Documenti di Didattica
Documenti di Professioni
Documenti di Cultura
Ktt211 18 Electron Rules PDF
Caricato da
Ahmad MuslihinTitolo originale
Copyright
Formati disponibili
Condividi questo documento
Condividi o incorpora il documento
Hai trovato utile questo documento?
Questo contenuto è inappropriato?
Segnala questo documentoCopyright:
Formati disponibili
Ktt211 18 Electron Rules PDF
Caricato da
Ahmad MuslihinCopyright:
Formati disponibili
04-Ktt212-18elect-09.
pptx
5-Sep-10
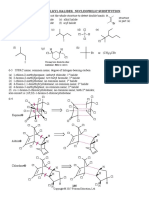
The 18 electron rule
For organic chemists its the octet rule, for organometallic chemists its the 18 electron rule. Useful for purposes of predicting reactivity. Each derives from a simple count of the number of electrons that may be accommodated by the available valence orbitals (one s and three p for organic chemists; organometallic chemists get five bonus dorbitals in which to place their electrons).
Slide 1
The 18 electron rule
states will follow the 18electron rule; metals in earlier rows and endrows do not. high field ligands will also observe the 18 electron rule.(later in the course) stability of complex.
Organometallic complexes with oxidation Complexes with backbonding electron or
18electron rule can assist in predicting the
Slide 2
p1
04-Ktt212-18elect-09.pptx
5-Sep-10
What is the 18 Electron Rule?
n + 2(CN) = 18
n = the number of valence d electrons on the metal CN is the coordination number
this 'rule' arise from the closed shell configuration d10s2p6 (Noble gas rule) analogous to octet rule for main group applies to most organometallic compounds
Slide 3
Cr(CO)6
Cr (0) a d6 central metal 6 CO with 2 e per CO
6 12
18
Cr(CO)7 [Cr(CO)6]
Cr (0) a d6 metal 7 CO with 2 e per CO Cr (0) a d6 metal 6 CO with 2 e per CO cas neg.
6 14 6 12 1
20
19
Slide 4
p2
04-Ktt212-18elect-09.pptx
5-Sep-10
CH3Re(CO)5 Re(I) CH3 donates 2 elect. 5 carbonyls (CO) Cp2Ru Ru(II) = 6 2 Cp with 6 elect. (2*(5 + 1)) = 12 Fe(0) Fe2(CO)9 3 terminal CO gives 2 elec. 3 bridging CO gives 1 elec. FeFe bond gives 1 elec. Cr(0) Cr(CO)6 6 Co gives 2 elec. each
6 2 10 6 12 8 6 3 1 6 12
18
18
18
18
Slide 5
Ru(II) = 6 2 Cp with 6 elect. (2*(5 + 1)) = 12
Fe OS = 0; No. of electrons = 8 3 bridging COs; 3*1 = 3
3 terminal COs; 3*2 = 6
Slide 6
p3
04-Ktt212-18elect-09.pptx
5-Sep-10
We can predict that less stable complexes are due to number of electrons; complexes can be stabilized by adding or removing electrons. Complexes that do not obey the rule will undergo reactions to provide a situation whereby the rule is observed: 1. V(CO)6 17elec. will oxidize [V(CO)6] which is an 18electron system. 2.[Fe(C5H5)2]+ with 17elec. will be reduced Fe(C5H5)2 which obeys the 18electron
Slide 7
* No of electrons 1st row
3 Sc Y (La)
4 Ti Zr Hf
5 V Nb Ta
6 Cr Mo W
7 Mn Tc Re
8 Fe Ru Os
9 Co Rh Ir
10 Ni Pd Pt
11 Cu Ag Au
12 Zn Cd Hg
2nd row
3rd row
Oxidation State
No of electrons on the metal ions
O I II III IV
3 2 1 0
4 3 2 1 0
5 4 3 2 1
6 5 4 3 2
7 6 5 4 3
8 7 6 5 4
9 8 7 6 5
10 9 8 7 6
11 10 9 8 7
12 11 10 9 8
Slide 8
p4
04-Ktt212-18elect-09.pptx
5-Sep-10
Knowing how many valence electrons "belong to" a transition metal complex allows us to make predictions about the mechanisms of reactions and the possible modes of reactivity.
