Manualbhuppi KumarNessuna valutazione finora
- A New Model For W, Z, Higgs Bosons Masses Calculation and Validation Tests Based On The Dual Ginzburg-Landau TheoryDocumento28 pagineA New Model For W, Z, Higgs Bosons Masses Calculation and Validation Tests Based On The Dual Ginzburg-Landau TheoryengineeringtheimpossibleNessuna valutazione finora
- Electron Spin Resnance: 1 ObjectivesDocumento9 pagineElectron Spin Resnance: 1 ObjectivesKunal GargNessuna valutazione finora
- Tulipx - caltech.edu/groups/ch6/manual/Ch6-7EPR - Doc: Electron Paramagnetic Resonance (EPR) SpectrosDocumento15 pagineTulipx - caltech.edu/groups/ch6/manual/Ch6-7EPR - Doc: Electron Paramagnetic Resonance (EPR) SpectrosvgvijuNessuna valutazione finora
- Wenxian Zhang Et Al - Coherent Spin Mixing Dynamics in A Spin-1 Atomic CondensateDocumento5 pagineWenxian Zhang Et Al - Coherent Spin Mixing Dynamics in A Spin-1 Atomic CondensatePomac232Nessuna valutazione finora
- Magnetic Resonance SpectrosDocumento10 pagineMagnetic Resonance SpectrosJake DysonNessuna valutazione finora
- Stark Effect in Parabolic Quantum DotDocumento6 pagineStark Effect in Parabolic Quantum DottransfinitumNessuna valutazione finora
- Electron Spin Resonance: Theory and ApplicationsDocumento25 pagineElectron Spin Resonance: Theory and ApplicationswexlerNessuna valutazione finora
- Shapiro Steps in Josephson JunctionsDocumento19 pagineShapiro Steps in Josephson JunctionsChristopher WallaceNessuna valutazione finora
- GraphDocumento9 pagineGraphFarazNessuna valutazione finora
- Condensed Matter 3Documento9 pagineCondensed Matter 3Abhisha RameshNessuna valutazione finora
- Rubidium Hyperfine StructureDocumento4 pagineRubidium Hyperfine StructureKapila Wijayaratne100% (2)
- Expt 15Documento10 pagineExpt 15rameshamruthaluriNessuna valutazione finora
- Pulsed NMR Grad LabDocumento3 paginePulsed NMR Grad LabSumith SaleheenNessuna valutazione finora
- Lecture 14: Time-Dependent Perturbation Theory: Correspondence Principle + Sudden Approximation (11/10/2005)Documento6 pagineLecture 14: Time-Dependent Perturbation Theory: Correspondence Principle + Sudden Approximation (11/10/2005)bgiangre8372Nessuna valutazione finora
- Malik2012 PDFDocumento6 pagineMalik2012 PDFManendra ChaudhryNessuna valutazione finora
- Actoydrazsing Electron Spin Resonance SpectrometerDocumento4 pagineActoydrazsing Electron Spin Resonance SpectrometerSanketDesaiNessuna valutazione finora
- High-Field Laser Physics: ETH Zürich Spring Semester 2010 H. R. ReissDocumento34 pagineHigh-Field Laser Physics: ETH Zürich Spring Semester 2010 H. R. ReissjasmonNessuna valutazione finora
- Quantum Plasma Effects in The Classical Regime: PACS Numbers: 52.27.-h, 52.27.Gr, 67.57.LmDocumento10 pagineQuantum Plasma Effects in The Classical Regime: PACS Numbers: 52.27.-h, 52.27.Gr, 67.57.LmMubashar IqbalNessuna valutazione finora
- Inducing A Magnetic Monopole With Topological Surface StatesDocumento9 pagineInducing A Magnetic Monopole With Topological Surface StatesMike WestfallNessuna valutazione finora
- PH 3001 Tute - SolutionsDocumento59 paginePH 3001 Tute - Solutionsrandima fernandoNessuna valutazione finora
- K. S. Yi and J. J. Quinn - Charge and Spin Response of The Spin-Polarized Electron GasDocumento4 pagineK. S. Yi and J. J. Quinn - Charge and Spin Response of The Spin-Polarized Electron GasYamcsaNessuna valutazione finora
- Intensity Dependence of The Photoelectric Effect Induced by A Circularly Polarized Laser BeamDocumento5 pagineIntensity Dependence of The Photoelectric Effect Induced by A Circularly Polarized Laser BeamsaaedkhalidNessuna valutazione finora
