Documenti di Didattica
Documenti di Professioni
Documenti di Cultura
Dehydration of Secondary Alcohols
Caricato da
Irek Habrylo0 valutazioniIl 0% ha trovato utile questo documento (0 voti)
140 visualizzazioni11 pagineFeb 21, 1961
Copyright
© Attribution Non-Commercial (BY-NC)
Formati disponibili
PDF, TXT o leggi online da Scribd
Condividi questo documento
Condividi o incorpora il documento
Hai trovato utile questo documento?
Questo contenuto è inappropriato?
Segnala questo documentoFeb 21, 1961
Copyright:
Attribution Non-Commercial (BY-NC)
Formati disponibili
Scarica in formato PDF, TXT o leggi online su Scribd
0 valutazioniIl 0% ha trovato utile questo documento (0 voti)
140 visualizzazioni11 pagineDehydration of Secondary Alcohols
Caricato da
Irek HabryloFeb 21, 1961
Copyright:
Attribution Non-Commercial (BY-NC)
Formati disponibili
Scarica in formato PDF, TXT o leggi online su Scribd
Sei sulla pagina 1di 11
ON THE MECHANISM OF DEHYDRATION OF SECONDARY
ALCOHOLS OVER ALUMINA CATALYST. 11.*
EFFECT OF STRUCTURE ON RATE
K. KOCHLOEFL, M. KRAUS, CHOU CHIN-SHEN**, L. BERANEK and V. BAZANT
Institute, of Chemical Process Fundamentals, Czechoslovak Academy of Science, Prague
Received February 21st, 1961
The effect of the structure of 14 secondary alcohols on the rate of their
dehydration on y-alumina has been determined. The compounds studied
included some aliphatic alcohols, cyclic alcohols of various ring size and
some stereoisomeric alkylcyclohexanols. Activation energies have been
estimated and the reactivity of the alcohols has been compared on the basis of
reaction rate constants for 200. The effects of vicinal carbon atom substi-
tution, of ring size and of steric configuration of the reacting molecule are
discussed. The results indicate that the geometry of the reacting molecule
is of major importance in the dehydration of alcohols on solid catalysts.
qne of the most fruitful approaches to the elucidation of reaction mechanisms in
organic chemistry has been the study of structure effects on reactivity. This approach
is extensively used for homogeneous transformations and it was to be expected that
its application to heterogeneous catalytic reactions would equally contribute to a bet-
ter understanding of such processes.
For catalytic dehydration of alcohols some very divergent mechanisms have been
proposed recently (see
1
- 3) none of which explains satisfactorily all experimental
facts. Further work is therefore necessary and in this paper kinetic measurements
of the dehydration of a series of secondary alcohols are described. By varying the
structure of the reacting compound, the influence of carbon chain branching, of
ring size and of configuration was studied.
Kinetics of primary and secondary alcohol dehydration over alumina has previously been
studied by Bork and Tolstopjatova
4
; these authors have found the same activation energy values
for all primary alcohols and another for aU secondary alcohols. However, from the small number
of substances reported in their paper no conclusions concerning the effect of the structure of
carbon skeleton on the reaction rate can be derived. Recently, Miller
5
and Laible
6
have studied
dehydration of some alcohols on silica-alumina catalyst. Unfortunately, their results form no
basis for consideration of structure effects on reactivity.
* Part L: This Journal 25, 2513 (1960).
** On leave of absence from the Institute of Applied Chemistry, Chang-ehun, China.
Vol. 27 (1962) 1199
Kochloe/l, Kraus, Chou Chin-Shen, Beranek, Balant:
Experimental
Boiling points and melting points values are uncorrected. The purity of all substances used was controlled by gas-liquid
chromatography.
Prepara tion of Cyclic Alcohols
Cyclopentanol was prepared by sodium reduction of cyclopentanone in aqueous methanol-
ether solutions. Yield 75%, b. p. 139-140
0
/760mm.
Cyclohexanol. A commercial product was converted into cyclohexyl acetate which was frac-
tionated on a column of 30 TP. The fraction boiling 1735/760 mm was hydrolysed with sodium
hydroxide solution and the alcohol destilled through the same column. B. p. 160 -1605, m. p.
20, nfio 14650.
Cycloheptanol and cyclooctanol were prepared by reduction of the corresponding ketones with
lithium aluminium hydride
9
in 94% and 954% yield, respectively: Cyc1oheptanol boiled at
88 -90/18 mm, cyc1ooctanol at 103 -104/15 mm.
For the preparation of cyc1oheptanone and cyc1ooctanone the standard ring enlargement of
cyc1ohexanone by means of diazomethane was used
10
,11. The composition of the reaction pro-
ducts as determined by gas-liquid chromatography for different cyc1ohexanone-diazomethane
ratios is shown in Table 1. The crude reaction mixture was fractionated on a destillation column
of 30 TP and the individual ketones were purified as semicarbazones. Cyc1oheptanone, b. p.
63 -64 /12mm; semicarbazone,m.p. 1635 (methanol). Cyc1ooctanone, b.p.100 -1005 /35 mm,
m. p. 34; semicarbazone, m. p. 1710 (methanol).
2,2-Dimethylcyclohexanol. 2-Methylcyc1ohexanone was methylated
12
yielding 65% of 2,2-
dimethylcyc1ohexanone, b. p. 1695/740 mm, which was reduced by lithium aluminium hydride
to the alcohol in 90% yield. B. p. 169 -172/750 mm.