There are two distinct methods that are used to count electrons, (i) the neutral or covalent method and (ii) the effective atomic number (EAN) or ionic method. These are simply two different accounting systems that give us the same final answer.
Slide 9
Slide 10
p5
04-Ktt212-18elect-09.pptx
5-Sep-10
Method 1: The ionic (charged) model
The basic premise of this method is that (i) we remove all of the ligands from the metal and (ii) add the proper number of electrons to each ligand to bring it to a closed valence shell state. 1. E.g. we remove ammonia(NH3) from metal complex, NH3 has a completed octet and acts as a neutral molecule. 2. When it bonds to the metal center it does so through its lone pair (Lewis acidbase adduct). 3. We call ammonia a neutral twoelectron donor.
Slide 11
Condt Method 1: The ionic (charged) model
In contrast, if we remove a methyl group from the metal and complete its octet, then we formally have CH3. If we bond this methyl anion to the metal, the lone pair forms our metalcarbon bond and the methyl group acts as a twoelectron donor ligand. Notice that to keep charge neutrality we must oxidize the metal by one electron (i.e. assign a positive charge to the metal). This will reduce the delectron count of the metal center by one.
Slide 12
p6
04-Ktt212-18elect-09.pptx
5-Sep-10
Condt Method 2: The covalent (neutral) model
In this method, we remove all of the ligands from the metal, & we do whatever is necessary to make them neutral, e.g. ammonia, NH3
When removed from the metal, it is a neutral molecule with one lone pair of electrons.
NH3 is a neutral two electron donor as with the ionic model.
Slide 13
We diverge from the ionic model when we consider a ligand such as methyl.
When we remove it from the metal and make the methyl fragment neutral, we have a neutral methyl radical. Both the metal and the methyl radical must donate one electron each to form our metalligand bond. Therefore, the methyl group is a one electron donor, not a two electron donor as it is under the ionic formalism. Where did the other electron "go"? It remains on the metal and is counted there.
In the covalent method, metals retain their full complement of d electrons because we never change the oxidation state from zero; i.e. Fe will always count for 8 electrons
regardless of the oxidation state and Ti will always count for four.
Slide 14
p7
04-Ktt212-18elect-09.pptx
5-Sep-10
Notice that this method does not give us any immediate information about the formal oxidation state of the metal, so we must go back and assign that in a separate step. For this reason, many chemists (particularly those that work with high oxidation state complexes) prefer the ionic method.
Slide 15
(a) 10 e (Pt) + 4 e (4 Cl) + 2 e (charge 2) = 16 e (b) 8 e (Pt2+) + 8 e (4 Cl) = 16 e
[PtCl4]2:
[Mn(CO)5]:
(a) 7 e (Mn) + 10 e (5CO) + 1 e(charge 1) = 18 e (b) 8 e (Mn) + 10 e (5 CO) = 18 e
Slide 16
p8
04-Ktt212-18elect-09.pptx
5-Sep-10
[Mn2(CO)10] : 14 e (2 Mn) + 20 e (10 CO) + 2 e (Mn Mn) = 36 e (i.e., 18 e per Mn)
[Co2(CO)8]: 18 e (2 Co) + 16 e (8 CO) + 2 e (CoCo) = 36 e (i.e., 18 e per Co)
