Documenti di Didattica
Documenti di Professioni
Documenti di Cultura
34 Alcohols & Ethers - Problems For Practice - Level 1
Caricato da
Abuturab MohammadiDescrizione originale:
Titolo originale
Copyright
Formati disponibili
Condividi questo documento
Condividi o incorpora il documento
Hai trovato utile questo documento?
Questo contenuto è inappropriato?
Segnala questo documentoCopyright:
Formati disponibili
34 Alcohols & Ethers - Problems For Practice - Level 1
Caricato da
Abuturab MohammadiCopyright:
Formati disponibili
EXERCISE-I(A)
Q.1 Which of the following reaction is called as BouveaultBlanc reduction (A) Reduction of acyl halide with H2PdBaSO4 (B) Reduction of ester with Na/C2H5OH (C) Reduction of anhydride with LiAlH4 (D) Reduction of carbonyl compounds with Na/HgHCl Glycol on treatment with PI3 mainly gives (A) Ethylene (B) Ethylene iodide
Q.2
(C) Ethyl iodide
(D) Ethane
Q.3
Acrolein is formed when glycerol is heated with (A) Acidified KMnO4 (B) Br2 water (C) KHSO4 Glycerol on treatment with oxalic acid at 110C forms (A) Allyl alcohol (B) Formic acid (C) CO2 and CO
(D) HNO3 (D) Glyceric acid
Q.4
Q.5
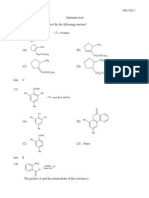
If the starting material is 1methyl1,2epoxy cyclopentane, of absolute configuration, decide which one compound correctly represent the product of its reaction with sodium methoxide in methanol.
(A)
(B)
(C)
(D)
Q.6
When phenol is treated with PCl5, the yield of chlorobenzene is generally poor because of the formation of (A) Benzoyl chloride (B) pchorophenol (C) ochlorophenol (D) Triphenyl phosphate In the following reaction, final product is
H ClCH 2 CH C H 2 NaOC 2 5 \ / O
Q.7
14
14
14
(A) ClCH 2CH C H 2OC 2 H 5 | OH (C) C H 2 CHCH 2 OC 2 H 5 \ / O
14
(B) ClCH 2CH C H 2ONa | OC 2 H 5 (D) CH 2 CH C H 2OC 2 H 5 \ / O
14
Q.8
Consider the reaction of HI with the following: I II (D) none
Which forms di-iodide on reaction with HI (excess)? (A) I and II both (B) II only (C) I only Q.9
Ethanol on reaction with acetic anhydride gives (A) Acetic ester (B) Formic ester (C) Ethanoic acid (D) Acetic ester and Ethanoic acid both Ethanol cannot be dried by anhydrous CaCl2 due to formation of the following solvated product (A) CaCl22C2H5OH (B) 2CaCl23C2H5OH (C) CaCl23C2H5OH (D) CaCl2C2H5OH Rate of hydration of
Q.10 Q.11
, will be in order: (A) I < II < III Q.12 Q.13 Q.14 Q.15 Q.16
(B) I < III < II
(C) II < I < III
(D) III < II < I
The reaction of CH3OC2H5 with HI gives (A) CH3I (B) C2H5OH
(C) CH3I + C2H5OH (D) C2H5I + CH3OH
The number of methoxy groups in a compound can be determined by treating it with (A) HI and AgNO3 (B) Sodium carbonate (C) Sodium hydroxide (D) Acetic acid Most acidic alcohol out of following compounds is (A) (CH3)3COH (B) CH3CH2OH (C) CH3OH Action of HNO2 on CH3NH2 gives following as major product (A) CH3OH (B) CH3OCH3 (C) CH3ON=O (D) PhOH (D) CH3NO2
A compound X with molecular formula C3H8O can be oxidised to a compound Y with the molecular formula C3H6O2, X is most likely to be (A) Primary alcohol (B) Secondary alcohol (C) Aldehyde (D) Ketone Diethyl ether and air gives ether hydroperoxide. The mechanism of the reaction is (A) Nucleophilic substitution (B) Free radical addition (C) Free radical substitution (D) None of the above Ether on carbonylation gives (A) Alkanoic acid (B) Alkanone
Q.17
Q.18
(C) Alkyl alkanoate
(D) Alkanal
Q.19