Preparation of Aliphatic Alcohols
2-Methylhexanol-(3). An attempt to purify the product of the reaction of isopropylmagnesium
bromide with butyraldehyde
13
(yield 46%) by distillation was unsuccessful. It was therefore
converted to the corresponding ketone by potassium bichromate oxidation in dilute sulphuric
acid
14
. The 2-methylhexanone-(3) obtained was then purified as the semicarbazone by crystal-
lisation from methanol; m. p. 117 -118. Reduktion of the ketone by lithium aluminium hydride
afforded 2-methylhexanol-(3) in 92% yield; b. p. 144/760 mm.
1200
Table I
Composition of the Crude Reaction Product from the Reaction of Cyc1ohexanone
with Diazomethane
Molar ratio of diazomethane to cyc1ohexanone: A 116, B 232.
Compound A B
Cyc1ohexanone 05 0
Methylenecyc10hexane epoxide 51 06
Cyc1oheptanone 788 50
Methylenecyc10heptane epoxide 39 76
Cyc100ctan one 89 820
Cyc1ononanone 28 48
,Collection Czechoslov. Chern. Commun.
Mechanism of Dehydration of Secondary Alcohols over Alumina Catalyst. 11.
Heptanol-(4). The crude product obtained by the reaction of propylmagnesium bromide with
ethyl formate
15
was purified using the same method as above. Heptanone-(4), b. p. 144/760 mm;
semicarbazone, m. p. 132 (methanol). Heptanol-(4), b. p. 153 -153'5/748 mm.
2,4-Dimethylpentanol-(3) was prepared from isopropylmagnesium bromide and isobutyral-
dehyde
16
. Fractionation on a column of 20 TP afforded the pure alcohol, b. p. 1384 -138'8/
744 mm, in 68% yield.
Preparation of Stereoisomeric Alkylcyc1ohexanols
For kinetic measurements 40 - 80 g of the pure alcohols were required and the known methods
for the preparation of the stereoisomeric alkylcyclohexanols were therefore modified to make
them more suitable for large scale work. In general, a crude mixture enriched in one of the stereo-
isomers was transformed into the crystalline ester which was then purified by repeated crystal-
lisation.
Table II
Sodium Reduction of the Ketones in Absolute Ethanol
Ketone
Alcohol Yield
Alcohol Composition, %
%
trans
I
cis
2-Methy lcyclohexanone 780 880 120
4-Methylcyclohexanone 823 871 129
4-t-Butylcyclohexanone 742 927 7'3
Table III
Hydrogenation of the Ketones on Platinum Black in Acetic Acid-Hydrochloric Acid Mixture
Ketone
Alcohol Yield
Alcohol Composition, %
%
cis
I
trans
2-Methy lcyclohexanone 863 950 50
4-t-Butylcyclohexanone 812 94'5 4'5
2-t-Butylcyclohexanone 943 810 190
The crude products for the preparation of trans-comp ounds were obtained by reduction of the
corresponding alkylcyclohexanones with sodium in absolute ethanol
17
(see Table II). For the
preparation of cis-compounds the ketones were hydrogen ated on platinum black in acidic medium
(see Table III). In the case of trans-2-methylcyclohexanol the difference in esterification rates
of the stereoisomers was made use of for its isolation fro m the product of o-cresol hydrogenation.
The starting alkylphenols were hydrogenated on a Raney-nickel catalyst in ethanol at 80
and 150 atm. The unreacted alkylphenol and the corresponding ketone formed as by-product
were separated from the alcohol by the usual procedure. The isomer composition of the alcohols
is given in Table IV.
Vol. 27 (1962) 1201
Kochloe/l, Kraus, Chou Chin-Shen, Berdnek, Bazant:
Table IV
Hydrogenation of Alkylphenols on Raney-nickel
Alkylphenol
Alcohol Composition, %
cis
I
trans
o-Cresol 250 750
p-Cresol 320 680
p-t-Butylphenol 295 705
o-t-Butylphenol 300 700
trans-2-Methylcyclohexanol
trans-2-Methylcyclohexanol p-nitrobenzoate. To a mixture of dry pyridine (145 ml) and 2-
methylcyclohexanol (114 g, 1 mole, 75% trans-isomer) a solution of p-nitrobenzoyl chloride
(120 g, 0649 mole) in dry benzene (500 ml) was added dropwise under cooling and mixing.
Mter standing for 20 minutes, the reaction product was poured on a mixture of hydrochloric acid
(180 ml) and ice. The benzene layer was separated, washed with sodium hydrogen carbonate
solution and the unreacted alcohol and benzene were removed by steam distillation. The p-nitro-
benzoate was :filtered off; yield 137 g (521 %). Analysis of a hydrolysed sample indicated a content
of 93% of trans-isomer in the ester. Four crystallisations from methanol afforded pure trans-2-
methylcyclohexyl p-nitrobenzoate (80 g), m. p. 635 -'- 64.
trans-2-Methylcyclohexanol. The p-nitrobenzoate (256 g, 0973 mole) was refluxed \\,'ith
a solution of potassium hydroxide (112 g, 2 mole) in water (900 ml) and methanol (400 ml). After
two hours, the methanol was distilled off through a column; the remaining mixture was diluted
with water and the trans-2-methylcyclohexanol taken up in ether. The pure product was obtained
by destillation in 55% yield (615 g); b. p. 165/750 mm, r;25 = 372 cPo
cis-2-Methylcyclohexanol
.2-Methylcyclohexanone was prepared by potassium bichromate oxidation of crude 2-methyl-
cyclohexanol in dilute sulphuric acid
14
in 812% yield. The ketone was purified as the semicar-
bazone, m. p. 1915 (methanol). The yield of the pure 2-methylcyclohexanone was 664%, b. p.
160 -161/746 mm.