- How To Measure Magnetic Flux With A Single Position Measurement?Documento5 pagineHow To Measure Magnetic Flux With A Single Position Measurement?liubingxyNessuna valutazione finora
- SpinDocumento21 pagineSpinRiyan AngelaNessuna valutazione finora
- C2 Zeeman Effect PDFDocumento9 pagineC2 Zeeman Effect PDFShweta SridharNessuna valutazione finora
- 4a1a 1348Documento4 pagine4a1a 1348guillhermeNessuna valutazione finora
- Experiment 5: Study of Hysteresis in Ferromagnetic SubstancesDocumento4 pagineExperiment 5: Study of Hysteresis in Ferromagnetic Substancescoolbro92Nessuna valutazione finora
- Manual Plancks ConstantDocumento5 pagineManual Plancks ConstantMonster DarkNessuna valutazione finora
- Semiconductor Nano RingsSemiconductorNanoringsDocumento8 pagineSemiconductor Nano RingsSemiconductorNanoringsZsolt SzakácsNessuna valutazione finora
- Numerical Analysis ProjectDocumento24 pagineNumerical Analysis ProjectMoshe DamkaniNessuna valutazione finora
- Simulado - 2 IPhODocumento12 pagineSimulado - 2 IPhOleonardo sathlerNessuna valutazione finora
- Hong 1999Documento4 pagineHong 1999yalocim666Nessuna valutazione finora
- Rydberg States and Momentum Bifurcation in Tunnel Ionization With Elliptically Polarized LightDocumento4 pagineRydberg States and Momentum Bifurcation in Tunnel Ionization With Elliptically Polarized LightBigMigTxNessuna valutazione finora
- P2214 Homework 14 Solutions - Spring 2011Documento7 pagineP2214 Homework 14 Solutions - Spring 2011calcyeeNessuna valutazione finora
- Deconvolution XPS SpectraDocumento5 pagineDeconvolution XPS SpectraLz Huerta100% (1)
- Wen Yang and Kai Chang - Intrinsic Spin Hall Effect Induced by Quantum Phase Transition in HgCdTe Quantum WellsDocumento5 pagineWen Yang and Kai Chang - Intrinsic Spin Hall Effect Induced by Quantum Phase Transition in HgCdTe Quantum WellsGreamxxNessuna valutazione finora
- Approximation Methods For Stationary States: 7.1 Time-Independent Perturbation TheoryDocumento16 pagineApproximation Methods For Stationary States: 7.1 Time-Independent Perturbation TheoryMarcelo MachadoNessuna valutazione finora
- 11B Exam Practice: Suggested Calculation: F FDocumento3 pagine11B Exam Practice: Suggested Calculation: F FsalmaNessuna valutazione finora
- Datasheet PDFDocumento8 pagineDatasheet PDFShohag MiaNessuna valutazione finora
- Ece III Network Analysis (10es34) NotesDocumento111 pagineEce III Network Analysis (10es34) NotesGautam Sharma80% (10)
- Motor Types NXP NotesDocumento4 pagineMotor Types NXP NotesdaveNessuna valutazione finora
- Busbar Eet414Documento34 pagineBusbar Eet414yahyeNessuna valutazione finora
- Trio 27Documento0 pagineTrio 27Cristi GrosuNessuna valutazione finora
- WIZ SeriesDocumento4 pagineWIZ SeriesJon HamonNessuna valutazione finora
- Module 3 TorsionDocumento13 pagineModule 3 TorsionJay LopezNessuna valutazione finora
- US20190295733A1 - Pasma Compression Fusion DeviceDocumento8 pagineUS20190295733A1 - Pasma Compression Fusion DevicedaveklodavekloNessuna valutazione finora
- Non Newtonian FluidsDocumento31 pagineNon Newtonian Fluidsikechukwujoel8Nessuna valutazione finora
- 1 s2.0 S0306261918311140 MainDocumento11 pagine1 s2.0 S0306261918311140 MainAllal BouzidNessuna valutazione finora
- 3sun M40Documento2 pagine3sun M40Anselmo Aguado cortesNessuna valutazione finora
