Documenti di Didattica
Documenti di Professioni
Documenti di Cultura
OCHEM Lab 13 Scribd
Caricato da
Matthew Harry0 valutazioniIl 0% ha trovato utile questo documento (0 voti)
2K visualizzazioni4 pagineMultistep Synthesis of Hexaphenylbenzene
Copyright
© © All Rights Reserved
Formati disponibili
DOCX, PDF, TXT o leggi online da Scribd
Condividi questo documento
Condividi o incorpora il documento
Hai trovato utile questo documento?
Questo contenuto è inappropriato?
Segnala questo documentoMultistep Synthesis of Hexaphenylbenzene
Copyright:
© All Rights Reserved
Formati disponibili
Scarica in formato DOCX, PDF, TXT o leggi online su Scribd
0 valutazioniIl 0% ha trovato utile questo documento (0 voti)
2K visualizzazioni4 pagineOCHEM Lab 13 Scribd
Caricato da
Matthew HarryMultistep Synthesis of Hexaphenylbenzene
Copyright:
© All Rights Reserved
Formati disponibili
Scarica in formato DOCX, PDF, TXT o leggi online su Scribd
Sei sulla pagina 1di 4
Organic Chemistry Lab Report 13: Multistep Synthesis of Hexaphenylbenzene
April 22, 2014
Purpose/ Reactions:
The purpose of this step was to perform the multistep parallel synthesis of
Hexaphenylbenzene. We began with the synthesis of meso-dibromostilbene and
then produced diphenylacetylene. We also synthesized
tetraphenylcyclopentadienone, which we reacted with the diphenylacetylene to
produce the Hexaphenylbenzene.
Results:
1. YIELDS
a. Synthesis of meso-Stilbene Dibromide
i. Final mass: 3.05 g
ii. Starting materials: 1.0 g trans-stilbene , 2.0 g pyridinium
tribomide
1. 1.0g/180.25 = 0.0055 mol ; 2.0 g/319.82 = 0.0063 mol
2. 0.0055 mol trans-stilbene is limiting reagent
3. 0.0055 mol trans-stilbene * (1 mol meso-Stilbene
Dibromide/ 1 mol trans-stilbene) * (340.05 g/ 1 mol
meso-Stilbene Dibromide) = 1.89 g meso-Stilbene
Dibromide theoretical yield
4. Yield = (3.05 g)/(1.89)*100 = 161.4% yield
b. Synthesis of diphenylacetylene
i. Final mass: 1.3 g
ii. Starting Material: 3.05 g meso-Stilbene Dibromide; KOH
1. 3.05 g meso-Stilbene Dibromide * (1 mol/ 340.05 g
meso-Stilbene Dibromide) = 0.0090 mol meso-Stilbene
Dibromide is limiting reagent
2. 0.0090 mol meso-Stilbene Dibromide * (1 mol
diphenylacetylene/ 1mol meso-Stilbene Dibromide) *
(178.23 g/ mol) = 1.60 g diphenylacetylene theoretical
yield
3. Yield = (1.3 g)/(1.6 g)*100 = 81.3% yield
c. Synthesis of tetraphenylcyclopentadienone
i. Final mass: 0.15 g
ii. Starting materials: 200 mg 1,3-diphenylacetone; 200 mg
benzil; 200 L benzyltrimethylammonium hydroxide
1. 0.2 g 1,3-diphenylacetone/ 210.27 = 9.512E-4 mol; 0.2 g
benzil/ 210.23 = 0.9513E-4 so the 1,3-diphenylacetone
is the limiting reagent but not by much
2. (0.9512E-4 mol 1,3-diphenylacetone) * (1 mol
tetraphenylcyclopentadienone/ 1mol 1,3-
diphenylacetone) * (384.47 g/mol) = 0.65 g
tetraphenylcyclopentadienone theoretical yield
3. Yield = (0.15 g)/(0.65 g)*100 = 23.1% yield
2. OVERALL YIELD
a. With a 23.1% yield, tetraphenylcyclopentadienone was the limiting
reagent for the overall reaction.