Hydrogenation of 2-methylcyclohexanone. The ketone (130 g, 116 mole) was hydrogenated on
platinum black catalyst (14 g) at atmospheric pressure and room temperature in glacial acetic
acid (600 ml) to which 80 ml of pure hydrochloric acid was added. When the hydrogen uptake has
ceased the catalyst was :filtered off and the solution diluted with water (600 ml). The oily layer
was separated, the aqueous layer was neutralised with sodium hydroxide solution and extracted
with ether. The combined products were washed and dried. Ether was distilled off and the residue
which contained some 2-methylcyclohexyl acetate was hydrolysed with potassium hydroxide
in a water- methanol solution. Working up of the crude product yielded 114 g (853%) of 2-methyl- .
cyclohexanol (95% cis-isomer).
cis-2-Methylcyclohexyl 3,5-dinitrobenzoate. To a mixture of the above alcohol (1472 g, 129
mole) and pyridine (300 ml) a solution of 3,5-dinitrobenzoyl chloride (300 g, 13 mole) in benzene
(1000 ml) was a::lded under cooling. After standing for 3 hours, the reaction product was worked
up in the same manner as in the case of trans-2-methylcyclohexyl p-nitrobenzoate. Five-fold re-
crystallisation from methanol yielded 267 g (672%) of the pure cis-ester, m. p. 975 - 980.
1202 Collection Czechoslov. Chern. Cornrnun.
Mechanism of Dehydration of Secondary Alcohols over Alumina Catalyst. II.
cis-2-MethylcyclohexatlOl. The 3,5-dinitrobenzoate (267 g, 0868 mole), potassium hydroxide
(112 g, 2 mole), water (900 ml) and methanol (3400 ml) were refluxed for two hours. The yield
of the pure alcohol was 743 g (75%), b. p. 162/740 mm, r;25 = 172 cPo '
trans-4- Me thylcyclohexanol
4-Methylcyclohexanone was prepared from the crude alcohol in the same way as 2-methyl-
cyc1ohexanone; yield 730%. An alternative preparation by oxidation with chromium trioxide
in acetic acid afforded only 442% of the ketone.
trans-4-Methylcyclohexyl hydrogen phthalate was obtained from 4-methylcyc1ohexanol (com-
position see Table II) and phtalic anhydride in pyridine by the standard method
2o
. The crude
product was purified by four-fold crystallisation from a light petroleum-ethyl acetate mixture
(5: 1). Yield 436% m. p. 1105 -1195.
trans-4-Methylcyclohexanol was prepared from the above ester by hydrolysis with alkali
in 841% yield. B. p. 172-173/760 mm.
trans-4-t-Butylcyclohexanol
4-t-Butylcyclohexanone was obtained from crude 4-t-butylcyc1ohexanol by chromium trioxide
oxidation in acetic acid
21
in 80% yield and purified as the semicarbazone, m. p. 212 (methanol).
The pure ketone has b. p. 92/ 10 mm, m. p. 495.
trans-4-t-Butylcyclohexyl hydrogen phthalate was prepared from the alcohol (see Table II)
by the standard method. Five-fold crystallisation from light petroleum-ethyl acetate (5 : 1) afforded
560% of the pure trans-isomer, m. p. 146.
trans-4-t-ButylcyclohexanoI
21
. The ester was hydrolysed with alkali and the alcohol isolated
by the same method as trans-2-methylcyc1ohexanol. Yield 950%, m. p. 815 -820 (light petro-
leum).
cis-4-t-Butylcyclohexanol
The crude alcohol (Table III) was converted to the hydrogen phtalate, purified and hydrolysed
in the same way as the trans-isomer. Hydrogen phtalate, m. p. 141 -142; cis-4-t-butylcyc10-
hexanol, overall yield 838%, m. p. 81 - 82.
cis-2-t-Butylcyc lohexanol
2-t-Butylcyclohexanone was obtained from the alcohol by oxidation with chromium trioxide
in acetic acid
17
. The crude product (yield 803%) was converted to the semicarbazone, m. p.
181 -182 (methanol). Hydrolysis of the semicarbazone gave the pure ketone in 640% yield;
b. p. 105 -106/38 mm.
cis-2-t-Butylcyclohexyl p-nitrobenzoate. The same method as for cis-2-methylcyc1ohexanol
3,5-dinitrobenzoate was used. Yield 384%, m. p. 885 - 890 (ethanol).
cis-2-t-Buty!cyc!ohexanoI
17
. Alkali hydrolysis of the p-nitro benzoate afforded the pure alcohol
in 897% yield; m. p. 560 - 570 (light petroleum).
Catalyst
The same lot of a y-alumina catalyst as in our previous work
7
was used.
Analytical Methods
The course of the preparation of all compounds and of their purification was control1ed after
each step or operation by gas chromatographic analysis of the products. Glycerol was used as.
the stationary phase, celite as the support, and nitrogen as the carrier gas (see
22
); the temperature
Vol. 27 (1962)
1203
Kochloe/l, Kraus, Chou Chin-Shen, Beranek, Bazant:
of the column and the gas velocity was waried according to the boiling point of the particular
alcohol or ketone.
Conversion in kinetic measurements was calculated from determinations of the unreacted
alcohol by the acetylation method
23
. For the aliphatic and simple cyclic alcohols 25 -27 fold
molar excess of acetic anhydride and one hours reaction time gave satisfactory results. For the
substituted cyclohexanols the acetylation time had to be 15 hour. In the case of cis-2-t-butyl-
cyclohexanol a 13 fold molar excess of acetic anhydride and 5 hours of reaction time were neces-
sary for complete reaction.
Procedure
The apparatus used for kinetic measurement is described in our previous paper
7
The partial
pressure of the reacting alcohol was adjusted by addition of a suitable amount of toluene. As
a sample for analysis the middle fraction of the reaction products (corresponding to steady state
conditions) was separated and pyridine added until the aqueous and toluene layers disappeared .
