Documenti di Didattica
Documenti di Professioni
Documenti di Cultura
Concentration Calcs With Answers
Caricato da
Khadija KarimDescrizione originale:
Copyright
Formati disponibili
Condividi questo documento
Condividi o incorpora il documento
Hai trovato utile questo documento?
Questo contenuto è inappropriato?
Segnala questo documentoCopyright:
Formati disponibili
Concentration Calcs With Answers
Caricato da
Khadija KarimCopyright:
Formati disponibili
1
Chem!stry
Name: ________________ ( )
Class: _______
Date: ____ / ____ / ____
Concentration
Concentration is a measure of the amount of a solute
dissolved in a solvent to make one unit volume of a
solution.
Unit volume: dm
3
(1 dm
3
=1000 cm
3
)
Unit of Concentration: mol/dm or g/dm
concentration in mol/dm
3
=
number of moles of solute
volume of solution measured in dm
3
concentration in g/dm
3
=
mass of solute measured in g
volume of solution measured in dm
3
Concentration in g/dm
3
can be changed into mol/dm
3
, and vice versa, using the following
relationship:
concentration in mol/dm
3
=
concentration in g/dm
3
relative molecular mass of the solute
Exercises:
1. Calculate the concentration of the following solution, in mol/dm
3
:
(a) 0.8 mol of H
2
SO
4
in 2 dm
3
of solution
(b) 0.25 mol of HCl in 250 cm
3
of solution
(c) 0.02 mol of NaOH in 50 cm
3
of solution
(d) 29.25g of NaCl in 4 dm
3
of solution
(e) 9.8g of H
2
SO
4
in 250 cm
3
of solution
2
2. Calculate the number of moles of:
(a) NaOH in 2 dm
3
of 0.5 mol/dm
3
NaOH
(b) H
+
in 4 dm
3
of 0.5 mol/dm
3
of H
2
SO
4
(c) KOH in 25 cm
3
of 0.4 mol/dm
3
of KOH
(d) HNO
3
in 100cm
3
of 6.3 g/dm
3
of HNO
3
(e) Na
+
in 500 cm
3
of 5.3 g/dm
3
of Na
2
CO
3
3. A solution of sodium hydroxide has a concentration of 0.2 mol/dm
3
. Calculate the
concentration of the solution in g/dm
3
.
4. A solution is made by dissolving 4.9g of sulphuric acid to make 1000 cm
3
solution.
Calculate the concentration of the solution in mol/dm
3
.
5. 200 cm
3
of solution contains 3.65g of HCl. Calculate the concentration in
(a) g/dm
3
(b) mol/dm
3
6. Calculate the number of moles of sulphuric acid in 25.0 cm
3
of 2.0 mol/dm
3
sulphuric solution.
7. What mass of potassium hydroxide must be dissolved in 1000 cm
3
to make a
250 cm
3
of solution of concentration 0.4 mol/dm
3
?
8. 50cm
3
of hydrogen peroxide, which has a concentration of 2.0 mol/dm
3
is
decomposed. The equation of the reaction is given below:
2H
2
O
2 (aq)
2H
2
O
(l)
+ O
2 (g)
(a) How many moles of hydrogen peroxide are there in 50 cm
3
of the solution?
(b) How many moles of oxygen are formed when 50 cm
3
of the hydrogen
peroxide decomposes?
(c) Calculate the volume of oxygen produced when 50 cm
3
of hydrogen peroxide
solution decomposes.
9. 30 cm
3
of 0.1 mol/dm
3
NaOH reacted completely with 25.0 cm
3
of H
2
SO
4
in a
titration. Calculate the concentration of H
2
SO
4
in:
(a) mol/dm
3
(b) g/dm
3
10. 18.0 cm
3
of 0.2 mol/dm
3
sulphuric acid reacts with 24.0 cm
3
of potassium hydroxide
solution to form salt and water. Calculate the concentration of the potassium
hydroxide solution in:
(a) mol/dm
3
(b) g/dm
3
3
11. Calculate the volume of 0.5 mol/dm
3
solution of sodium hydroxide required to
neutralize 24.7 cm
3
of 0.1 mol/dm
3
sulphuric acid.
12. What volume of hydrochloric acid of concentration 0.25 mol/dm
3
is required to
neutralize 22.5 cm
3
of 0.40 mol/dm
3
sodium hydroxide solution?