OC CO OC CO
OC
OC
Mn
Mn
CO
O C Co C O Co
CO
OC CO OC CO
OC
CO CO
Slide 17
OC
Electron counting for [Rh2Cl2(CO)4]:
(a) 18 e (2 Rh) + 8 e (4 CO) + 6 e (2 bridging Cl) = 32 e (i.e., 16 e per Rh) (b) 16 e (2 Rh+) + 8 e (4 CO) + 8 e (2 bridgingCl) = 32 e (i.e., 16 e per Rh)
Slide 18
p9
04-Ktt212-18elect-09.pptx
5-Sep-10
(C5H5)Fe(CO)2Cl Fe(II) 6 e (C5H5) 6 e 2(CO) 2 x 2 = 4 e Cl 2 e Total 18 e : Electron Counting: Donor Pairs
Fe Cl
CO CO
Slide 19
[Fe(CO)4]2 Fe(2) d10 thus 10 +2(4) = 18 Fe2(CO)9 Fe(0) d8 thus: 2 metals 2(8) + CO ligands 9(2) MM bond 2 Total = 36 can view bridging CO; as 3 center 2 electron bond
OC OC OC Fe
O C C Fe O C O
CO CO CO
Slide 20
p10
04-Ktt212-18elect-09.pptx
5-Sep-10
Slide 21
Ligands
H-, [alkyl]- e.g. ([CH3]-, [C2H5]- dsb.), [aryl]- e.g. ([C6H5]-, [C6H5CH2]-, etc., CO, CS, CSe, PR3, P(OP)3, SbR3, F-, Cl-, Br-, I-, carbene(=CR2), alkene NO, NS, carbienes(CR) Dienes, [-allyl]-, [C5H5]-, [C4H4]2-, [C7H7]+ arene(C6H6, C6H5CO2CH3, etc.) [C3H3]3teraenes(C8H8) [C8H8]22
No. of electrons donated
3 4 6 8 10
Slide 22
p11
04-Ktt212-18elect-09.pptx
5-Sep-10
Slide 23
OC OC OC Fe OC
O C Fe
CO CO CO H
Fe = 8 e3 terminal CO = 6 eFe-Fe = 1 e1 H = 1 e2 CO = 2eTotal = 18 e-
Ph3 P Ni Ni P Ph3
Ni = 10 e5 -C5H5 = 5 e2-PPh3 = 3 Total = 18 eSlide 24
p12
04-Ktt212-18elect-09.pptx
5-Sep-10
Slide 25
Cr OC OC
Cr = 6 e6 -C6H6 = 6 e3 CO = 3*2 = 6 eTotal = 18 e-
OC Rh OC
Cl Rh Cl
CO
CO
CO
Rh = 9 e-Cl (1 e- Rh# 2 e- Rh##) = 3 e2CO = 4 eTotal = 16 e-
Slide 26
p13
04-Ktt212-18elect-09.pptx
5-Sep-10
OC OC OC Fe OC
O C Fe
CO CO CO H
Fe = 8 e3 terminal CO = 6 eFe-Fe = 1 e1 H = 1 e2 CO = 2eTotal = 18 eSlide 27
p14
Potrebbero piacerti anche
- Catalytic Oxidation of VOCs - ManuelaDocumento31 pagineCatalytic Oxidation of VOCs - ManuelaSantiago Sánchez GómezNessuna valutazione finora
- Group 1 - ETHYLBENZENE PRODUCTIONDocumento7 pagineGroup 1 - ETHYLBENZENE PRODUCTIONQuỳnh Như PhạmNessuna valutazione finora
- Asignment - Chapter 2Documento9 pagineAsignment - Chapter 2Nguyễn Đạt100% (1)
- PS1 08-SolutionDocumento7 paginePS1 08-SolutionNguyễn Vũ Quang Thành100% (1)
- Real InDictionary of Chemistry 4 BookDocumento33 pagineReal InDictionary of Chemistry 4 Booktire farrokhzadNessuna valutazione finora
- Giao Trinh Hoa Vo Co - Hay - NhatDocumento194 pagineGiao Trinh Hoa Vo Co - Hay - NhatNguyễnn Linhh ChiiNessuna valutazione finora
- Electro Chemistry FinalDocumento51 pagineElectro Chemistry FinalManoj50% (2)
- ASTM Plastic TestingDocumento2 pagineASTM Plastic TestingAnish KumarNessuna valutazione finora
- Experiment 1Documento11 pagineExperiment 1Kelvin LimNessuna valutazione finora
- Nguyễn Thị Hương Giang Unit 1: Chemistry And Its BranchesDocumento3 pagineNguyễn Thị Hương Giang Unit 1: Chemistry And Its BranchesGiang Nguyen Thi HuongNessuna valutazione finora
- German Problems 2014 Thu Nghiem 1Documento447 pagineGerman Problems 2014 Thu Nghiem 1Hảo ĐặngNessuna valutazione finora
- BT tuần 2Documento2 pagineBT tuần 2Đặng NhungNessuna valutazione finora
- Temperature Programmed Desorption TPDDocumento18 pagineTemperature Programmed Desorption TPDyiyiNessuna valutazione finora