. With this homogenised sample two or three parallel determinations were performed.
At different temperatures kinetic measurement included usually 4 different parcial pressures
for which two to four separate conversion
determinations were made. An example of
experimental values for trans-2-methylcyclo-
hexanol is presented in Fig. 1. The points
represent averages from two to four conver-
Q 6 . - - - - - - - - . - - - - - - - - - - - - - - - - - ~
x
sion measurements. 0,4
Fig. 1 Q2
Dehydration of trans-2-Methylcyclohexanol
Conversion (x) dependence on reciprocal
space velocity (W/F). 1 193C, 2 204C,
32145C.
0,3
Kinetic Equation
It was ascertained by preliminary experiments that a linear relationship exists between reci-
procal space velocity (contact time) and conversion of the reacting alcohol approximately to
a value of the latter quantity of 04. At constant temperature a single line in conversion against
reciprocal space velocity graphs was obtained for different parcial pressures of the alcohol in the
feed. It was therefore possible to calculate the rate constant k by means of the integral equation
k = xF/W
(1)
where x is the conversion of the alcohol, F the feed rate of the alcohol [mole/h] and W the volume
of the catalyst bed [1].
On the basis of Langmuir-Hinshelwood kinetics of catalytic reactions an equation of this
type can be obtained in two cases: 1. For a irreversible reaction if the rate determining step is
desorption of the products from the surface of the catalyst. In this case, the x- W/ F plot should
be linear in the whole range of conversions; however, this condition is not fulfilled here. 2. For
a irreversible reaction if a) the rate determining step is a surface reaction, b) the alcohol is adsorbed
on a single center, c) the adsorption coefficient of the alcohol is high in general and higher than
1204 . Collection Czechoslov. Chem. Commun.
Mechanism of Dehydration of Secondary Alcohols over Alumina Catalyst. II.
the adsorption coefficient of water, and d) the olefin formed is practically not adsorbed. In such
case it is possible to 'Omit the terms 1, KRPR and KsPs in the basic rate equation
(2)
where K
A
, KR and Ks denote adsorption coefficients of the alcohol, water and the olefin, respec-
tively, and P A' PR and Ps the corresponding partial pressures. Equation (2) simplifies to
r=k (3)
which on integration gives Equation (1).
For higher water contents, i. e. for higher conversions, it is not possible to omit the term KRPR;
this gives
(4a)
After rearrangement we get
(4b)
For confirmation ofthis hypothesis the experimental data of 4-methylcyclohexanol dehydration
from our previous paper
7
were used. The reaction conditions in these experiments were: temper-
ature 215C, partial pressure of the alcohol PA = 0095 atm, dilution with toluene and nitrogen.
In the x- WI F plot the tangent was found for wi F = 0 which practically coincided with a curve
drawn through experimental points up to x = 03. This gave with the help of equation (1) the
value of the k. Additional tangents were measured at points of x = 04 and 0'6, respectively,
and from their values and equation (4b) an estimate of KRIKA ratio was obtained (approximately
0'5). Using this value and the rate constant, the dependence of the x on the wlF was calculated
and good agreement with all experimental points was obtained.
From this we conclude that dehydration of secondary alcohols over our y-alumina catalyst is
governed by a surface reaction as the rate-determining step. This confirms that our kinetic data
described the rate of a chemical reaction and not the rate of adsorption or of any other process.
Results and Discussion
A comparison of the appropriate values for the aliphatic alcohols (Table V and
Fig. 2) shows no substantial influence of chain branching in the neighbourhood of the
hydroxyl group on the rate and activa-
tion energy. This indicates that electronic
effects, which usually play an important
role in liquid phase reactions, have little
influence under our conditions.
Fig. 2
Arrhenius Plots of Rate Constants for Ali-
phatic and Simple Cyclic Alcohols
1 Heptanol-(4), 2 2-methylhexanol-(3), 32,4-
dimethylpentanol-(3), 4 cyclopentanol, 5 cyc-
lohexanol, 6 cycloheptanol, 7 cyclooctanol,
8 2,2-dimethylcyclohexa?ol.
Vol. 27 (1962)
-1,0...--.------.-------.-------.-----,
logk
0,5
o
-0,5
1205
Kochloejl, Kraus, Chou Chin-Shen, Beranek, Baiant:
Table V
Rate Constants and Activation Energies of Secondary Alcohol Dehydration over y-Alumina
k
k
re1
Alcohol for 200
0 E
mole/ h I
for 200
0
kcal / mole
,
Heptanol-(4) 049 10 33
2-Methylhexanol-(3) 051 10 34
2,4-Dimethylpentanol-(3) 065 13 32
Cyclopentanol 095 19 36
Cyclohexanol 051 10 35
Cycloheptanol 119 23 35
Cyclooctanol 437 86
-
2,2-Dimethylcyclohexanol 047 09 34
trans-2- Methylcyclohexanol 092 18 44
trans-4- Methylcyclohexanol 096 19 38
trans-4-t-Buty 1cyclohexanol 105 21 38
cis-2-Methylcyclohexanol 617 121 21
cis-2-t-Buty1cyclohexanol 202 403 19
cis-4-t-ButyIcyclohexanol 692 136 27
A more marked dependence may be observed on the ring size of simple secondary
cyclic alcohols. However, the values of activation energy are practically the same
for all members of this group and differ only slightly from those for the aliphatic
alcohols. The order of reactivity for the cyclic alcohols is in agreement with observ-
ations of ring size influence on some related reactions in solution reported by various
authors
25
- 28. Quantitative treatment of our findings is not yet possible, not even for
the liqujd phase reactions a satisfactory explanation has been presented although
some hypotheses have been proposed
25
,26.