13. The table shows the results of experiments to measure the volume of dilute
sulphuric acid (column 1) needed to neutralize the mass of sodium hydroxide
(column 2) dissolved in the volume of water (column 3):
Experiment
Column 1
Volume of sulphuric
acid required / cm
3
Column 2
Mass of sodium
hydroxide / g
Column 3
Volume of water /
cm
3
1
2
3
4
20
40
40
?
1
2
2
1
20
40
20
40
From these results, what volume of sulphuric acid is needed to neutralize the
sodium hydroxide in the fourth experiment?
(A) 5 cm
3
(B) 10 cm
3
(C) 20 cm
3
(D) 40 cm
3
(E) 80 cm
3
4
Answers to Concentration Calculations
1.
a) 0.8 mol of H
2
SO
4
dissolved in 2.0 dm
3
=0.8 (1 2) =0.400 mol/dm
3
b) 0.25 mol of HCl dissolved in 250 cm
3
=0.25 (1000 250) =1.00 mol/dm
3
c) 0.02 mol of NaOH dissolved in 50 cm
3
=0.02 (1000 50) =0.400 mol/dm
3
d) 29.25 g of NaCl =29.25 58.5 =0.5 mol
0.5 mol of NaCl dissolved in 4.0 dm
3
=0.5 (1 4) =0.125 mol/dm
3
e) 9.8 g of H
2
SO
4
=9.8 98 =0.1 mol
0.1 mol of H
2
SO
4
dissolved in 250 cm
3
=0.1 (1000 250) =0.400 mol/dm
3
2. moles =(c v) 1000 c =concentration in mol/dm
3
v =volume in cm
3
moles =mass in g relative molecular mass
a) moles of NaOH =(0.5 2000) 1000 =1.00 mol
b) H
2
SO
4
2H
+
+SO
4
2
0.5 mol/dm
3
H
2
SO
4
will contain 2 0.5 =1.0 mol/dm
3
H
+
moles of H
+
=(1.0 4000) 1000 =4.00 mol
c) moles of KOH =(0.4 25.0) 1000 =0.0100 mol
d) concentration of HNO
3
=6.3 63.0 =0.1 mol 0.1 mol/dm
3
moles of HNO
3
=(0.1 100.0) 1000 =0.0100 mol
e) concentration of Na
2
CO
3
=5.3 106.0 =0.05 mol/dm
3
Na
2
CO
3
2Na
+
+CO
3
2
0.05 mol/dm
3
of Na
2
CO
3
will contain 2 0.05 =0.1 mol/dm
3
Na
+
moles of Na
+
=(0.1 500) 1000 =0.0500 mol
3. relative molecular mass of NaOH =23.0 +16.0 +1.0 =40.0
mass of NaOH =0.2 40.0 =8.0g concentration =8.00 g/dm
3
4. moles of H
2
SO
4
used =4.9 98.0 =0.05 mol
0.05 mol of H
2
SO
4
dissolved in 1000 cm
3
=0.0500 mol/dm
3
5.
a) 3.65 g of HCl dissolved in 200 cm
3
=3.65 200 g dissolved in 1 cm
3
=(3.65 200) 1000
=18.25 g/dm
3
b) moles of HCl =3.65 36.5 =0.1 mol
0.1 mol of HCl dissolved in 200 cm
3
=0.1 200 mol dissolved in 1 cm
3
=(0.1 200) 1000
=0.500 mol/dm
3
6. 2.0 mol in 1000 cm
3
=2.0 1000 mol in 1 cm
3
=(2.0 1000) 25.0 mol in 25.0 cm
3
=0.0500 mol
7. relative molecular mass of KOH =39.1 +16.0 +1.0 =56.1
mass of 0.4 mol of KOH =0.4 56.1 =22.4 g
5
8.
a) 2.0 mol of H
2
O
2
in 1000 cm
3
=2.0 1000 mol in 1 cm
3
=(2.0 1000) 50 mol in 50 cm
3
=0.100 mol
b) from the balanced chemical equation, 2 mol of H
2
O
2
produce 1 mol of O
2
0.1 mol of H
2
O
2
will produce 0.1 0.5 =0.0500 mol of O
2
c) volume of gas =mol 24.0 dm
3
=0.05 24.0 =1.20 dm
3
9.
