Documenti di Didattica
Documenti di Professioni
Documenti di Cultura
Biology - Toothpickase Lab
Caricato da
lanichungCopyright
Formati disponibili
Condividi questo documento
Condividi o incorpora il documento
Hai trovato utile questo documento?
Questo contenuto è inappropriato?
Segnala questo documentoCopyright:
Formati disponibili
Biology - Toothpickase Lab
Caricato da
lanichungCopyright:
Formati disponibili
Lani Chung Period 2B September 13, 2011
Toothpickase Lab
Part A: Rate of Product Formation in an Enzyme-Facilitated Reaction: 4. Graph the results.
Rate of Product Formation in an Enzyme-Facilitated Reaction 80 80 80 79 77 70 62 60 49 56
80 80
80
Number of Toothpicks Catalyzed
40 29 19 10
39
20
0 20 40 60 80 100 120 140 160 180 200 220 240 260 280 300 Time in Seconds
5. Calculate the rate of enzyme action in toothpicks per second for each 60 second interval: How many toothpicks were broken after 1 minute, 2 minutes, 3 minutes, etc. For the first interval about 0.48 toothpicks were broken per second, in the second interval about 0.45 toothpicks were broken per second, in the third interval about 0.35 toothpicks were broken per second, in the fourth interval about 0.05 toothpicks were broken per second, and in the last interval no toothpicks were broken because all 80 toothpicks were already broken. After one minute 29 toothpicks were broken, after two minutes 56 toothpicks were broken, after three minutes 77 toothpicks were broken, after four minutes 80 toothpicks were broken, and after five minutes 80 toothpicks were broken. 6. Discuss your results under the graph. Explain what happens to the rate and summarize enzyme activity. Our results showed that enzyme activity steadily increased about 10 toothpicks for every 20 second interval until in neared about 200 seconds because there werent enough toothpicks left to catalyze. Therefore, the rate stopped increasing and stayed at 80 toothpicks from the 220 second mark to the 300 second mark since
all toothpicks were already catalyzed. This means that enzyme activity will steadily increase over time until it runs out of substrates to catalyze and has a stabilized rate of increase. Part B: Effect of Substrate Concentration on Reaction Rate: 4. Graph the results.
Effect of Substrate Concentration on Reaction Rate 12 11 10 10 9 8 8 8 8 7 10
Number of Toothpicks Catalyzed (In 20 Second Intervals)
7 6
0 10 20 30 40 50 60 70 80 90 100 Number of Toothpicks
5. Discuss your results and explain why the rates were different at different concentrations. Summarize the effect of substrate concentration on enzyme action. For the first six trials, I saw a steady increase in the rate of enzyme activity. But in the next trials, the rate jumped up and down from 10 to 8 to 11 to 7 to 10. Although my results showed consistency in the beginning, it was inconsistent towards the last trials. The rates were different at different concentrations because the toothpickase person generally had a harder time finding toothpicks when there was a low concentration of toothpicks while they had an easier time finding toothpicks when there was a high concentration of toothpicks. This is because when there is a high ratio or concentration of toothpicks mixed in with the twist ties, there is a higher probably that you would find a toothpick and vice versa. Because of this, when substrate concentration is high, is may cause enzyme action to increase while low substrate concentrations might cause enzyme action to decrease.