From the similarity between the activation energy values for both aliphatic and
simple cyclic alcohols we assume that the mechanism of the dehydration of this two
groups of substances is identical or, if more mechanisms are in operation, the degrees
of their participation are of the same magnitude.
In studying the dehydration of 2,2-dimethylcyclohexanol we intended to determine
whether a decrease in the number of hydrogen atoms in the neighbourhood of the
hydroxyl group will influence the reaction rate. From comparison of the values i.t;l
Table V for cyclohexanol and its 2,2-dimethyl derivative is evident that the presence
of a quaternary carbon atom diminishes the reactivity only slightly.
It appeared further important to study the dehydration of those alcohols which
have a fixed position of the hydroxyl group in respect to other atoms in the molecule.
This condition is not fulfilled in the case of the aliphatic alcohols where there is no
1206 Collection Czechoslov. Chern. Cornrnun.
Mechanism of Dehydration of Secondary Alcohols over Alumina Catalyst. II.
powerfull restriction to an arbitrary arrangement of the molecule on the surface of the
catalyst; Even in 'the case of unsubstituted cyclic alcohols the position of the hydroxyl
group is not strictly defined because of ready interconversion of different conform at-
ions
29
The same is true for 2,2-dimethylcyclohexanol.
For these reasons we studied the dehydration of some stereoisomeric 2- and 4-alkyl-
cyclohexanols (Table V and Fig. 3). The t-butylcyclohexanols were most suitable
because of their conformational homogeneity: the cis-isomers have the hydroxyl
group in the axial and the t-butyl group in the equatorial positions whereas both
substituents of-the trans-isomers are equatorial. Since the bulky t-butyl group could
exert specific effect on the reaction rate we also examined the dehydration of the cor-
responding 2- and 4-methyl derivates. With these compounds steric conditions are
not so clear-cut; however, deviations
from conformational homogeneity are
small enough not to be taken in account
in the present study.
Fig. 3
Arrhenius Plots of Rate Constants for Ste-
reoisomeric Alky lcyc1ohexanols
1 trans-2-Methyicyc1ohexanol, 2 cis-2-me-
thylcyc1ohexanol, 3 trans-4-t-butylcyc1ohe-
xanol, , 4 cis-4-t-butylcyc1ohexanol, 5 cis-
2-t-butylcyc1ohexanol, 6 trans-4-methy1cyc1o-
hexanol, 7 cyc1ohexanol.
1,0
logk
2
a
' 0,5
2,05
Largest differences in reaction rates and activation energies were found among
stereoisomeric alkylcyclohexanols. Table V shows that on the average the activation
energy of the cis-isomers is substantially lower than that of the trans-isomers. For the
aliphatic and simple cyclic alcohols this quantity has values between those for the
alkylcyclohexanol stereoisomers. Such distinct dissimilarity in activation energies
of individual conformations is clearly connected with a different geometry of reacting
molecules and we regard it as evidence for different mechanisms of dehydration.
One can suppose that for the formation of the activated complex on the surface of
the catalyst a specific steric arrangement of a reacting molecule is necessary. A low
activation energy can then be expected in the case of substances in which a suitable
geometry is largely preformed in their free state. However, a precise picture of the
form of the activated complex is hardly possible to propose on the basis of the pre-
sent data since chemisorbed species and not free molecules are involved in its format-
ion; however, this does not mean than the form of the molecule is totally changed
by chemisorption.
Vol. 27 (1962)
1207
Koc;hloe/l, Kraus, Chou Chin-Shen, Beranek, Bazant:
In the series of trans-alkylcyclohexanols our results shows no effect of alkyl struc-
ture on the rate whereas the opposite has been observed for the cis-compounds.
To explain this fact we assume a different behaviour of the hydrogen atoms vicinal
to the OH group which mayor may not take part in the formation of the activated
complex. The first possibiljty probably holds for the cis-alcohols while no participat-
ion of these hydrogen atoms has to be expected for the trans-isomers. Then for this
second group of alcohols some type of non-synchronous mechanism must be valid;
for example, it may involve the formation of a surface alkoxide like that proposed
by Topcijeva
3o
,31 or the formation of a bridged carbonium ion
17
However, on the
basis of our previous results
7
we exclude the possibility of a classical carbonium
ion mechanism which has been proposed by Brey and Krieger32 to explain dehydra-
tion over solid catalysts.
In the case of the dehydration of cis-alkylcyclohexanols a synchronous mechanism
is probable, for example of the type that has been proposed by Eucken
3
3, or quite
recently by Pines
3
High reaction rate of the cis-2-t-butylcyclohexanol dehydration cannot be explained
solely by suitable geometry of the molecule but steric effects of the bulky alkyl group
have evidently also to be considered. In this Laboratory a similar phenomenon was
observed in the case of the catalytic dealkyla60n of 2-t-butylphenoI
34
.
Unfortunately, a comparison of our results with corresponding measurements of
dehydration in the liquid phase is not possible as no equivalent study has been pub-
lished until now, although a number of papers has been devoted to the mechanism
of the homogeneous dehydration of alcohols
2
,17,3s-38. Growing evidence from our
and other authors' kinetic studies indicates (see
34
,39-42) that specific structure effects
having no direct analogy to reactions in solution may playa decisive role in hetero-
geneous catalytic reactions.
We wish to thank Dr J. Sicher Institute of Organic Chemistry and Biochemistry, Prague, for
helpful discussions in the course of our work and in interpretation of results.
We are further indebted to Dr R. Komers for gas-chromatographic analysis and to Miss V. Miko-
to va for technical assistance.