i. 0.15 g tetraphenylcyclopentadienone * (1 mol/ 384.47 g) =
3.90E-4 mol tetraphenylcyclopentadienone ; 1.3 g
diphenylacetylene * (1 mol /178.23 g) = 0.0073 mol
ii. 3.90E-4 mol tetraphenylcyclopentadienone is the limiting
reagent
iii. 3.90E-4 * (1 mol hexaphenylbenzene/ 1mol
tetraphenylcyclopentadienone) * (534.69 g/ mol
hexaphenylbenzene) = 0.21 g hexaphenylbenzene
iv. Yield = (0.13 g)/(0.21 g)*100 = 62.0% yield
hexaphenylbenzene
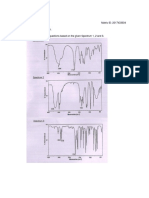
3. IR SPECTRA
a. tetraphenylcyclopentadienone
i. I had key peaks at 3054 cm
-1
and 2986 cm
-1
representing the
aromatic C-H bonds, 1265 cm
-1
representing the C-O double
bond of the ketone carbonyl, and 738 cm
-1
representing the C-C
bonds of the phenyl groups. These all fall in accordance with
the literature values for each of these respective bonds.
b. hexaphenylbenzene
i. I had key peaks at 3054 cm
-1
representing the C-H bonds in the
aromatic phenyls, 2963 cm
-1
representing the weak overtones,
1700 cm
-1
, 1670 cm
-1
, and 1595 cm
-1
representing the C-C
bonds in the aromatics and 693 cm
-1
representing the aromatic
C-H bonds.
Discussion:
1. PURITY
a. meso-Stilbene Dibromide
i. The literature value melting point for this molecule is 241C
and my recorded melting point range was 234C-236C,
indicating a very pure product. In addition, the literature
describes the appearance of his compound as a white power,
which I also recorded.
b. tetraphenylcyclopentadienone
i. The literature value melting point for this molecule is 219C-
220C and I recorded a melting point range of 216.8C-
219.3C. The literature describes
tetraphenylcyclopentadienone as a dark purple crystalline
solid. I had similar observations as I described the compound
as a dark purple crystalline solid. Therefore, my product was
also very pure.
c. diphenylacetylene
i. The literature value melting point for this molecule is 62.5C
and I recorded a melting point range of 59.5C-62C. The
literature describes diphenylacetylene as a clear crystalline
solid. I had similar observations as I described the compound
as a clear, crystal-structured solid. Therefore, my product for
diphenylacetylene was pure.
2. The key difference between the tetraphenylcyclopentadienone and the
hexaphenylbenzene should be the absence of the carbonyl (ketone) peak in
the latter molecule. Upon the addition of diphenylacetylene and heat,
tetraphenylpentadienone will lose a Carbon and Oxygen atom and will form
the hexaphenylbenzene, which itself is compromised of only the phenyl
groups surrounding a benzene ring. My IR spectra show this analysis to be
true as I observed a peak at 1265 cm
-1
representing the C-O double bond of
the ketone carbonyl in the tetraphenylcyclopentadienone and no peak at this
value for the hexaphenylbenzene. A lack of the carbonyl group illustrates the
success of this reaction because it shows that the ketone was removed
completely.
3. All the melting points in this synthesis are relatively high (the melting point
of the meso-stilbene dibromide is around 240C) so activation energy for the
reaction must be reached for the reaction to commence specifically, in the
synthesis of the hexaphenylbenzene, all of the diphenylacetylene and the
tetraphenylcyclopentadienone need to react for a good yield since we are
already in parallel synthesis and will only get a 73% yield. Therefore, the
temperature needs to be above 260C so that the activation energy of the
individual components is reached and they can react to form
hexaphenylbenzene. Furthermore, both the reagents in the synthesis of the
hexaphenylbenzene are solids so there would need to be a high temperature
to get the two substances to dissolve and to mix together to react.