a) 0.1 mol of NaOH in 1000 cm
3
=0.1 1000 mol in 1 cm
3
=(0.1 1000) 30 mol in 30 cm
3
=0.003 mol
2NaOH
(aq)
+H
2
SO
4(aq)
Na
2
SO
4(aq)
+2H
2
O
(l)
from the balanced chemical equation, 2 mol of NaOH react with 1 mol of H
2
SO
4
0.003 mol of NaOH react with 0.003 0.5 =0.0015 mol of H
2
SO
4
there are 0.0015 mol of H
2
SO
4
dissolved in 25.0 cm
3
of solution
=0.0015 25.0 mol of H
2
SO
4
dissolved in 1.0 cm
3
of solution
=(0.0015 25.0) 1000 mol of H
2
SO
4
dissolved in 1000 cm
3
of solution =0.0600 mol/dm
3
b) relative molecular mass of H
2
SO
4
=(2 1.0) +32.0 +(4 16.0) =98.0
mass concentration of H
2
SO
4
=98.0 0.06 =5.88 g/dm
3
10.
a) 0.2 mol of H
2
SO
4
in 1000 cm
3
=0.2 1000 mol in 1 cm
3
=(0.2 1000) 18.0 mol in 18.0 cm
3
=0.0036 mol
2KOH
(aq)
+H
2
SO
4(aq)
K
2
SO
4(aq)
+2H
2
O
(l)
from the balanced chemical equation, 1 mol of H
2
SO
4
react with 2 mol of KOH
0.0036 mol of H
2
SO
4
react with 2 0.0036 =0.0072 mol of KOH
there are 0.0072 mol of KOH dissolved in 24.0 cm
3
of solution
=0.0072 24.0 mol of KOH dissolved in 1.0 cm
3
of solution
=(0.0072 24.0) 1000 mol of KOH dissolved in 1000 cm
3
of solution =0.300 mol/dm
3
b) relative molecular mass of KOH =39.1 +16.0 +1.0 =56.1 mass concentration =0.3 56.1
=16.8 g dm
3
11.
2NaOH
(aq)
+H
2
SO
4(aq)
Na
2
SO
4(aq)
+2H
2
O
(l)
moles of sulphuric acid =0.1 mol in 1000 cm
3
=0.1 1000 mol in 1 cm
3
=(0.1 1000) 24.7 mol
in 24.7 cm
3
=0.00247 mol
from the balanced chemical equation, 2 mol of NaOH react with 1 mol of H
2
SO
4
amount of NaOH =0.00247 2 =0.00494 mol
volume of NaOH =0.00494 (0.5 1000) =9.88 cm
3
6
12.
NaOH
(aq)
+HCl
(aq)
NaCl
(aq)
+H
2
O
(l)
moles of sodium hydroxide =0.4 mol in 1000 cm
3
=0.4 1000 mol in 1 cm
3
=(0.4 1000) 22.5 mol in 22.5 cm
3
=0.009 mol
from the balanced chemical equation, 1 mol of NaOH react with 1 mol of HCl
amount of HCl =amount of NaOH =0.009 mol
volume of HCl =0.009 (0.25 1000) =36.0 cm
3
Potrebbero piacerti anche
- Electron Transfer Reactions of Complex Ions in SolutionDa EverandElectron Transfer Reactions of Complex Ions in SolutionNessuna valutazione finora
- Application of Mole ConceptDocumento12 pagineApplication of Mole ConceptSonal GiraseNessuna valutazione finora
- Critical Reasoning: Drill 1: SolutionsDocumento17 pagineCritical Reasoning: Drill 1: SolutionslognNessuna valutazione finora
- CHM270 - Tutorial 3 (Chemical Kinetics)Documento7 pagineCHM270 - Tutorial 3 (Chemical Kinetics)Azrie HizadNessuna valutazione finora
- Worksheet ElectrochemistryDocumento1 paginaWorksheet ElectrochemistryShreya GiriNessuna valutazione finora
- 2012 Redox Tutorial-TutorDocumento11 pagine2012 Redox Tutorial-TutorKarunya NarayanamurthyNessuna valutazione finora
- Unit-1 MCQDocumento6 pagineUnit-1 MCQAnupa MedhekarNessuna valutazione finora
- Unit Practice Test: Gas Laws: Multiple ChoiceDocumento8 pagineUnit Practice Test: Gas Laws: Multiple Choiceanj pianoNessuna valutazione finora
- Workbk 10Documento177 pagineWorkbk 10Manushree NayakNessuna valutazione finora
- Titration Sample ProblemDocumento8 pagineTitration Sample ProblemPaulAcademicsNessuna valutazione finora
- Reacting MassesDocumento2 pagineReacting MassesTsz Wai WONGNessuna valutazione finora
- Kinetics Homework 3Documento4 pagineKinetics Homework 3RizkiNessuna valutazione finora
- PS As CB X Chem 1.2 Chemical Reactions and EquationsDocumento2 paginePS As CB X Chem 1.2 Chemical Reactions and EquationsHemant PadalkarNessuna valutazione finora