References
1. Winfield M. E.: Catalysis, Vol. VII (P. H. Emmet ed.) p. 93. Reinhold, New York 1960.
2. Naro P. A., Dixon J. A.: J. Am. Chern. Soc. 81, 1681 (1959).
3. Pines H.: J. Am. Chern. Soc. 82, 2401 (1960).
4. Bork A., Tolstopjatova A. A.: Acta Physicochim. URSS 8, 603 (1938).
5. Miller D. N.: Thesis. University of Wisconsin 1955.
6. Laible J. R.: ThesiS. University of Wisconsin 1958.
7. Beranek L., Kraus M., Kochloefl K., Bazant V.: This Journal 25, 2513 (1960).
8. Nenitcescu c., Ionescu C. N.: Bull. soc. chim. Romania 14, 65 (1932).
9. Smith P., Bauer D. R.: J. Am. Chem. Soc. 74, 6135 (1952).
10. De Boer J. Th., Backer J. H.: Org. Syntheses 34, 24 (1954).
11. Kohler E. P., Tishler M., Potter H., Thomson H. T.: J. Am. Chern. Soc. 61, 1057 (1939).
12. Bailey W., Medoff M.: J. Am. Chern. Soc. 76, 2708 (1954).
1208 Collection Czechoslov. Chern. Cornrnun.
Mechanism 0/ Dehydration 0/ Secondary Alcohols over Alumina Catalyst. II.
13. Whitmore F. C., Johnston F.: J. Am. Chern. Soc. 60, 2265 (1938).
14. Bachmann W. F., Rauno E. K.: J. Am. Chern. Soc. 72,2530 (1950).
15. Dillon R. T.: J . Am. Chern. Soc. 50, 1712 (1928).
16. Conant J. B., Blatt A. H.: J. Am. Chern. Soc. 51, 1227 (1929).
17. Goering H. L., Reeves R., Espy H. H.: J. Am. Chern. Soc. 78, 4926 (1956).
18. Huckel W., Hubelle A.: Ann. 613, 137 (1958).
19. Houben-Weyl: Methoden der Organischen Chemie 4/2, p. 165, IV. Ed. Thieme, Stutt-
gart 1955.
20. Stork G., White W. N.: J. Am. Chern. Soc. 78, 4604 (1965).
21. Winstein S., Holness N. J.: J. Am. Chern. Soc. 77, 5562 (1955).
22. Komers R., Kochloefi K., Bazant Y.: Chern. & Ind. (London) 1958, 1405.
23. Siggia S.: Quantitative Organic Analysis via Functional Groups, p. 4. Wiley, New York 1949.
24. Hougen .O. A., Watson K. H.: Chemical Process Principles, Part III. Wiley, New York 1947.
25. Brown H. c., Fletcher R. S., Johannesen R. B.: J. Am. Chern. Soc. 73, 212 (1951).
26. Brown H. c., Ham G.: J. Am. Chern. Soc. 78, 2738 (1956).
27. Kuivila H. G., Becker W. J.: J. Am. Chern. Soc. 74, 5329 (1952).
28. Mares F., Rocek J., Sieher J.: This Journal 26, 2355 (1961).
29. Eliel E. L.: J. Chern. Educ. 32, 126 (1960).
30. Topcieva K. V., Jun-Pin K.: Z. fiz. chim. 29, 1854 (1955).
31. Topcieva K. V., Jun-Pin K.: Z. fiz. chim. 29, 2076 (1955).
32. Brey W. S., Krieger K. A.: J. Am. Chern. Soc. 71, 3637 (1949).
33. Eucken A., Wicke E.: Naturwiss. 32, 165 (1945).
34. Schneider P., Kraus M., Bazant V.: This Journal, 27, 9 (1962).
35. Chiurdoglu G.: Bull. soc. chirn. Belges 47, 241 (1938).
36. Vavon G.: Bull. soc. chirn. [4] 49, 937 (1933).
37. Price Ch. C., Karabinos J. V.: J. Am. Chern. Soc. 62, 1159 (1940).
38. Schaeffer H. J., Collins C. J.: J. Am. Chern. Soc. 78, 124 (1956).
39. Schneider P., Kraus M., Bazant V.: This Journal 26, 1636 (1961).
40. Kadlec J . , Jost F., Bazant V.: This Journal 26, 818 (1961).
41. Kadlec J., Bazant V.: This Journal 26, 1201 (1961).
42. Rase H. F., Kirk R. S.: Chern. Eng. Progr. 50, 35 (1954).
Translated by the Author (M. K.).
PellOMe
K. KoxJIe<pJIb, M. Kpayc, "Ioy "IHH-illeH, JI. EepaHeK H B. Ea)KaHT: 0 MeXaltU3Me oe-
zuOpamaquu 6mOpUllltblX cnupm06 Ita ZilUlt03eMltOM KamallU3amope. II. Bllulllt.ue cmpoeltUIl Ita CKO-
pocmb oezuopamaquu. Onpe.n;eJIeHO BJlIDIHlIe CTpoeHmr 14 BTOPH'iHbIX CllHPTOB Ha CKOPOCTb HX .n;e-
rH.n;paTa:l(HH Ha y-rJIHH03eMe. HCCJIe.n;oBaHHlO no.n;BepraJIHCb HeKOTopble aJIH<paTH'ieCme CllHPTbI,
IUiKJIH'ieCKHe cnHpTbI c pa3JIH'iHOH BeJIH':IHHOH KOJIen; H HeKOTopbIe CTepeOH30MepHbIe aJIKHJII.J;HKJIO-
reKCaHOJIbI. On;eHeHbI 3HeprHH aKTHBaI.J;HH H peaKI.J;HOHHaH cnoco6HoCTb cnHpTOB cpaBHeHa Ha OCHO-
Bamm KOHCTaHT CKOPOCTH peaKn;HH npH 200C. 06cY)K.n;eHbI BJIHHHHe 3aMemeHHH Ha coce.n;HeM aTO-
Me ymepo.n;a, BeJIH':IHHbI KOJIbIn;a H npOCTpaHCTBeHHOH KOH<pHrypaI.J;HH pearHPYIOmeH MOJIeKYJIbI.