I R O R ' RI + R'OH true about this mechanism | H
(A) SN1 in gas phase (C) both of the above Q.20 Phenol with Hinsbergs reagent gives (A) Sulphone (B) Sulphanilic acid
(B) SN2 in aqueous phase (D) none (C) Sulphonic ester (D) Sulphonal
Q.21
Select the odd structure out (A) CH3CH(OH)CH2CH3 (C) CH3CH(OH)CH3
4 Glycerol 4 A B A and B are: (A) Acrolein, allyl alcohol (C) Allyl alcohol, acrolein
(B) CH3CH(OH)CH2CH2CH3 (D) CH3CH2CH(OH)C2H5
Q.22
KHSO
LiAlH
(B) Glyceryl sulphate, acrylic acid (D) Only acrolein (B is not formed)
Q.23
An ether is heated with phosphorous penta sulphide to give (A) Alkanthiol (B) Dialkyl sulphide (C) Hydrogen sulphide (D) Thioester
O i ) NaOH Phenol ( A 2 B Al2 C 3 H+ / H O
Q.24
( ii ) CO 2 / 140C
CH3COOH ,
In this reaction, the end product C is: (A) salicylaldehyde (B) salicylic acid Q.25
(C) phenyl acetate
(D) aspirin
In the Liebermanns nitroso reaction changes in the colour of phenol occurs as: (A) Brown or red-greenish red-deep blue (B) red-deep blue-green (C) red-green-white (D) white-red-green
Q.26
A. A is
H 3O
(A)
(B)
(C)
(D)
Q.27 A and B are: (A)
cold A CrO3 B alkaline KMnO
4
AcOH
(B)
(C)
(D) no formation of A and B
Q.28
B 4
NaBH
2 CH = CH CHO A
H / Pt
A and B are: (A) (B) (C) (D) CH2CH2CHO, CH2CH2CH2OH, CH = CHCH2OH CH = CH CH2OH
CH= CHCH2OH in both cases CH2CH2CH2OH in both cases
Q.29
3 B
CH OH
2 A
H O18 H+
CH 3ONa
A and B are CH 3 | (A) CH 3 C CH 2 | | OH OH 18
CH 3 | , CH 3 C CH 2 | | OH OCH 3
CH 3 CH 3 | | (B) CH 3 C CH 2 , CH 3 C CH 2 | | | | OH18OH OH OCH 3
CH 3 CH 3 | | (D) CH 3 C CH 2 , CH C CH 3 2 | | | | 18 OCH 3 OH 18OH OH
CH 3 CH 3 | | (C) CH 3 C CH 2 , CH 3 C CH 2 | | | | 18 OH OH OH OH 18
18
Q.30
Oxalic acid + A hence A conc. H 2SO 4 B, B is: (C) CH 2 O CH 2 | | OH OH
(A)
(B)
(D) None
Q.31
Select schemes A, B, C out of I acid catalysed hydration (A) I in all cases (B) I, II, III Q.32 Dehydration of the alcohols
II
HBO (C) II, III, I
III
oxymercuration-demercuration (D) III, I, II
will be in order (A) IV > III > II > I
(B) I > II > III > IV
H O+
(C) IV > II > III > I
(D) II > IV > I > III
Q.33
CH3MgBr + E is (A)
HCHO HBr Mg / ether 3 HI A B C D E H O+
3
(B)
(C)
(D)
Q.34
H ? Product is:
(A)
(B)
(C)
(D)
Q.35 (A) (C)
Alc. Major product is KOH (B) (D)
Q.36
RMgX ? Product Obtained is:
H 2O
R | (A) R 'C CH 2OH | R''
R' R R | | | (B) RCH 2 C OH (C) R ' CH 2 C OH (D) R ' ' CH 2 C OH | | | R' ' R' R''
Q.37
I / NaOH
+ A, A is H
(A)
(B)
(C)
(D)
Q.38
3 (A)
CH OH CH3O
3 (B) A & B are:
H 2SO 4
CH OH
(A)
&
(B)
&
(C)
&
(D)
&
Q.39
3 ? Major Product is:
AlCl
(A)
(B) H3COC
OH
(C)
(D)
Q.40
? Product is:
(A)
(B)
(C)
(D) no reaction
Q.41
3 [X] here X is:
( i ) CHCl + KOH
(ii ) CH 2I 2 + NaOH
(A)
(B)
(C)
(D)
Q.42
(CH 3 ) 2 C C(CH 3 ) 2 | | OH OH (A) OH at C2 is more basic than that of at C5 (B) OH at C2 is more acidic than at C5. (C) both have same basicity (D) both have same acidic strength . In this diol
2 (CH3)2C = C(CH3)2 A OH B
Q.43
H 2O
(A)
(B) (CH 3 ) 2 C C(CH 3 ) 2 | | OH Cl (D) None
(C)
Q.44 A is
2 A
NaNO HCl
(A)
(B)
(C)
(D)
Q.45
CH 2 = CHCHCH 2CH 2OH | OH (A) CH 2 = CHCCH 2 CH 2OH || O
A. A is
(B)
(C)
(D)
Q.46
HCO 3H H3O Acetophenone A B + C Indicator (D) +
Pthalic Anhydride H
C & D are
(A) CH3OH &
(B) PhOH &
(C) PhOH &
(D) CH3OH &
MnO CH 2 CHCHCH 2CHO CHO =2 CHCCH 2O || || CH 2 = CHCCH 2COH OH O || O
Q.47
m-Aminophenol on treatment with NaOH and CO2 gives which of the following as major product?