- Stoichiometry Questions & AnswersDocumento2 pagineStoichiometry Questions & Answersnosirat aladeNessuna valutazione finora
- IUPAC - DPP-1 To 10 - English - Print - 600Documento28 pagineIUPAC - DPP-1 To 10 - English - Print - 600Avijeet kumar Mishra100% (1)
- Titration ProblemsDocumento8 pagineTitration ProblemsAngela KocevskaNessuna valutazione finora
- Ch123 Exam II Practice Exam Spring2011Documento7 pagineCh123 Exam II Practice Exam Spring2011christopher92530% (1)
- SCH3U Exam Review QUESTIONSDocumento3 pagineSCH3U Exam Review QUESTIONSChen Dingna100% (1)
- ElectrolysisDocumento25 pagineElectrolysisMuhammad UmerNessuna valutazione finora
- Salts (NOTES)Documento32 pagineSalts (NOTES)Sabriza Hassan Assa'ariNessuna valutazione finora
- Acid Base Problems SolutionsDocumento20 pagineAcid Base Problems SolutionsAnusha PatelNessuna valutazione finora
- XI Chemistry Chapterwise Advanced Study MaterialDocumento537 pagineXI Chemistry Chapterwise Advanced Study MaterialregisNessuna valutazione finora
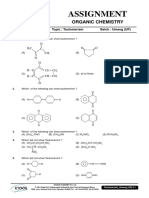
- Assignment: Organic ChemistryDocumento6 pagineAssignment: Organic ChemistryWalid EbaiedNessuna valutazione finora
- Che 02Documento5 pagineChe 02lakashl14Nessuna valutazione finora
- H2 Atomic Structure, Stiochiometry QuestionsDocumento8 pagineH2 Atomic Structure, Stiochiometry QuestionskitoniumNessuna valutazione finora
- Haloalkanes and Haloarenes - MCQSDocumento3 pagineHaloalkanes and Haloarenes - MCQSDivyam GargNessuna valutazione finora
- Mole Concept-1 JEE Main and Advanced PDFDocumento6 pagineMole Concept-1 JEE Main and Advanced PDFAryan Jaiswal100% (1)
- Practice Exam 2 ChemistDocumento5 paginePractice Exam 2 ChemistFATIN FARHANAH BINTI HALIDIN MoeNessuna valutazione finora
- PLTL Ch. 16 AssignmentDocumento6 paginePLTL Ch. 16 AssignmentJules BrunoNessuna valutazione finora
- 102 MSJC 13Documento11 pagine102 MSJC 13noelNessuna valutazione finora
- 3 - Chemical Thermodynamics and ThermochemistryDocumento3 pagine3 - Chemical Thermodynamics and ThermochemistryYawn D ArtistNessuna valutazione finora
- Net Ionic Equation W.S. AnswersDocumento2 pagineNet Ionic Equation W.S. Answersgimarreyes23Nessuna valutazione finora
- Alcohols Ethers and Phenol-03 - Assignments (New)Documento26 pagineAlcohols Ethers and Phenol-03 - Assignments (New)Raju SinghNessuna valutazione finora
- Chemistry Worksheet 2Documento8 pagineChemistry Worksheet 2Marie BozemanNessuna valutazione finora
- Quiz - Coordination Compounds PDFDocumento2 pagineQuiz - Coordination Compounds PDFAman JaiswalNessuna valutazione finora
- Sicmyb - DPP Mole ConceptDocumento6 pagineSicmyb - DPP Mole ConceptBorn to fightNessuna valutazione finora
- SL Score ! /30: Practice Exam: Paper 1 Topic 4: BondingDocumento7 pagineSL Score ! /30: Practice Exam: Paper 1 Topic 4: Bondingraja_tanukuNessuna valutazione finora
- Supplementary ProblemsDocumento30 pagineSupplementary ProblemsMike PatenaudeNessuna valutazione finora
- Buffers&titrationsquestions ReviewDocumento6 pagineBuffers&titrationsquestions Reviewapi-279595789Nessuna valutazione finora
- Topic 10 20 MC PracticeDocumento17 pagineTopic 10 20 MC PracticePipen 5Nessuna valutazione finora
- Atomicstructurequestions PDFDocumento42 pagineAtomicstructurequestions PDFNfor KlinsmanNessuna valutazione finora
- Physical Chemistry: Shailendra KRDocumento6 paginePhysical Chemistry: Shailendra KR1harshikaNessuna valutazione finora