Part C: Effect of Temperature Substrate Concentration on Reaction Rate: 3. Calculate the rate of enzyme action in toothpicks per second. Compare the two rates. The rate of enzyme action for hands in iced water was about 0.36 toothpicks per second while the rate of enzyme action for hands that werent in iced water was about 0.48 toothpicks per second. The rate for hands in iced water was a little lower than that of the hands not in iced water, showing that the hands that werent in iced water performed faster. 4. Discuss your results and explain why the rates were different at different temperatures. Summarize the effect of temperature on enzyme action. My results showed that hands placed in iced water could break 10 toothpicks in 28 seconds while hands not placed in iced water could break 10 toothpicks in 21 seconds. Because hands that are placed in iced water lose feeling and are often very painful, it is harder to do a task than using hands not affected by the cold. This is why the rates were different at different temperatures. This also goes for enzyme action because if an enzyme is not at its optimum temperature, it cannot perform as well as it could if it were in an ideal temperature. In this case, the cold would slow down the molecules, cause less collisions, and as a result cause less reactions. a) Explain what would happen to an enzyme-facilitated reaction if temperature were increased. Be sure to include the effect if temperature were increased to 100 degrees Celsius. If the temperature in an enzyme-facilitated reaction increased, the enzymes might be able to react more often and perform faster because heat makes molecules more faster, causing more collisions and reactions. However if the temperature reached 100 degrees Celsius, it is quite possible that the enzyme wouldnt be able to handle the heat. This could cause the enzyme to lose its shape and get denatured, making it lose its ability to work. b) What is the optimal temperature (degrees Celsius) for enzymes in the human body? The optimal temperature for enzymes in the human body is 37 degrees Celsius or 96.8 degrees Fahrenheit which is body temperature.
Potrebbero piacerti anche
- Enzyme Lab Temperature and Concentration EffectsDocumento6 pagineEnzyme Lab Temperature and Concentration EffectsDJ ISAACSNessuna valutazione finora
- Toothpickase LabDocumento2 pagineToothpickase Labjoannekimm150% (1)
- OfficialtoothpickthingDocumento3 pagineOfficialtoothpickthingapi-275583436Nessuna valutazione finora
- Toothpickase Lab BortzyDocumento4 pagineToothpickase Lab BortzyJunaid SiddiqiNessuna valutazione finora
- Enzyme Activity Lab ResultsDocumento6 pagineEnzyme Activity Lab Resultsrsenser2100% (2)
- Exercise 4:: Organic Components: CarbohydratesDocumento20 pagineExercise 4:: Organic Components: CarbohydratespikachuzingungaNessuna valutazione finora
- Interpreting and Constructing Cladograms LabDocumento5 pagineInterpreting and Constructing Cladograms Labapi-375285021100% (1)
- Experiment 3: Le Chatelier's PrincipleDocumento4 pagineExperiment 3: Le Chatelier's PrinciplespaghetticurlersNessuna valutazione finora
- Enzyme Action Testing Catalase Activity Lab ReportDocumento4 pagineEnzyme Action Testing Catalase Activity Lab ReportAnthony100% (3)
- 02 01 Properties of Water Lab ReportDocumento3 pagine02 01 Properties of Water Lab ReportkyleaNessuna valutazione finora