(A)
(B)
(C)
(D)
Question No 48 to 50 (3 questions)
Q.48
Compound 'B' is? (A) (B) (C) (D)
Q.49
Organic compound A does not undergo de carboxylation reaction because? (A) Intermediate does not follow Saytzeff'n rule (B) Intermediate does not follow Hofmann's rule (C) Intermediate does not follow Bredict's rule (D) Intermediate does not follow MarkwoniKoffs rule Total No. of stereoisomers of B are? (A) 2 (B) 4 (C) 3 (D) 6
Q.50
EXERCISE-I(B)
Q.1 Compound which gives alcohol on reduction is/are (A) Me C Cl || O Q.2 (B) (C) (D)
Phenol and ethanol are distinguished by the reaction with (A) Red litmus (B) NaHCO3 (C) FeCl3
H
(D) NaOH
Q.3
dil. H 2SO 4 2 2O In the reaction sequence, CaC2 H A B C, true about the product C is +2 Hg Ni
(A) give yellow ppt. with NaOI (B) its final oxidation product is carbonyl compound (C) its final oxidation product is CO2 and H2O (D) its final oxidation product is CH3COOH Q.4 Which can be cleaved by HIO4? O O || || (A) CH CH CCH CCH 3 2 2 3 OH O O | O Me C CH 2 CH NH |2| | |||\ /| | (B) CH 3CHCCH CH Me O O Me2 3 C OC O O || || (D) CH 3CCH 2 CHCH 2CCH 3 | OH
(C)
Q.5
HBO, oxymercuration-demercuration and acid catalysed hydration will not give same product in (A) (B) (C) (D)
Q.6
Diethyl ether reacts with PCl5 to form (A) Ethyl chloride (C) Ethenol
(B) Phosphorous oxy trichloride (D) Ethene
Q.7
Select the correct synthesis products (A)
2 2 3 NaOH
BH THF
H O , OH
(B)
2 4 NaOH
Hg ( OAc) H 2O
NaBH
(C) (D) Q.8
mCPBA
CH 2Cl2
The molecules of ether dehydrates in the presence of (A) Al2O3 (B) H3PO4 (C) H2S2O7 Anhydride of alcohol is (A) Ether (C) Alkyl hydrogen sulphere (B) Aldehyde (D) Alkene
(D) liq. NH3
Q.9
Q.10
Lucas test is used to make distinction between 1, 2 and 3 alcohols ROH + HCl anhydrous ZnCl 2
conc.
RCl
White turbidity
+ H2O
This shows that (A) ROH behaves as a base (B) greater the value of pKa (alcohol), greater the reactivity with conc. HCl and thus sooner the formation of white turbidity. (C) alcohol which reacts fastest with Na metal, will give turbidity at fastest rate (D) alcohol which gives red colour during Victor Mayor test, will give turbidity at slower rate then those giving blue or white colour during Victor Mayor test. Q.11 If ethanol dissolves in water, then which of the following would be done (A) Absorption of heat (B) Emission of heat (C) Increase in volume (D) Contraction in volume Which method is useful for the synthesis of ether? (A) (B) C2H5ONa + (CH3)2SO4 (C) CH3ONa + CH3CH2OSO2 (D) (CH3)3CBr + CH3CH2ONa Q.13 Which of the following can react with TsCl (A) Glycerol (C) Oil of wintergreen (B) Oximinoacetone (D) dimethyl amine aq. NaOH
Q.12
Q.14
Which is/are correct statements? (A)
(B)
(C) This is only affected in reduction to 2 alcohol
NaBH
CH3OH
(D)
Q.15
3-methyl-3-hexanol can be prepared by (A) CH3MgI and 3-hexanone, followed by hydrolysis (B) C2H5MgI and 2-pentanone, followed by hydrolysis (C) C3H7MgI and 2-butananone, followed by hydrolysis (D) C4H9MgI and propanone, followed by hydrolysis In which cases product formed are not according to reaction? (A) (B) + HNO3 H 2SO 4 + HNO3 H 2SO 4
Q.16
(C) CH2 = CHCHO + LiAlH4 CH3 CH2CH2OH (D) Q.17 + CH3ONa
Dehydration of alcohols take place more rapidly with POCl3 than with H2SO4. Select the correct statement(s) about the following dehydration reaction.