- Organic Chemistry: GladiatorDocumento4 pagineOrganic Chemistry: GladiatorArihant KumarNessuna valutazione finora
- Chem G-9 Lesson 7 IGCSE Qs - Rates of ReactionDocumento24 pagineChem G-9 Lesson 7 IGCSE Qs - Rates of ReactionKarim WaelNessuna valutazione finora
- Acids and Derivatives TutorialDocumento18 pagineAcids and Derivatives TutorialChen ZhihaoNessuna valutazione finora
- AP Chem CH 7 Practice QuizDocumento8 pagineAP Chem CH 7 Practice QuizOmprakash LatiyalNessuna valutazione finora
- Assignment 151Documento5 pagineAssignment 151Hai Xuan DoNessuna valutazione finora
- Oxford Resources For IB: Structure 3.1 - The Periodic Table: Classification of ElementsDocumento19 pagineOxford Resources For IB: Structure 3.1 - The Periodic Table: Classification of ElementsGian Paolo GerzonNessuna valutazione finora
- Back TitrateDocumento16 pagineBack Titratepicket1019Nessuna valutazione finora
- AP Chem CH 6 Practice QuizDocumento3 pagineAP Chem CH 6 Practice Quizprin ppNessuna valutazione finora
- Sem1 Unit8 Phase EquilibriaDocumento5 pagineSem1 Unit8 Phase EquilibriashehdilanunNessuna valutazione finora
- Mole Concept Solution Practice Set Objective by S.K.sinha See Chemistry Animations atDocumento1 paginaMole Concept Solution Practice Set Objective by S.K.sinha See Chemistry Animations atmyiitchemistry50% (2)
- 8 - Sample Titration ProblemsDocumento15 pagine8 - Sample Titration ProblemsGerald LimNessuna valutazione finora
- Percentage YieldDocumento2 paginePercentage YieldHal OgleNessuna valutazione finora
- Experiment I1 Preparation of Some Cobaltammine ComplexesDocumento7 pagineExperiment I1 Preparation of Some Cobaltammine ComplexesIftitah HauriyahNessuna valutazione finora
- CBSE Class 10 Chemistry Worksheet - Chemical Reactions and EquationsDocumento4 pagineCBSE Class 10 Chemistry Worksheet - Chemical Reactions and EquationsAsh snowNessuna valutazione finora
- MCQ Chapter 9 Haloalkanes and HaloarenesDocumento2 pagineMCQ Chapter 9 Haloalkanes and HaloarenesNinaNessuna valutazione finora
- Physical Chemistry QuestionsDocumento22 paginePhysical Chemistry QuestionshanaNessuna valutazione finora
- S7 11012021 Acid Base Titrations WS With ANSWERSDocumento7 pagineS7 11012021 Acid Base Titrations WS With ANSWERSFatima Ahmed-VeriterNessuna valutazione finora
- S.4 Mole (2) + TitrationDocumento27 pagineS.4 Mole (2) + TitrationS4C07 Lai Yik TsunNessuna valutazione finora
- Nestle Pakistan - Case Study ClassDocumento4 pagineNestle Pakistan - Case Study ClassKhadija KarimNessuna valutazione finora
- Example, Straight Line DepreciationDocumento12 pagineExample, Straight Line DepreciationKhadija Karim100% (2)
- Welcome: Business Law/ Corporate LawDocumento7 pagineWelcome: Business Law/ Corporate LawKhadija KarimNessuna valutazione finora
- Food NutrientDocumento3 pagineFood NutrientKhadija KarimNessuna valutazione finora
- Potash Feldspar K2o Al2o3 6sio2Documento1 paginaPotash Feldspar K2o Al2o3 6sio2Vikash AgarwalNessuna valutazione finora
- File 3Documento22 pagineFile 3Talha ZubayerNessuna valutazione finora
- 0620 - s06 - QP - 6 (ALTERNATE TO PRACTICAL) PDFDocumento16 pagine0620 - s06 - QP - 6 (ALTERNATE TO PRACTICAL) PDFconsultunnikrishnan1850Nessuna valutazione finora
- Treatment of Real Printing Wastewater Using Electrocoagulation Process With Titanium and Zinc ElectrodesDocumento9 pagineTreatment of Real Printing Wastewater Using Electrocoagulation Process With Titanium and Zinc ElectrodesCUEVA VASQUEZ BRYAN ALBERTONessuna valutazione finora