- Lab Introduction To Gel ElectrophoresisDocumento3 pagineLab Introduction To Gel Electrophoresisapi-382372564100% (1)
- Ch27 Sample PreparationDocumento74 pagineCh27 Sample PreparationkevsayeghNessuna valutazione finora
- Lab 3 Le Chatelier's Principle - and Chemical Equilibrium BCCDocumento9 pagineLab 3 Le Chatelier's Principle - and Chemical Equilibrium BCCXavier BuenoNessuna valutazione finora
- Formal Lab Report Chem 113Documento22 pagineFormal Lab Report Chem 113api-367122872100% (1)
- UST College of Science Analytical Chemistry 3 Laboratory:, 36 PagesDocumento3 pagineUST College of Science Analytical Chemistry 3 Laboratory:, 36 PagesKENT BENEDICT PERALESNessuna valutazione finora
- EXPT 2 LAB REPORT Extraction and Characterization of Proteins Group 2Documento11 pagineEXPT 2 LAB REPORT Extraction and Characterization of Proteins Group 2Teach LesnitoNessuna valutazione finora
- Biology Lab Report 4Documento5 pagineBiology Lab Report 4ai_eiNessuna valutazione finora
- Egg Osmosis LabDocumento4 pagineEgg Osmosis Labapi-371081506Nessuna valutazione finora
- Effect of Temperature On The Reaction RateDocumento5 pagineEffect of Temperature On The Reaction RateChristy Joy RetanalNessuna valutazione finora
- IB Bio 1 Lab Mitosis in Onion Root Tip CellsDocumento3 pagineIB Bio 1 Lab Mitosis in Onion Root Tip CellsDani RodriguezNessuna valutazione finora
- The Effect of Substrate Concentration On The Activity of EnzymesDocumento7 pagineThe Effect of Substrate Concentration On The Activity of Enzymesjosephine100% (1)
- Kinetics LabDocumento12 pagineKinetics LabJesseNessuna valutazione finora
- Web Site 3 Module 4 Worksheet - Website Evaluation 2Documento10 pagineWeb Site 3 Module 4 Worksheet - Website Evaluation 2api-548496615Nessuna valutazione finora
- Lab 4 Determination of An Equilibrium Constant 1Documento7 pagineLab 4 Determination of An Equilibrium Constant 1Mohammad IzadiNessuna valutazione finora
- Titration of Acetic Acid in VinegarDocumento3 pagineTitration of Acetic Acid in VinegarChristi ViajeNessuna valutazione finora
- Chem 17 Formal ReportDocumento4 pagineChem 17 Formal Reportchemeister111Nessuna valutazione finora
- The Rate Laws of An Iodine Clock ReactionDocumento10 pagineThe Rate Laws of An Iodine Clock Reactionboria11Nessuna valutazione finora
- Chemistry of Living Organisms Measuring PH Lab Activity PDFDocumento6 pagineChemistry of Living Organisms Measuring PH Lab Activity PDFHaris KhanNessuna valutazione finora
- Back Titration of ASPIRINDocumento9 pagineBack Titration of ASPIRINJylla Angway100% (1)
- Observing cyanobacteria Oscillatoria and Anabaena under microscopeDocumento2 pagineObserving cyanobacteria Oscillatoria and Anabaena under microscopeNabilah SyafiqahNessuna valutazione finora
- Ap Photosynthesis LabDocumento6 pagineAp Photosynthesis Labapi-382372564Nessuna valutazione finora
- Acid Base InvestigationDocumento3 pagineAcid Base InvestigationJonathan IrwantoNessuna valutazione finora
- Ap Cellular Respiration LabDocumento8 pagineAp Cellular Respiration Labapi-382372564Nessuna valutazione finora
- 105 Lab ReportDocumento4 pagine105 Lab ReportDan WooNessuna valutazione finora
- Enzymes Lab ReportDocumento11 pagineEnzymes Lab Reportcodybearden100% (2)
- Lab Report Sds-Page WB - PT 1 (1-5)Documento5 pagineLab Report Sds-Page WB - PT 1 (1-5)Ezad juferiNessuna valutazione finora
- Fermentation Lab Report Example With Guidelines To Write Lab ReportsDocumento6 pagineFermentation Lab Report Example With Guidelines To Write Lab ReportsbellasyazanaNessuna valutazione finora