4. The cyclopentadienone is itself very unstable but when the four phenyl
groups are added to it, they serve to protect the carbonyl core from any
external reagents and reactants. The phenyl groups repulse each other so
that they spread around the carbonyl group at four of the five Carbons on the
cyclopentadienone and therefore, due to their large size relative to the
carbonyl, sterically prevent other reactants from approaching the oxygen.
5. You can test the hexaphenylbenzene for optical rotation and see if it
illustrates both R and S characteristics. If the optical rotation shows zero
degree rotation than the mixture is racemic since the R and S cancel each
other out.
6.
Potrebbero piacerti anche
- Experiment 10: Iodine Clock ReactionDocumento11 pagineExperiment 10: Iodine Clock ReactionJohn NdambukiNessuna valutazione finora
- EXPERIMENT 5 Robinson Annulation ReactionDocumento13 pagineEXPERIMENT 5 Robinson Annulation ReactionMuhammad Amirul Afifi100% (1)
- FerroceneDocumento20 pagineFerroceneKalfakNessuna valutazione finora
- Discussions Exp 14 RecrystallizationDocumento4 pagineDiscussions Exp 14 RecrystallizationEdwin fooNessuna valutazione finora
- 3 Determination of Complex Ion by Jobs MethodDocumento2 pagine3 Determination of Complex Ion by Jobs Methodvishwanathz47Nessuna valutazione finora
- Outline of Article Analysis/Elc550: © Hak Cipta Universiti Teknologi MARADocumento7 pagineOutline of Article Analysis/Elc550: © Hak Cipta Universiti Teknologi MARAAliya GhazaliNessuna valutazione finora
- ObjectiveDocumento8 pagineObjectivenaim rashidNessuna valutazione finora
- Experiment 3Documento8 pagineExperiment 3ohhiNessuna valutazione finora
- CHM557 Exp 3Documento22 pagineCHM557 Exp 3syafNessuna valutazione finora
- Chm580 Experiment 3Documento9 pagineChm580 Experiment 3ohhiNessuna valutazione finora
- Robinson Annulation Reaction of NItrochalconeDocumento10 pagineRobinson Annulation Reaction of NItrochalconeMohd Nakirudin Muhamad NorNessuna valutazione finora
- Analysis of Aspirin in Commercial Apc Tablet Using Ftir SpectrosDocumento5 pagineAnalysis of Aspirin in Commercial Apc Tablet Using Ftir SpectrosMay Lee100% (1)
- NITRATION OF METHYL BENZOATE (ELECTROPHILIC AROMATIC SUBSTITUITION - Idayu Razali - Academia - Edu PDFDocumento7 pagineNITRATION OF METHYL BENZOATE (ELECTROPHILIC AROMATIC SUBSTITUITION - Idayu Razali - Academia - Edu PDFyawsNessuna valutazione finora
- Experiment Baking SsodaDocumento7 pagineExperiment Baking Ssodaatynzaty0% (1)
- ALDOL Synth - DibenzalacetoneDocumento7 pagineALDOL Synth - DibenzalacetoneJames CookeNessuna valutazione finora
- Lab Report 2 (Bio462)Documento4 pagineLab Report 2 (Bio462)Allisya NasirNessuna valutazione finora
- CHM 475 Inorganic Chemistry: (Experiment 8)Documento5 pagineCHM 475 Inorganic Chemistry: (Experiment 8)FAtma HAnysNessuna valutazione finora
- Experiment 2 Sodium Borohydride Reduction of CyclohexanoneDocumento6 pagineExperiment 2 Sodium Borohydride Reduction of CyclohexanoneSarah HannisNessuna valutazione finora
- 4d Application of Complexometric TitrationDocumento16 pagine4d Application of Complexometric TitrationnajwaNessuna valutazione finora
- Experiment 5 AASDocumento15 pagineExperiment 5 AASnn bbNessuna valutazione finora