- Bài tập Hoá lý 1 (Physical Chemistry 1 - Homework)Documento13 pagineBài tập Hoá lý 1 (Physical Chemistry 1 - Homework)Minh ThưNessuna valutazione finora
- 2016 Chimie Internationala Proba Teoretica SubiectebaremeDocumento51 pagine2016 Chimie Internationala Proba Teoretica SubiectebaremeCristinaNessuna valutazione finora
- Estimation of PKaDocumento3 pagineEstimation of PKaLiliana Andrea Pacheco Miranda100% (1)
- Final Exam - 2017 PDFDocumento7 pagineFinal Exam - 2017 PDFOlla 8352Nessuna valutazione finora
- Aromatic Nitro Compounds PDFDocumento20 pagineAromatic Nitro Compounds PDFRitam GhoshNessuna valutazione finora
- Easy Way To Determine R, S ConfigurationDocumento11 pagineEasy Way To Determine R, S ConfigurationHimNessuna valutazione finora
- Walkley Blackmethod PDFDocumento9 pagineWalkley Blackmethod PDFgagileNessuna valutazione finora
- Organic Chemistry: CarbanionDocumento12 pagineOrganic Chemistry: CarbanionDiana DedoNessuna valutazione finora
- Silver BromideDocumento9 pagineSilver BromideEric MonsalveNessuna valutazione finora
- Homework 1 FMDocumento3 pagineHomework 1 FMCynthia Guerra0% (1)
- HW 1 2018Documento4 pagineHW 1 2018Huy TranNessuna valutazione finora
- 39th IChO SyllabusDocumento12 pagine39th IChO SyllabuskyzzzNessuna valutazione finora
- (123doc) - Tieu-Luan-Phan-Ung-Cong-AeDocumento60 pagine(123doc) - Tieu-Luan-Phan-Ung-Cong-Ae58.Nguyễn Thị Diệu LinhNessuna valutazione finora
- Chapter 19. Aldehydes and Ketones: Nucleophilic Addition ReactionsDocumento98 pagineChapter 19. Aldehydes and Ketones: Nucleophilic Addition Reactions張湧浩Nessuna valutazione finora
- Donnee Exo SupplDocumento5 pagineDonnee Exo SupplMohamud LhafNessuna valutazione finora
- Plastic StandardsDocumento10 paginePlastic StandardsMarian BelmisNessuna valutazione finora
- Taber D.F., Lambert T. - Organic Synthesis - State of The Art 2013-2015 (2017)Documento286 pagineTaber D.F., Lambert T. - Organic Synthesis - State of The Art 2013-2015 (2017)Hi HiNessuna valutazione finora
- HW 5Documento2 pagineHW 5msoccerdude291Nessuna valutazione finora
- Electron Transfer Reactions of Complex Ions in SolutionDa EverandElectron Transfer Reactions of Complex Ions in SolutionNessuna valutazione finora
- Ethylene Oxidation On Silver Catalysts eDocumento22 pagineEthylene Oxidation On Silver Catalysts ePaola GarciaNessuna valutazione finora
- Chem 41c Quiz 1Documento6 pagineChem 41c Quiz 1Minh TieuNessuna valutazione finora
- BÁO CÁO THÍ NGHIỆM HÓA LYDocumento23 pagineBÁO CÁO THÍ NGHIỆM HÓA LYNhat Quang PhanNessuna valutazione finora
- Ebook Phân tích Lí - Hóa - GS.TS. Hồ Viết Quý - 655370 PDFDocumento571 pagineEbook Phân tích Lí - Hóa - GS.TS. Hồ Viết Quý - 655370 PDFTú QuyênNessuna valutazione finora
- 1-50 QuestionsDocumento48 pagine1-50 Questionsbolla reddyNessuna valutazione finora
- Enu Tour1 TaskDocumento9 pagineEnu Tour1 TaskĐinh Đại VũNessuna valutazione finora
- PhotoChemistry PDFDocumento22 paginePhotoChemistry PDFAnkitranjan GogoiNessuna valutazione finora
- Problem Set No. 3Documento1 paginaProblem Set No. 3CJ GoradaNessuna valutazione finora