- Grade 6 Science Term 3 Exam Table of Specification: Unit Objective(s) Level Questions PercentageDocumento2 pagineGrade 6 Science Term 3 Exam Table of Specification: Unit Objective(s) Level Questions PercentageMazanda YalinduaNessuna valutazione finora
- Fundamental Concepts in Substation Design PDFDocumento26 pagineFundamental Concepts in Substation Design PDFvishalkumarNessuna valutazione finora
- 17a03g - Mosfet - DualDocumento5 pagine17a03g - Mosfet - DualEletronica01 - BLUEVIXNessuna valutazione finora
- CATALOGO EVOLIS 24kvDocumento80 pagineCATALOGO EVOLIS 24kvOgaihtsantosNessuna valutazione finora
- Thermo NotesDocumento12 pagineThermo NotesSamriddha Das GuptaNessuna valutazione finora
- 2CDS211001R0324 sh201 c32Documento4 pagine2CDS211001R0324 sh201 c32Jean Luc FrancisNessuna valutazione finora
- Electrical System A40F Copy Prosis - 3.4.0.12Documento857 pagineElectrical System A40F Copy Prosis - 3.4.0.12Bontor Tua100% (3)
- Performance Analysis of Multilevel Inverter Using Matlab/SimulinkDocumento63 paginePerformance Analysis of Multilevel Inverter Using Matlab/Simulinkvinayak jith100% (1)
- Selection Calculations For Motors PDFDocumento17 pagineSelection Calculations For Motors PDFbibhansuNessuna valutazione finora
- Fundamentals of Electronics - Awais YasinDocumento185 pagineFundamentals of Electronics - Awais YasinAsif HameedNessuna valutazione finora
- SF 17 Sect 14 ALL WebDocumento94 pagineSF 17 Sect 14 ALL WebcachiletNessuna valutazione finora
- E&I QAP's - Submitted To Contractor HZADocumento33 pagineE&I QAP's - Submitted To Contractor HZASuraj ShettyNessuna valutazione finora
- Unit 11 - Paper 1 and 2 HLDocumento13 pagineUnit 11 - Paper 1 and 2 HLUncharted FireNessuna valutazione finora
- Experimental and Numerical Investigations On The Leakage Flow Characteristics of The Labyrinth Brush SealDocumento10 pagineExperimental and Numerical Investigations On The Leakage Flow Characteristics of The Labyrinth Brush SealНиколайNessuna valutazione finora
- Department of Computer Engineering 22215 EEC MCQ (Elements of Electrical Engineering)Documento34 pagineDepartment of Computer Engineering 22215 EEC MCQ (Elements of Electrical Engineering)Viraj TiwareNessuna valutazione finora
- Unit 1 Force Systems PDFDocumento17 pagineUnit 1 Force Systems PDFpeter vanderNessuna valutazione finora
- Waves 3 by Shyam Mohan BhaiyaDocumento17 pagineWaves 3 by Shyam Mohan BhaiyaArmaan GarnayakNessuna valutazione finora
- Description Mechanical Dimensions: 6.8V To 400V GPP Transient Voltage SuppressorsDocumento4 pagineDescription Mechanical Dimensions: 6.8V To 400V GPP Transient Voltage SuppressorsRabshaqaNessuna valutazione finora
- Summary and Interpretation of Reality TransurfingDa EverandSummary and Interpretation of Reality TransurfingValutazione: 5 su 5 stelle5/5 (5)
- A Brief History of Time: From the Big Bang to Black HolesDa EverandA Brief History of Time: From the Big Bang to Black HolesValutazione: 4 su 5 stelle4/5 (2193)
- Giza: The Tesla Connection: Acoustical Science and the Harvesting of Clean EnergyDa EverandGiza: The Tesla Connection: Acoustical Science and the Harvesting of Clean EnergyNessuna valutazione finora
- Dark Matter and the Dinosaurs: The Astounding Interconnectedness of the UniverseDa EverandDark Matter and the Dinosaurs: The Astounding Interconnectedness of the UniverseValutazione: 3.5 su 5 stelle3.5/5 (69)
- Knocking on Heaven's Door: How Physics and Scientific Thinking Illuminate the Universe and the Modern WorldDa EverandKnocking on Heaven's Door: How Physics and Scientific Thinking Illuminate the Universe and the Modern WorldValutazione: 3.5 su 5 stelle3.5/5 (64)