3
POCl
pyridine
(A) It does not involve carbocation. (B) It involves ROPOCl2 with OPOCl2 as a better leaving group. (C) It involves E2 mechanism as pyridine base abstracts proton from the adjacent carbon as the same time at which OPOCl2 is leaving. (D) It is E1 reaction without formation of carbocation.
Q.18
Which of the following will get oxidised by Br2 / KOH into carboxylic acid? (A) CH3CH2OH (B)
CH CH 3 | OH
(C) Q.19
CH2OH
(D)
In Kolbe-Schmidt reaction, o-hydroxy benzoic acid is predominantly formed. This is because (A) salicylate anion is a stronger base than phenoxide ion (B) salicylate anion is a weaker base than phenoxide ion (C) p-hydroxy benzoate ion is a stronger base than phenoxide ion (D) p-hydroxy benzoate ion is a stronger base than salicylate ion Methanol can be distinguished from ethanol by (A) Heating with I2 and alkali (B) Treating with schiffs reagent (C) Treating with CrO3 solution in dil. H2SO4 (D) Treating with Lucas reagent Products form by following reactions are
Q.20
Q.21
CHCl +KOH
(A)
(B)
(C)
(D)
Q.22
For the reactions shown below, identify the correct statement(s) with regard to the products formed: (i) MeOH, P H
+
(ii)
NaOMe Q / MeOH
(A) P and Q are identical (B) P is recemic and Q is optically active (C) P and Q are positional isomers (D) both are optically active Q.23
Tilden NaNO2 NH C2N5NH2 (i) 3 (ii) (iii). The product (iii) can be reagent
HCl
(A) Alcohol
(B) Ether
(C) Alkyl chloride
(D) Alkyl nitrite
Q.24
Products form during dehydration of following alcohols are
(A) Q.25
(B)
(C)
(D)
Consider the following compound A (below)
Select the correct statement(s) (A) It is more acidic than CH3OH (C) It reacts very fast with Lucas reagent Q.26 Match the following: Reaction (A)
2 4
O || (B) It is more acidic than CH 3COH (D) It is a diacid base Mechanism
Conc.H SO
(P)
change in number of carbon in cycle
(B)
HIO
(Q)
Pinacol rearrangement
(C)
H
heat H O heat
(R)
Oxidative bond cleavage
(D) Q.27
(S)
Ketone as product
Match the column: Column I (A) Identification of 1, 2& 3 Alcohols (B) Identification of 1, 2& 3 Nitro alkanes (C) Formation of alcohol by anti Markovnikov's addition of H2O (D) Formation of alcohol by Markovnikov's addition
(P) (Q) (R) (S) (T)
Column II Oxymercuration demercuration Cu/300 heat Victor Mayer's test Hydroboration oxidation Lucas test
Q.28
Match the column: Column I (A) Oxidation of 1alcohol in aldehyde (B) CrO3 . .HCl
(P) (Q)
Column II KMnO4/ Collin's reagent
(C) (D) Q.29
CrO3 .2 Oxidation of alkene into acid
(R) (S)
Jone's reagent P.C.C
Match the products of following: Column I (A) (B)
HI
Column II (P) (Q) I CH2OH
Violet color is obtain by the reaction of neutral FeCl3 with
(C)
Reaction of benzaldehyde with LiAlH4 / H2O gives
(R)
OH
(D)
+ KI ?