- STDNT KS3 Year 7 C1 Chapter 4 Acids and AlkalisDocumento8 pagineSTDNT KS3 Year 7 C1 Chapter 4 Acids and Alkalis21tahiradamNessuna valutazione finora
- 2.5 Insulation & RefractoriesNDocumento43 pagine2.5 Insulation & RefractoriesNgaurang1111Nessuna valutazione finora
- Isolation and Qualitative Analysis of Nucleic Acids (DNA From Onion)Documento3 pagineIsolation and Qualitative Analysis of Nucleic Acids (DNA From Onion)Elina Lantion100% (1)
- Laundry Detergents Formulation With ExplanationsDocumento13 pagineLaundry Detergents Formulation With Explanationsramu_uppada89% (9)
- Organic QuestionsDocumento51 pagineOrganic Questionshemab30851Nessuna valutazione finora
- M1-L2 - Hardness Determination EDTADocumento9 pagineM1-L2 - Hardness Determination EDTAShivika MittalNessuna valutazione finora
- Rop 2019Documento17 pagineRop 2019Alya PolapaNessuna valutazione finora
- Earth and Life Science: Quarter 1 - Module 3: MineralsDocumento24 pagineEarth and Life Science: Quarter 1 - Module 3: MineralsSarah SanchezNessuna valutazione finora
- Act B1 PCC Interim AssignmentDocumento6 pagineAct B1 PCC Interim AssignmentHassan ZakariaNessuna valutazione finora
- Sikalite®PK100: Powder Intergral Waterproofer For Mortar and ConcreteDocumento2 pagineSikalite®PK100: Powder Intergral Waterproofer For Mortar and ConcretewaqasNessuna valutazione finora
- Isolation of DNA From Given Sample.Documento2 pagineIsolation of DNA From Given Sample.gauravpawar5971252Nessuna valutazione finora
- Limiting Reagent ProblemsDocumento6 pagineLimiting Reagent ProblemsFarisDonNessuna valutazione finora
- Experiment 6 - Elementary Analysis REVISEDDocumento4 pagineExperiment 6 - Elementary Analysis REVISEDAlex Borja100% (3)
- Jenny Ultimate Blue Synthetic Compressor Oil - Product SpecificationsDocumento2 pagineJenny Ultimate Blue Synthetic Compressor Oil - Product SpecificationsAnibal RiosNessuna valutazione finora
- GuideDocumento8 pagineGuideMichael A. HackneyNessuna valutazione finora
- Spec Epoxyprene Enr TdsDocumento4 pagineSpec Epoxyprene Enr Tdsapi-286010429100% (1)
- Rezaei 2009Documento10 pagineRezaei 2009Química InvestChimNessuna valutazione finora
- CML 100 Inorganic Part Home Assignment and Solved Problems For Self Study-Part 3Documento1 paginaCML 100 Inorganic Part Home Assignment and Solved Problems For Self Study-Part 3RhombiNessuna valutazione finora
- Novel Drug Delivery SystemDocumento20 pagineNovel Drug Delivery SystemZainab ArshadNessuna valutazione finora
- Investigating Effect of Temperature On The Activity of Lipase Ss 35Documento3 pagineInvestigating Effect of Temperature On The Activity of Lipase Ss 35Chelsea MontrichardNessuna valutazione finora
- Priscilla Proposal For Chemistry STPM1-2Documento4 paginePriscilla Proposal For Chemistry STPM1-2SalinKaurNessuna valutazione finora
- 37 Metals Non Metals Metalloids Long TestDocumento2 pagine37 Metals Non Metals Metalloids Long TestMyrna Appal100% (2)
- Unit-2-Question Bank PDFDocumento17 pagineUnit-2-Question Bank PDFSujith.VetriMaaranNessuna valutazione finora
- Finishes in Textiles: Identification Finishes in Textiles: IdentificationDocumento7 pagineFinishes in Textiles: Identification Finishes in Textiles: IdentificationMilagros WieczorekNessuna valutazione finora