- Egg Osmosis Lab ResultsDocumento8 pagineEgg Osmosis Lab ResultsJueNessuna valutazione finora
- Experiment III Lab Report (Alcohol in Wine)Documento6 pagineExperiment III Lab Report (Alcohol in Wine)Clayton VerBerkmös50% (2)
- AP Biology Enzyme Lab ReportDocumento3 pagineAP Biology Enzyme Lab ReportPatrick100% (6)
- EFFECTS OF PH AND TEMPERATURE ON THE REACTION RATES OF ENZYMECATALYZED REACTIONSDocumento5 pagineEFFECTS OF PH AND TEMPERATURE ON THE REACTION RATES OF ENZYMECATALYZED REACTIONSJocee Mae EbdaniNessuna valutazione finora
- Amylase Experiment Lab ReportDocumento7 pagineAmylase Experiment Lab ReportCHLOE IANNAH CALVADORESNessuna valutazione finora
- Microbiology Practical: Isolating Bacteria Using Differential and Selective MediaDocumento31 pagineMicrobiology Practical: Isolating Bacteria Using Differential and Selective MediaAyman ElkenawyNessuna valutazione finora
- Bio Paper FinalDocumento8 pagineBio Paper FinalNiem PhamNessuna valutazione finora
- Heart Dissection, Lab Report GuideDocumento5 pagineHeart Dissection, Lab Report GuideJohn OsborneNessuna valutazione finora
- AP Biology Lab Two: Enzyme CatalysisDocumento4 pagineAP Biology Lab Two: Enzyme CatalysisCoolAsianDude95% (37)
- The Effect of Substrate Concentration PracDocumento4 pagineThe Effect of Substrate Concentration PracjimslibraryNessuna valutazione finora
- Effect of pH on Catalase Reaction RateDocumento10 pagineEffect of pH on Catalase Reaction Rateanon0% (1)
- Enzymes Lab Report - Activty 8Documento6 pagineEnzymes Lab Report - Activty 8JengNessuna valutazione finora
- CHEM120 OL Solution PreparationDocumento2 pagineCHEM120 OL Solution PreparationYsabela Dela PazNessuna valutazione finora
- Lab ReportDocumento7 pagineLab ReportAlliedschool DefencecampusNessuna valutazione finora
- DNA Exraction LabDocumento5 pagineDNA Exraction Labssullivan611Nessuna valutazione finora
- Intro - Yeast Fermentation by SugarDocumento2 pagineIntro - Yeast Fermentation by SugarMis SheilaNessuna valutazione finora
- Enzyme Lab Report - Effect of Time on Hydrogen Peroxide DecompositionDocumento7 pagineEnzyme Lab Report - Effect of Time on Hydrogen Peroxide DecompositionRob Becerra100% (1)
- Chemical Composition of Cells RevealedDocumento5 pagineChemical Composition of Cells RevealedJustin Riel63% (8)
- Pre Test Yeast and Glucose ExperimentDocumento4 paginePre Test Yeast and Glucose ExperimentAisya Omeira100% (1)
- Analysis of A Commercial Bleach LabDocumento7 pagineAnalysis of A Commercial Bleach Labapi-358133276100% (2)
- Acid-Base Indicators Spectrophotometric Ka LabDocumento6 pagineAcid-Base Indicators Spectrophotometric Ka Labmuskaan0% (2)
- Lab 10 Toothpickase 2005Documento2 pagineLab 10 Toothpickase 2005S. SpencerNessuna valutazione finora
- Enzyme Station Lab 1Documento21 pagineEnzyme Station Lab 1api-233187566Nessuna valutazione finora
- Letter To The EditorDocumento1 paginaLetter To The EditorlanichungNessuna valutazione finora
- Chemistry - GraphDocumento1 paginaChemistry - GraphlanichungNessuna valutazione finora
- Bible Study WorshipDocumento2 pagineBible Study WorshiplanichungNessuna valutazione finora
- B'More - Photograph CritiquesDocumento1 paginaB'More - Photograph CritiqueslanichungNessuna valutazione finora
- Chemistry - GraphDocumento1 paginaChemistry - GraphlanichungNessuna valutazione finora