- CHM 510 Exp 1 GCDocumento8 pagineCHM 510 Exp 1 GCNurul HaziqahNessuna valutazione finora
- Preparation of 1-Bromobutane From 1-ButanolDocumento5 paginePreparation of 1-Bromobutane From 1-ButanolHidayu AdnanNessuna valutazione finora
- LONG CAFFEINE UVLong Caffeine UvDocumento8 pagineLONG CAFFEINE UVLong Caffeine UvzaNessuna valutazione finora
- Covid-19 Laboratory Report Exp 5Documento7 pagineCovid-19 Laboratory Report Exp 5Nasuha AriffinNessuna valutazione finora
- EXPERIMENT 2: Determgcnation of Water Hardness by Atomic Absorption Spectroscopy (AAS)Documento7 pagineEXPERIMENT 2: Determgcnation of Water Hardness by Atomic Absorption Spectroscopy (AAS)Afiqah SamanNessuna valutazione finora
- NaBH4 Reduction of CyclohaxanoneDocumento5 pagineNaBH4 Reduction of Cyclohaxanonenurul1110Nessuna valutazione finora
- Exp 2Documento11 pagineExp 2ohhiNessuna valutazione finora
- CHM510 - SpeDocumento7 pagineCHM510 - SpeafifiNessuna valutazione finora
- Spectrochemical Method of Analysis (CHM 580) EXPERIMENT 1:qualitative Analysis of Aspirin Phenacetin Caffeine and Sample Using FTIR and NMRDocumento9 pagineSpectrochemical Method of Analysis (CHM 580) EXPERIMENT 1:qualitative Analysis of Aspirin Phenacetin Caffeine and Sample Using FTIR and NMRbatrisyia hazirahNessuna valutazione finora
- Determination of Vitamin CDocumento2 pagineDetermination of Vitamin CWalwin HareNessuna valutazione finora
- The Friedel-Crafts Reaction PDFDocumento7 pagineThe Friedel-Crafts Reaction PDFIsaac Nicholas NotorioNessuna valutazione finora
- 4A F16 Exp 04 Absorbance and Fluorescence Spectros PDFDocumento27 pagine4A F16 Exp 04 Absorbance and Fluorescence Spectros PDFWanqing HeNessuna valutazione finora
- Ioron Determination in WaterDocumento6 pagineIoron Determination in WaterGobe JamNessuna valutazione finora
- Sodium Borohydride Reduction of Cyclohex PDFDocumento8 pagineSodium Borohydride Reduction of Cyclohex PDFhahadindongNessuna valutazione finora
- Phase Transfer 0Documento3 paginePhase Transfer 0Jeannine CoxNessuna valutazione finora
- Experiment 7Documento8 pagineExperiment 7Shinichi KudoNessuna valutazione finora
- Hardness of WaterDocumento6 pagineHardness of WaterJamesShiq0% (1)
- Uv Vis Spectroscopy Lab Report of The Detection of Analytes From Environmental Samples Using UvDocumento13 pagineUv Vis Spectroscopy Lab Report of The Detection of Analytes From Environmental Samples Using UvSaba Naseer100% (1)
- Anal Chem 3 - Test 1-2016Documento4 pagineAnal Chem 3 - Test 1-2016Buhle BuhleNessuna valutazione finora
- Spectro ReportDocumento30 pagineSpectro ReportIbrahim Muhamad100% (2)
- Objectives: FIGURE A: Example of Coordination CompoundsDocumento7 pagineObjectives: FIGURE A: Example of Coordination CompoundsNurul izzatiNessuna valutazione finora
- Discussion Lab Report ACDocumento2 pagineDiscussion Lab Report ACNur AsiahNessuna valutazione finora
- Lab Report chm457 Exp 3Documento3 pagineLab Report chm457 Exp 3pufff witchesNessuna valutazione finora
- Laboratory Report: Chm457 - October 2020 - Odl Lab AssessmentDocumento5 pagineLaboratory Report: Chm457 - October 2020 - Odl Lab AssessmentHakim SunaeNessuna valutazione finora
- MIC481 Lab ReportDocumento6 pagineMIC481 Lab ReportAbg Khairul Hannan Bin Abg AbdillahNessuna valutazione finora