- 1 Eksplorasi Geoffrey de Jong PTFI Prosiding FixDocumento7 pagine1 Eksplorasi Geoffrey de Jong PTFI Prosiding FixRidwan KotoNessuna valutazione finora
- Periodic Tales: A Cultural History of the Elements, from Arsenic to ZincDa EverandPeriodic Tales: A Cultural History of the Elements, from Arsenic to ZincValutazione: 3.5 su 5 stelle3.5/5 (137)
- Process Plant Equipment: Operation, Control, and ReliabilityDa EverandProcess Plant Equipment: Operation, Control, and ReliabilityValutazione: 5 su 5 stelle5/5 (1)
- Chemistry for Breakfast: The Amazing Science of Everyday LifeDa EverandChemistry for Breakfast: The Amazing Science of Everyday LifeValutazione: 4.5 su 5 stelle4.5/5 (14)
- It's Elemental: The Hidden Chemistry in EverythingDa EverandIt's Elemental: The Hidden Chemistry in EverythingValutazione: 4 su 5 stelle4/5 (10)
- Chemistry: a QuickStudy Laminated Reference GuideDa EverandChemistry: a QuickStudy Laminated Reference GuideValutazione: 5 su 5 stelle5/5 (1)
- ICH Quality Guidelines: An Implementation GuideDa EverandICH Quality Guidelines: An Implementation GuideAndrew TeasdaleNessuna valutazione finora
- The Nature of Drugs Vol. 1: History, Pharmacology, and Social ImpactDa EverandThe Nature of Drugs Vol. 1: History, Pharmacology, and Social ImpactValutazione: 5 su 5 stelle5/5 (5)
- The Nature of Drugs Vol. 1: History, Pharmacology, and Social ImpactDa EverandThe Nature of Drugs Vol. 1: History, Pharmacology, and Social ImpactValutazione: 5 su 5 stelle5/5 (1)
- The Regenerative Grower's Guide to Garden Amendments: Using Locally Sourced Materials to Make Mineral and Biological Extracts and FermentsDa EverandThe Regenerative Grower's Guide to Garden Amendments: Using Locally Sourced Materials to Make Mineral and Biological Extracts and FermentsValutazione: 5 su 5 stelle5/5 (3)
- Monkeys, Myths, and Molecules: Separating Fact from Fiction, and the Science of Everyday LifeDa EverandMonkeys, Myths, and Molecules: Separating Fact from Fiction, and the Science of Everyday LifeValutazione: 4 su 5 stelle4/5 (1)
- Troubleshooting Process Plant Control: A Practical Guide to Avoiding and Correcting MistakesDa EverandTroubleshooting Process Plant Control: A Practical Guide to Avoiding and Correcting MistakesValutazione: 1 su 5 stelle1/5 (2)
- Handbook of Formulating Dermal Applications: A Definitive Practical GuideDa EverandHandbook of Formulating Dermal Applications: A Definitive Practical GuideNessuna valutazione finora
- Practical High-Performance Liquid ChromatographyDa EverandPractical High-Performance Liquid ChromatographyNessuna valutazione finora
- Chemistry for Breakfast: The Amazing Science of Everyday LifeDa EverandChemistry for Breakfast: The Amazing Science of Everyday LifeValutazione: 4.5 su 5 stelle4.5/5 (90)
- The Nature of Drugs Vol. 2: History, Pharmacology, and Social ImpactDa EverandThe Nature of Drugs Vol. 2: History, Pharmacology, and Social ImpactNessuna valutazione finora
- The Production of Volatile Oils and Perfumery Plants in the United StatesDa EverandThe Production of Volatile Oils and Perfumery Plants in the United StatesNessuna valutazione finora
- The Elements We Live By: How Iron Helps Us Breathe, Potassium Lets Us See, and Other Surprising Superpowers of the Periodic TableDa EverandThe Elements We Live By: How Iron Helps Us Breathe, Potassium Lets Us See, and Other Surprising Superpowers of the Periodic TableValutazione: 3.5 su 5 stelle3.5/5 (22)