- FHPM - Employer Mandate SummaryDocumento2 pagineFHPM - Employer Mandate SummarylanichungNessuna valutazione finora
- Analyzing Concepts of "HomeDocumento6 pagineAnalyzing Concepts of "HomelanichungNessuna valutazione finora
- Genetics Limerick Explains DNA, Trait InheritanceDocumento1 paginaGenetics Limerick Explains DNA, Trait InheritancelanichungNessuna valutazione finora
- Biology - North Pole Cell AnalogyDocumento1 paginaBiology - North Pole Cell AnalogylanichungNessuna valutazione finora
- TOK - EssayDocumento6 pagineTOK - EssaylanichungNessuna valutazione finora
- Biology - The Genetics of Parenthood AnalysisDocumento2 pagineBiology - The Genetics of Parenthood Analysislanichung100% (5)
- Biology - The Genetics of Parenthood AnalysisDocumento2 pagineBiology - The Genetics of Parenthood Analysislanichung100% (5)
- B'More - Photograph CritiquesDocumento1 paginaB'More - Photograph CritiqueslanichungNessuna valutazione finora
- Biology - Spring Break Reading AssignmentDocumento17 pagineBiology - Spring Break Reading AssignmentlanichungNessuna valutazione finora
- Black and White Perspective PortfolioDocumento1 paginaBlack and White Perspective PortfoliolanichungNessuna valutazione finora
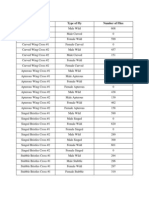
- Biology - Fly Lab Raw Data CollectionDocumento3 pagineBiology - Fly Lab Raw Data CollectionlanichungNessuna valutazione finora
- Chung 1 Reaction Time Lab AnalysisDocumento3 pagineChung 1 Reaction Time Lab Analysislanichung67% (3)
- Biology - Heart Rate Inquiry LabDocumento2 pagineBiology - Heart Rate Inquiry LablanichungNessuna valutazione finora
- Biology - Cell LimerickDocumento1 paginaBiology - Cell LimericklanichungNessuna valutazione finora
- Biology - Genetics Review 2Documento15 pagineBiology - Genetics Review 2lanichung100% (1)
- Genetics Limerick Explains DNA, Trait InheritanceDocumento1 paginaGenetics Limerick Explains DNA, Trait InheritancelanichungNessuna valutazione finora
- Expository Writing - Assignment 1.2 RevisionsDocumento3 pagineExpository Writing - Assignment 1.2 RevisionslanichungNessuna valutazione finora
- Biology - Diffusion Through Membranes Lab Analysis QuestionsDocumento2 pagineBiology - Diffusion Through Membranes Lab Analysis Questionslanichung0% (3)
- Biology - Fly Lab ResultsDocumento17 pagineBiology - Fly Lab ResultslanichungNessuna valutazione finora
- Biology - Enzyme Lab ConclusionDocumento4 pagineBiology - Enzyme Lab ConclusionlanichungNessuna valutazione finora
- Biology - DNA MessageDocumento1 paginaBiology - DNA MessagelanichungNessuna valutazione finora
- Biology - Design A FlowerDocumento1 paginaBiology - Design A FlowerlanichungNessuna valutazione finora
- Chung Assignment 2.3Documento2 pagineChung Assignment 2.3lanichungNessuna valutazione finora
- Expository Writing - Writing Assignment 3.2Documento2 pagineExpository Writing - Writing Assignment 3.2lanichungNessuna valutazione finora
- Design Criteria - Senior High - Mechanical (ACMV)Documento9 pagineDesign Criteria - Senior High - Mechanical (ACMV)Jethro AbanadorNessuna valutazione finora
- Cambridge International Examinations Cambridge International General Certificate of Secondary EducationDocumento24 pagineCambridge International Examinations Cambridge International General Certificate of Secondary EducationWENDY MENDIOLANessuna valutazione finora
- Mecschsyll PDFDocumento161 pagineMecschsyll PDFVinay V GowdaNessuna valutazione finora