- Preparation of Acetaline Notes PDFDocumento6 paginePreparation of Acetaline Notes PDFAnonymous Wwxatt3oIK100% (1)
- Sample Question and Answer Scheme For Past Year Chapter 10 - Molecular Mass SpectrosDocumento8 pagineSample Question and Answer Scheme For Past Year Chapter 10 - Molecular Mass SpectrostirahNessuna valutazione finora
- Preparation of 4-Methylcyclohexene From Dehydration of 4-MethylcyclohexanolDocumento8 paginePreparation of 4-Methylcyclohexene From Dehydration of 4-MethylcyclohexanolHidayu AdnanNessuna valutazione finora
- EXPERIMENT 4 The Aldol CondenstaionDocumento8 pagineEXPERIMENT 4 The Aldol CondenstaionMuhammad Amirul AfifiNessuna valutazione finora
- CHM 421 - ToPIC 1 - CalculationsDocumento46 pagineCHM 421 - ToPIC 1 - CalculationsthemfyNessuna valutazione finora
- Potentiometric Titration of A Weak Acid: Chemistry 135 Clark CollegeDocumento14 paginePotentiometric Titration of A Weak Acid: Chemistry 135 Clark CollegeMay LeeNessuna valutazione finora
- Preparation of Tetraamminecopper II Sulphate.Documento10 paginePreparation of Tetraamminecopper II Sulphate.DaizLee Ahmad25% (4)
- Robinson Annulation Reaction of 3-Nitrochalcone With Ethyl AcetoacetateDocumento9 pagineRobinson Annulation Reaction of 3-Nitrochalcone With Ethyl AcetoacetateAmirul Azhar100% (5)
- Lab Ins 1 (Spectronic 20)Documento7 pagineLab Ins 1 (Spectronic 20)Ayale Mn100% (3)
- Results For Experiment 3Documento5 pagineResults For Experiment 3Syahriezan HaminNessuna valutazione finora
- Temperature Programmed Desorption With ReactionDocumento42 pagineTemperature Programmed Desorption With ReactionaquacobiaNessuna valutazione finora
- Lab Report 1 LahDocumento5 pagineLab Report 1 Lahsarra nazamNessuna valutazione finora
- Essays on Analytical Chemistry: In Memory of Professor Anders RingbomDa EverandEssays on Analytical Chemistry: In Memory of Professor Anders RingbomErkki WänninenNessuna valutazione finora
- Comparison of Prerogative Power and Tyranny in John Locke's Second Treatise of GovernmentDocumento13 pagineComparison of Prerogative Power and Tyranny in John Locke's Second Treatise of GovernmentMatthew HarryNessuna valutazione finora
- Seneca: Thyestes Act I in EnglishDocumento16 pagineSeneca: Thyestes Act I in EnglishMatthew HarryNessuna valutazione finora
- Seneca: Thyestes Act I in EnglishDocumento16 pagineSeneca: Thyestes Act I in EnglishMatthew HarryNessuna valutazione finora
- Thucydides: Peloponnesian Wars: Nicias and AlcibiadesDocumento8 pagineThucydides: Peloponnesian Wars: Nicias and AlcibiadesMatthew Harry100% (1)
- Buku Praktikum Kiia OrganikDocumento91 pagineBuku Praktikum Kiia OrganikKhairunnisah SiagianNessuna valutazione finora
- Pre Lab 1-4 Module DK024 (Question)Documento12 paginePre Lab 1-4 Module DK024 (Question)Irdina WnNessuna valutazione finora
- Oxidation of AlcohalDocumento49 pagineOxidation of Alcohalpramod kumar yadavNessuna valutazione finora
- Chemistry of Natural Products PDFDocumento21 pagineChemistry of Natural Products PDFhosseini_9864Nessuna valutazione finora
- 16H Carbonyl PDFDocumento60 pagine16H Carbonyl PDFJose Erick Ortega ValenciaNessuna valutazione finora
- Carbonyl ChemsketchDocumento3 pagineCarbonyl ChemsketchJenica RiaNessuna valutazione finora