- Determining Molar Mass Using CryosDocumento6 pagineDetermining Molar Mass Using CryosValentin-AngeloUzunovNessuna valutazione finora
- Chapter 6-Alkyl Halides Nucleophilic Substitution: You May Have Drawn The Other Enantiomer. Either Is CorrectDocumento23 pagineChapter 6-Alkyl Halides Nucleophilic Substitution: You May Have Drawn The Other Enantiomer. Either Is Correct張湧浩Nessuna valutazione finora
- A General Review of Thermodynamic Concepts: KMU220 Chemical Engineering Thermodynamics IDocumento24 pagineA General Review of Thermodynamic Concepts: KMU220 Chemical Engineering Thermodynamics ISaud Hashmi100% (1)
- Bài Tập Chuỗi Chuyển Hóa Hữu Cơ 2Documento10 pagineBài Tập Chuỗi Chuyển Hóa Hữu Cơ 2A4K74 HUP100% (1)
- Chapter18 Answer KeyDocumento6 pagineChapter18 Answer KeyRavindra KempaiahNessuna valutazione finora
- Chapter 11Documento24 pagineChapter 11Biotechnology IIUM Kuantan100% (2)
- Structure of Ceramics PDFDocumento14 pagineStructure of Ceramics PDFTejinder SinghNessuna valutazione finora
- PhotochemistryDocumento24 paginePhotochemistryVijay PradhanNessuna valutazione finora
- 4.5 Exercise 1 Aldehydes and KetonesDocumento1 pagina4.5 Exercise 1 Aldehydes and Ketonesloly62006Nessuna valutazione finora
- Impact StrengthDocumento13 pagineImpact StrengthManishUpadhyay100% (1)
- Anti Baldwin CyclizationsDocumento14 pagineAnti Baldwin CyclizationsLeandro SasiambarrenaNessuna valutazione finora
- Cahn-Ingold-Prelog Priority RulesDocumento5 pagineCahn-Ingold-Prelog Priority RulesBer GuzNessuna valutazione finora
- HSAB TheoryDocumento11 pagineHSAB TheoryMahesh Babu SriNessuna valutazione finora
- Brochure Transcutol P For Efficient Skin PenetrationDocumento24 pagineBrochure Transcutol P For Efficient Skin PenetrationJoaozinhoMéndez100% (1)
- 2013 Group II Lecture Notes (Student) .UnlockedDocumento20 pagine2013 Group II Lecture Notes (Student) .UnlockedKwok Yee TherNessuna valutazione finora
- Thao Nguyen ReportDocumento12 pagineThao Nguyen ReportHoàng Nguyễn100% (1)
- Electroanalysis: Theory and Applications in Aqueous and Non-Aqueous Media and in Automated Chemical ControlDa EverandElectroanalysis: Theory and Applications in Aqueous and Non-Aqueous Media and in Automated Chemical ControlNessuna valutazione finora
- Enzymes: A Practical Introduction to Structure, Mechanism, and Data AnalysisDa EverandEnzymes: A Practical Introduction to Structure, Mechanism, and Data AnalysisValutazione: 4 su 5 stelle4/5 (2)
- Full Scale AnalysisDocumento43 pagineFull Scale Analysispaulogud6170Nessuna valutazione finora
- B2 Stability of Structures 2nded Dover2003Documento9 pagineB2 Stability of Structures 2nded Dover2003nicu1212Nessuna valutazione finora
- Vectors in Two Dimensions: What Is A Vector?Documento13 pagineVectors in Two Dimensions: What Is A Vector?Lazarus MukwekweNessuna valutazione finora
- Es TG Module 2 Climate RevisedDocumento23 pagineEs TG Module 2 Climate RevisedNikkieIrisAlbañoNovesNessuna valutazione finora
- ChemDraw TrainingDocumento64 pagineChemDraw Trainingslash_de_law7484Nessuna valutazione finora