Pe3YJIbTaTbI CBH,l1;eTeJIbCTBYIOT 0 TOM, 'ITO reOMeTpHH pearHpyromeH MOJIeKYJIbI HMeeT BecbMa
Ba)l(HOe 3Ha'IeHHe npH .n;erH.n;paTan;HH cnHpTOB Ha TBep.n;bIX KaTaJIH3aTopax.
Vol. 27 (1962)
1209
Potrebbero piacerti anche
- A Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryDa EverandA Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryValutazione: 3.5 su 5 stelle3.5/5 (231)
- The Sympathizer: A Novel (Pulitzer Prize for Fiction)Da EverandThe Sympathizer: A Novel (Pulitzer Prize for Fiction)Valutazione: 4.5 su 5 stelle4.5/5 (121)
- Grit: The Power of Passion and PerseveranceDa EverandGrit: The Power of Passion and PerseveranceValutazione: 4 su 5 stelle4/5 (588)
- Devil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaDa EverandDevil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaValutazione: 4.5 su 5 stelle4.5/5 (266)
- The Little Book of Hygge: Danish Secrets to Happy LivingDa EverandThe Little Book of Hygge: Danish Secrets to Happy LivingValutazione: 3.5 su 5 stelle3.5/5 (399)
- Never Split the Difference: Negotiating As If Your Life Depended On ItDa EverandNever Split the Difference: Negotiating As If Your Life Depended On ItValutazione: 4.5 su 5 stelle4.5/5 (838)
- Shoe Dog: A Memoir by the Creator of NikeDa EverandShoe Dog: A Memoir by the Creator of NikeValutazione: 4.5 su 5 stelle4.5/5 (537)
- The Emperor of All Maladies: A Biography of CancerDa EverandThe Emperor of All Maladies: A Biography of CancerValutazione: 4.5 su 5 stelle4.5/5 (271)
- The Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeDa EverandThe Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeValutazione: 4 su 5 stelle4/5 (5794)
- The World Is Flat 3.0: A Brief History of the Twenty-first CenturyDa EverandThe World Is Flat 3.0: A Brief History of the Twenty-first CenturyValutazione: 3.5 su 5 stelle3.5/5 (2259)
- The Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersDa EverandThe Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersValutazione: 4.5 su 5 stelle4.5/5 (344)
- Team of Rivals: The Political Genius of Abraham LincolnDa EverandTeam of Rivals: The Political Genius of Abraham LincolnValutazione: 4.5 su 5 stelle4.5/5 (234)
- The Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreDa EverandThe Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreValutazione: 4 su 5 stelle4/5 (1090)
- Hidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceDa EverandHidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceValutazione: 4 su 5 stelle4/5 (895)
- Her Body and Other Parties: StoriesDa EverandHer Body and Other Parties: StoriesValutazione: 4 su 5 stelle4/5 (821)
- Elon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureDa EverandElon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureValutazione: 4.5 su 5 stelle4.5/5 (474)
- The Unwinding: An Inner History of the New AmericaDa EverandThe Unwinding: An Inner History of the New AmericaValutazione: 4 su 5 stelle4/5 (45)
- The Yellow House: A Memoir (2019 National Book Award Winner)Da EverandThe Yellow House: A Memoir (2019 National Book Award Winner)Valutazione: 4 su 5 stelle4/5 (98)
- On Fire: The (Burning) Case for a Green New DealDa EverandOn Fire: The (Burning) Case for a Green New DealValutazione: 4 su 5 stelle4/5 (73)
- Ammonia Plant Basic EquationsDocumento87 pagineAmmonia Plant Basic Equationschichosango100% (2)
- O LVL Chem Definitions ListDocumento6 pagineO LVL Chem Definitions Listacsbr4science170% (10)
- Photocatalytic CementDocumento30 paginePhotocatalytic CementRehan Ravi100% (1)
- TOPSOE Seminar - Catalysts and Reactions PDFDocumento132 pagineTOPSOE Seminar - Catalysts and Reactions PDFAhmad Nakash100% (3)
- A Review On Biodiesel Production Using Catalyzed TransesterificationDocumento13 pagineA Review On Biodiesel Production Using Catalyzed TransesterificationAndre HaywardNessuna valutazione finora
- MbocDocumento1 paginaMbocIrek HabryloNessuna valutazione finora
- U WritingDocumento1 paginaU WritingIrek HabryloNessuna valutazione finora
- Find Locations: Ship A Package Send Mail Manage Your Mail Shop Business Solutions Quick ToolsDocumento2 pagineFind Locations: Ship A Package Send Mail Manage Your Mail Shop Business Solutions Quick ToolsIrek HabryloNessuna valutazione finora
- Find Locations: Ship A Package Send Mail Manage Your Mail Shop Business Solutions Quick ToolsDocumento2 pagineFind Locations: Ship A Package Send Mail Manage Your Mail Shop Business Solutions Quick ToolsIrek HabryloNessuna valutazione finora
- AcknowledgmentsDocumento2 pagineAcknowledgmentsIrek HabryloNessuna valutazione finora