- Nomenclature of A2Documento5 pagineNomenclature of A2SophieStevensNessuna valutazione finora
- Aldol CondensationDocumento4 pagineAldol Condensationrjbagh08Nessuna valutazione finora
- Grignard ReactionDocumento5 pagineGrignard ReactionNur HidayantiNessuna valutazione finora
- Carboxylic Acid & NitrilesDocumento19 pagineCarboxylic Acid & NitrilesDante Luis SilvaNessuna valutazione finora
- Efficient Aldolase Catalytic Antibodies The Enamine Mechanism of NaturalDocumento5 pagineEfficient Aldolase Catalytic Antibodies The Enamine Mechanism of NaturalShyam SathyamoorthiNessuna valutazione finora
- IUPAC NomenclatureDocumento36 pagineIUPAC NomenclatureRx Nadeem ChhipaNessuna valutazione finora
- Lab Report Expt 10Documento4 pagineLab Report Expt 10Eyvette GoNessuna valutazione finora
- IUPAC Provisional Recommendations: Chapter P-1 Nomenclature of Organic CompoundsDocumento94 pagineIUPAC Provisional Recommendations: Chapter P-1 Nomenclature of Organic CompoundsJuan Carlos SihotangNessuna valutazione finora
- Chm260 Tutorial 2Documento4 pagineChm260 Tutorial 2adlina asnawiNessuna valutazione finora
- Keto Prime Diet ReviewDocumento4 pagineKeto Prime Diet Reviewvanessa varversNessuna valutazione finora
- Aldehydes KetonesDocumento51 pagineAldehydes Ketonesadarsh mohanNessuna valutazione finora
- EAMCET QR Chemistry SR Chem 17.organic Chemistry Carbonyl CompoundsDocumento11 pagineEAMCET QR Chemistry SR Chem 17.organic Chemistry Carbonyl CompoundsJagadeesh GoliNessuna valutazione finora
- CH-105 - (4) Chemistry of Carbonyl CompoundsDocumento29 pagineCH-105 - (4) Chemistry of Carbonyl CompoundsK T Prajwal PrathikshNessuna valutazione finora
- Handbook5 Sunscreens 2Documento5 pagineHandbook5 Sunscreens 2ardanNessuna valutazione finora
- Infrared Spectroscopy: WWU ChemistryDocumento34 pagineInfrared Spectroscopy: WWU ChemistryTrung HoNessuna valutazione finora
- Systematic Organic AnalysisDocumento6 pagineSystematic Organic Analysisapi-19520338100% (5)
- AldehydeDocumento29 pagineAldehydeJan michael ChivaNessuna valutazione finora
- Aldehydes Ketones and Carboxylic Acids - NCERT SolutionsDocumento27 pagineAldehydes Ketones and Carboxylic Acids - NCERT SolutionsVyjayanthiNessuna valutazione finora
- Aldehydes and KetonesDocumento81 pagineAldehydes and Ketonesrouben09castroNessuna valutazione finora
- Instruction Manual FOR Vacuum Drying Ovens: Models: DP23 DP33 DP43 DP63Documento52 pagineInstruction Manual FOR Vacuum Drying Ovens: Models: DP23 DP33 DP43 DP63Deri CahyadiNessuna valutazione finora
- Subject ChemistryDocumento15 pagineSubject ChemistryDhirendra Singh RathoreNessuna valutazione finora
- Class 12th Chemistry Chapter 12 (Aldehydes, Ketones and Carboxylic Acids) Important Unsolved QuestionsDocumento13 pagineClass 12th Chemistry Chapter 12 (Aldehydes, Ketones and Carboxylic Acids) Important Unsolved QuestionsSanjiv KumarNessuna valutazione finora
- Organic Chemistry: Richard F. Daley and Sally J. DaleyDocumento47 pagineOrganic Chemistry: Richard F. Daley and Sally J. DaleySai Swaroop MandalNessuna valutazione finora
- The Cerric Nitrate Test: Shows Positive Test ForDocumento9 pagineThe Cerric Nitrate Test: Shows Positive Test ForPraveen KumarNessuna valutazione finora