- Chemistry 1A Lecture 3 and 4 Revised 2021 2022Documento51 pagineChemistry 1A Lecture 3 and 4 Revised 2021 2022edwinjugunaNessuna valutazione finora
- INOVA Gear Flyer A4 EnglischDocumento2 pagineINOVA Gear Flyer A4 EnglischBhagesh LokhandeNessuna valutazione finora
- Assignment in Power SystemDocumento5 pagineAssignment in Power SystemRhiza NavallascaNessuna valutazione finora
- Jack Sarfatti - Hologram UniverseDocumento9 pagineJack Sarfatti - Hologram UniverseFrankOfTomorrowNessuna valutazione finora
- Searching For CometsDocumento2 pagineSearching For CometspianomagicianNessuna valutazione finora
- VSP Final ReportDocumento53 pagineVSP Final ReportAnand GautamNessuna valutazione finora
- Synthesis and Characterization of Pegylated Polypropyleneimine (Ppi) Dendrimer Loaded Prednisolone For Antileukemic ActivityDocumento11 pagineSynthesis and Characterization of Pegylated Polypropyleneimine (Ppi) Dendrimer Loaded Prednisolone For Antileukemic ActivityRAPPORTS DE PHARMACIENessuna valutazione finora
- Differentiation Formulas Integration FormulasDocumento7 pagineDifferentiation Formulas Integration FormulasnirobNessuna valutazione finora
- Ch6a, Drude Model and Conductivity of A MetalDocumento120 pagineCh6a, Drude Model and Conductivity of A Metalashraf habibNessuna valutazione finora
- MAGNETS PPT FinalDocumento14 pagineMAGNETS PPT FinalCHELCEE C. ENARIONessuna valutazione finora
- Science 9 2nd QuarterDocumento2 pagineScience 9 2nd QuarterJocelyn Acog Bisas Mestizo100% (1)
- Multiple Choice Questions in Electricity and Magnetism FundamentalsDocumento7 pagineMultiple Choice Questions in Electricity and Magnetism FundamentalsKen NyNessuna valutazione finora
- Mri Physics en Rev1.3Documento76 pagineMri Physics en Rev1.3Arbjan Rusi100% (3)
- 7 Hydraulic ConductivityDocumento31 pagine7 Hydraulic ConductivityDarya Memon100% (2)
- Статья - 1 фнт НабойченкоDocumento8 pagineСтатья - 1 фнт НабойченкоОльга НабойченкоNessuna valutazione finora
- Kakutani Fixed-Point TheoremDocumento9 pagineKakutani Fixed-Point TheoremNicolas MarionNessuna valutazione finora
- Abaqus 6.12: Abaqus Keywords Reference ManualDocumento1.545 pagineAbaqus 6.12: Abaqus Keywords Reference ManualIojanan Ben AvrahamNessuna valutazione finora
- 08 - Mook Test SP - 6Documento14 pagine08 - Mook Test SP - 6AnkurNessuna valutazione finora
- RheologyDocumento23 pagineRheologyJasmine LeeNessuna valutazione finora
- HC Docx1Documento13 pagineHC Docx1ayushsekhariNessuna valutazione finora
- 1525950185module-2 Unit5 COM-IDocumento8 pagine1525950185module-2 Unit5 COM-ISimranNessuna valutazione finora
- Sensors & Instrumentation Notes Unit-02Documento47 pagineSensors & Instrumentation Notes Unit-02shivangi sharmaNessuna valutazione finora
- Ch2QM PDFDocumento26 pagineCh2QM PDFfractalgeoNessuna valutazione finora
- Quantum Number and Electron Orbital Review GameDocumento3 pagineQuantum Number and Electron Orbital Review GameMonette CabugayanNessuna valutazione finora
- Laplace Transform: Aim: Mathematical BackgroundDocumento10 pagineLaplace Transform: Aim: Mathematical BackgroundSahil KalingNessuna valutazione finora