- A Beginner's Guide to Constructing the Universe: The Mathematical Archetypes of Nature, Art, and ScienceDa EverandA Beginner's Guide to Constructing the Universe: The Mathematical Archetypes of Nature, Art, and ScienceValutazione: 4 su 5 stelle4/5 (51)
- The Beauty of Falling: A Life in Pursuit of GravityDa EverandThe Beauty of Falling: A Life in Pursuit of GravityNessuna valutazione finora
- Lost in Math: How Beauty Leads Physics AstrayDa EverandLost in Math: How Beauty Leads Physics AstrayValutazione: 4.5 su 5 stelle4.5/5 (125)
- Quantum Spirituality: Science, Gnostic Mysticism, and Connecting with Source ConsciousnessDa EverandQuantum Spirituality: Science, Gnostic Mysticism, and Connecting with Source ConsciousnessValutazione: 4 su 5 stelle4/5 (6)
- The End of Everything: (Astrophysically Speaking)Da EverandThe End of Everything: (Astrophysically Speaking)Valutazione: 4.5 su 5 stelle4.5/5 (157)
- Midnight in Chernobyl: The Story of the World's Greatest Nuclear DisasterDa EverandMidnight in Chernobyl: The Story of the World's Greatest Nuclear DisasterValutazione: 4.5 su 5 stelle4.5/5 (410)
- Bedeviled: A Shadow History of Demons in ScienceDa EverandBedeviled: A Shadow History of Demons in ScienceValutazione: 5 su 5 stelle5/5 (5)
- Vibration and Frequency: How to Get What You Want in LifeDa EverandVibration and Frequency: How to Get What You Want in LifeValutazione: 4.5 su 5 stelle4.5/5 (13)
- The Power of Eight: Harnessing the Miraculous Energies of a Small Group to Heal Others, Your Life, and the WorldDa EverandThe Power of Eight: Harnessing the Miraculous Energies of a Small Group to Heal Others, Your Life, and the WorldValutazione: 4.5 su 5 stelle4.5/5 (54)
- The 60 Minute Quantum Physics Book: Science Made Easy For Beginners Without Math And In Plain Simple EnglishDa EverandThe 60 Minute Quantum Physics Book: Science Made Easy For Beginners Without Math And In Plain Simple EnglishValutazione: 4.5 su 5 stelle4.5/5 (4)
- Quantum Physics: What Everyone Needs to KnowDa EverandQuantum Physics: What Everyone Needs to KnowValutazione: 4.5 su 5 stelle4.5/5 (49)
- The Beginning of Infinity: Explanations That Transform the WorldDa EverandThe Beginning of Infinity: Explanations That Transform the WorldValutazione: 5 su 5 stelle5/5 (60)
- The Magick of Physics: Uncovering the Fantastical Phenomena in Everyday LifeDa EverandThe Magick of Physics: Uncovering the Fantastical Phenomena in Everyday LifeNessuna valutazione finora
- The Holographic Universe: The Revolutionary Theory of RealityDa EverandThe Holographic Universe: The Revolutionary Theory of RealityValutazione: 4.5 su 5 stelle4.5/5 (78)
- Let There Be Light: Physics, Philosophy & the Dimensional Structure of ConsciousnessDa EverandLet There Be Light: Physics, Philosophy & the Dimensional Structure of ConsciousnessValutazione: 4.5 su 5 stelle4.5/5 (57)
- The Illustrated Theory of Everything: The Origin and Fate of the UniverseDa EverandThe Illustrated Theory of Everything: The Origin and Fate of the UniverseValutazione: 5 su 5 stelle5/5 (1)
- Packing for Mars: The Curious Science of Life in the VoidDa EverandPacking for Mars: The Curious Science of Life in the VoidValutazione: 4 su 5 stelle4/5 (1396)
- AP Physics 1 Premium, 2024: 4 Practice Tests + Comprehensive Review + Online PracticeDa EverandAP Physics 1 Premium, 2024: 4 Practice Tests + Comprehensive Review + Online PracticeNessuna valutazione finora