- Neural Substrates of Awakening Probed With Optogenetic Control of Hypocretin Neurons / Antoine Adamantidis / Nature 2007Documento1 paginaNeural Substrates of Awakening Probed With Optogenetic Control of Hypocretin Neurons / Antoine Adamantidis / Nature 2007Irek HabryloNessuna valutazione finora
- Track MapDocumento1 paginaTrack MapIrek HabryloNessuna valutazione finora
- Amadeus TranscriptDocumento55 pagineAmadeus TranscriptIrek HabryloNessuna valutazione finora
- Monday Through Friday: Eastbound WestboundDocumento4 pagineMonday Through Friday: Eastbound WestboundIrek HabryloNessuna valutazione finora
- Eula Microsoft Visual StudioDocumento3 pagineEula Microsoft Visual StudioqwwerttyyNessuna valutazione finora
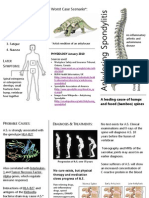
- Physio AnkylosingDocumento2 paginePhysio AnkylosingIrek HabryloNessuna valutazione finora
- Lab 3 KineticsDocumento6 pagineLab 3 KineticsMichelle SamayoaNessuna valutazione finora
- Influence of Metal Salts On Bitumen Oxidation: Speros E. Moschopedis and James G. SpeightDocumento6 pagineInfluence of Metal Salts On Bitumen Oxidation: Speros E. Moschopedis and James G. SpeightLarissa PimentaNessuna valutazione finora
- GEN BIO EnzymesDocumento17 pagineGEN BIO EnzymesTurn2ndTurN P.ONessuna valutazione finora
- Ethylene Propylene Rubbers (Epdm) Dry Chart: Ordered by Ethylene Content and Af NityDocumento6 pagineEthylene Propylene Rubbers (Epdm) Dry Chart: Ordered by Ethylene Content and Af NityvibhuNessuna valutazione finora
- MSDW Mwi Maxsat WebpagepdfDocumento5 pagineMSDW Mwi Maxsat Webpagepdfdonald55555Nessuna valutazione finora
- Arti Ficial Photosynthesis at E Ciencies Greatly Exceeding That of Natural PhotosynthesisDocumento6 pagineArti Ficial Photosynthesis at E Ciencies Greatly Exceeding That of Natural PhotosynthesisAudry F. DewiNessuna valutazione finora
- Cigre - 2 - 2015 PDFDocumento10 pagineCigre - 2 - 2015 PDFbenlahnecheNessuna valutazione finora
- Chapter 21 22 - Chemical Reactions Balancing Chemical EquationsDocumento34 pagineChapter 21 22 - Chemical Reactions Balancing Chemical EquationsAlwielland BelloNessuna valutazione finora
- Catalysts 07 00323 v2Documento27 pagineCatalysts 07 00323 v2Thaís OliveiraNessuna valutazione finora
- Selective Catalytic Dehydrogenation of Alkanes To Alkenes: Mark J. Burk and Robert H. CrabtreeDocumento8 pagineSelective Catalytic Dehydrogenation of Alkanes To Alkenes: Mark J. Burk and Robert H. CrabtreeAnupama D. MishraNessuna valutazione finora
- What Are ZeolitesDocumento26 pagineWhat Are ZeolitesborgiamatriceNessuna valutazione finora
- F5 Chapter 1 Rate of ReactionDocumento18 pagineF5 Chapter 1 Rate of ReactionSiti Aishah ZolkanainNessuna valutazione finora
- Cracking Hydrocarbons: 1 of 28 © Boardworks LTD 2016Documento28 pagineCracking Hydrocarbons: 1 of 28 © Boardworks LTD 2016Nuro Josine JosineNessuna valutazione finora
- Biochemistry QuestionsDocumento7 pagineBiochemistry QuestionsCatherine JennensNessuna valutazione finora
- Wang 2012Documento5 pagineWang 2012Carmen AguilarNessuna valutazione finora
- Principles of Heterogeneous Catalysis CHE633A, IIT Kanpur: Course Conduct and Content Introduction and A Bit of HistoryDocumento13 paginePrinciples of Heterogeneous Catalysis CHE633A, IIT Kanpur: Course Conduct and Content Introduction and A Bit of HistoryRajendra SahuNessuna valutazione finora
- Phosphoric Acid Fuel Cell (Pafc)Documento10 paginePhosphoric Acid Fuel Cell (Pafc)Else Feba Paul0% (1)
- CH 15Documento51 pagineCH 15IshNessuna valutazione finora
- Module 1 Kinetics of Materials Reading MaterialsDocumento8 pagineModule 1 Kinetics of Materials Reading MaterialsmayNessuna valutazione finora
- Transesterification Reaction and ComparativeDocumento13 pagineTransesterification Reaction and ComparativeKa HinaNessuna valutazione finora
- Regioselective Acylation, Alkylation, Silylation, and Glycosylation of MonosaccharidesDocumento37 pagineRegioselective Acylation, Alkylation, Silylation, and Glycosylation of MonosaccharidesRiaz LourencoNessuna valutazione finora
- Structure, Functionality and Tuning Up of LaccasesDocumento17 pagineStructure, Functionality and Tuning Up of LaccasesjwalantkbhattNessuna valutazione finora
- Che QP 5Documento20 pagineChe QP 5Shreeranga RbNessuna valutazione finora
- UNIT 5 WorksheetDocumento10 pagineUNIT 5 WorksheetlorNessuna valutazione finora
- 2009 Evaluation of Tritium Removal and Mitigation Technologies For Wastewater TreatmentDocumento51 pagine2009 Evaluation of Tritium Removal and Mitigation Technologies For Wastewater TreatmentugyaNessuna valutazione finora