- Ficha Tecnica de Los Inverter Yhke Hasta 3 Tons.Documento4 pagineFicha Tecnica de Los Inverter Yhke Hasta 3 Tons.Proyectos Instalaciones RAVENessuna valutazione finora
- Thermodynamics: Chapter 2 Topic 1Documento9 pagineThermodynamics: Chapter 2 Topic 1April Galope OlaliaNessuna valutazione finora
- Solving A Conjugate Heat Transfer Problem Using ANSYS FluentDocumento34 pagineSolving A Conjugate Heat Transfer Problem Using ANSYS Fluentjose antonioNessuna valutazione finora
- Heat Conduction - Basic ResearchDocumento362 pagineHeat Conduction - Basic ResearchJosé Ramírez100% (1)
- CTV Part1Documento39 pagineCTV Part1تيسير صادق تركيNessuna valutazione finora
- Fujitsu ACDocumento16 pagineFujitsu ACNora SmithNessuna valutazione finora
- Polo Cool Pc53 Pc44 ManualDocumento19 paginePolo Cool Pc53 Pc44 Manualjuniperharru8Nessuna valutazione finora
- Boiling Point Elevation of Sucrose Solutions.109125520Documento12 pagineBoiling Point Elevation of Sucrose Solutions.109125520Mahesh SalotagiNessuna valutazione finora
- ADVANCED THERMODYNAMICS COURSEDocumento2 pagineADVANCED THERMODYNAMICS COURSEVivek VenugopalNessuna valutazione finora
- Gay-Lussac's Pressure-Temperature RelationshipDocumento20 pagineGay-Lussac's Pressure-Temperature RelationshipMichelle EscalienteNessuna valutazione finora
- Tianjin Jieda Temperature Instruments Factory Bimetallic Thermometer SpecsDocumento18 pagineTianjin Jieda Temperature Instruments Factory Bimetallic Thermometer SpecsbunkeringNessuna valutazione finora
- Julabo MP Controller 19Documento23 pagineJulabo MP Controller 19Migue ZabaletaNessuna valutazione finora
- MCQ Thermal PhyDocumento14 pagineMCQ Thermal PhyGajendraNessuna valutazione finora
- Overall Heat Transfer Coefficient Table Charts and EquationDocumento5 pagineOverall Heat Transfer Coefficient Table Charts and Equationmiguel_vera6592Nessuna valutazione finora
- Sequence of OperationsDocumento12 pagineSequence of Operationseyad100% (2)
- Refrigeration Service Manual Thermostat GuideDocumento83 pagineRefrigeration Service Manual Thermostat GuideIordan Adrian100% (2)
- Fluent-Intro 14.5 L02 BoundaryConditionsSolverSettings PDFDocumento61 pagineFluent-Intro 14.5 L02 BoundaryConditionsSolverSettings PDFAnonymous XzqXVMjNessuna valutazione finora
- Solid State and Submerged Fermentation and Their ApplicationsDocumento12 pagineSolid State and Submerged Fermentation and Their ApplicationsAbishekNessuna valutazione finora
- Specific Heat Capacities of Metals: Experiment 9Documento4 pagineSpecific Heat Capacities of Metals: Experiment 9Pinto PintoNessuna valutazione finora
- Arun BLS4Documento2 pagineArun BLS4Eric E' Sandoval ANessuna valutazione finora
- Split Air Conditioner Pre-TestDocumento3 pagineSplit Air Conditioner Pre-TestkablasNessuna valutazione finora
- MAE 334 - Temp MeasurementDocumento5 pagineMAE 334 - Temp MeasurementKhoa Ngo NhuNessuna valutazione finora
- Landau Theory of Phase TransitionDocumento13 pagineLandau Theory of Phase TransitionKya Baat HaiNessuna valutazione finora
- IPC-TM-650 Test Methods ManualDocumento4 pagineIPC-TM-650 Test Methods ManualSSmyLoginNessuna valutazione finora
- Design and Operation of Double Pipe Heat Exchanger: February 2017Documento27 pagineDesign and Operation of Double Pipe Heat Exchanger: February 2017tasosNessuna valutazione finora
- Medium voltage switchgear diagramDocumento1 paginaMedium voltage switchgear diagramnailulfalah17Nessuna valutazione finora