Q.30
CH 3 CH CH = CH 2 | CH 3
which is true about alcohol and R? Alcohol (A)
(S) CH2 I CH 3 agent R CH CH 3 CH Re OH | | CH 3 CH CH 2CH 3 C OH 3 Alcohol | CH 3
Reagent B2H6 , H2O2 / NaOH
CH 3 CH CH 2 CH 2OH | CH 3
(B)
PdCl2, H2O, O2 / LAH
(C)
OH | CH 3 C CH 2CH 3 | CH 3
Hg(OAc)2, H2O / NaBH4
(D)
dil. H2SO4
Potrebbero piacerti anche
- Alcohols & EthersDocumento36 pagineAlcohols & EthersKaveesh KulkarniNessuna valutazione finora
- Iit Questions On Carbonyl Compounds & Carboxylic Acid and Its DerivativeDocumento12 pagineIit Questions On Carbonyl Compounds & Carboxylic Acid and Its DerivativeRaju SinghNessuna valutazione finora
- Alcohol EtherDocumento36 pagineAlcohol EtherAnand MurugananthamNessuna valutazione finora
- HydrocarbonDocumento39 pagineHydrocarbonSachin KumarNessuna valutazione finora
- Alcohol & Ether B Lord of The RingsDocumento13 pagineAlcohol & Ether B Lord of The RingsadityaNessuna valutazione finora
- Jee 2014 Booklet7 HWT Oxygen Containing Organic Compounds IIDocumento10 pagineJee 2014 Booklet7 HWT Oxygen Containing Organic Compounds IIvarunkohliinNessuna valutazione finora
- Class Test-1-Aldehydes & Ketones - PreparationDocumento5 pagineClass Test-1-Aldehydes & Ketones - PreparationSarthak VermaNessuna valutazione finora
- Aldehydes & KetonesDocumento23 pagineAldehydes & KetonesManthan JhaNessuna valutazione finora
- HC Docx1Documento13 pagineHC Docx1ayushsekhariNessuna valutazione finora
- Aromatic CompoundsDocumento16 pagineAromatic CompoundsadityaNessuna valutazione finora
- Halogen Containing CompoundsDocumento67 pagineHalogen Containing Compoundsonline_mktg100% (6)
- 3 - Aldehydes and Ketones (Assignment) Booklet-2Documento15 pagine3 - Aldehydes and Ketones (Assignment) Booklet-2kraken monsterNessuna valutazione finora
- Carbonyl Compounds SheetDocumento133 pagineCarbonyl Compounds Sheetadityavaish739Nessuna valutazione finora
- Aromatic Compounds (13th)Documento24 pagineAromatic Compounds (13th)Raju SinghNessuna valutazione finora
- CadDocumento8 pagineCadRamesh Babu GarlapatiNessuna valutazione finora
- 12TH CBSE DPP 37. Aldehydes, Ketones and Carboxylic Acids MCQ ASSERTION REASON CS QDocumento20 pagine12TH CBSE DPP 37. Aldehydes, Ketones and Carboxylic Acids MCQ ASSERTION REASON CS Q123Nessuna valutazione finora
- BT Alcolhols Phenls and Ethers - ExerciseDocumento20 pagineBT Alcolhols Phenls and Ethers - ExerciseSanjay KumarNessuna valutazione finora
- Corbonyl CompOUND AND Corboxilic AcidDocumento12 pagineCorbonyl CompOUND AND Corboxilic AcidApex InstituteNessuna valutazione finora
- Carbonyl Compounds 12thDocumento24 pagineCarbonyl Compounds 12thRaju SinghNessuna valutazione finora
- Alcohols & EtherDocumento18 pagineAlcohols & EtherRaju SinghNessuna valutazione finora
- 12 Carboxyloc AcidDocumento3 pagine12 Carboxyloc Acidimu4u_786Nessuna valutazione finora
- Organic Chemistry-JeeDocumento33 pagineOrganic Chemistry-JeeRamesh Babu GarlapatiNessuna valutazione finora
- Hydrocarbon 4Documento35 pagineHydrocarbon 4AjayNessuna valutazione finora
- NSEC Solved Paper 2010Documento7 pagineNSEC Solved Paper 2010whatismyusername1947Nessuna valutazione finora
- Carboxylic Acids and Acid Derivatives Carbonyl PDFDocumento124 pagineCarboxylic Acids and Acid Derivatives Carbonyl PDFrvignesh2809Nessuna valutazione finora
- Aldehydes, Ketones and Carboxylic AcidsDocumento7 pagineAldehydes, Ketones and Carboxylic Acidskavitha2511977Nessuna valutazione finora
- Exercise-01 Check Your Grasp: O CH HO HODocumento29 pagineExercise-01 Check Your Grasp: O CH HO HOHet PrajapatiNessuna valutazione finora
- P Block Elements QBDocumento12 pagineP Block Elements QBRajeev KaushikNessuna valutazione finora
- Alcohol & EtherDocumento217 pagineAlcohol & EtherAmitNessuna valutazione finora
- Exercise-01 Check Your Grasp: O CH HO HODocumento7 pagineExercise-01 Check Your Grasp: O CH HO HOChesta MalhotraNessuna valutazione finora
- National Standard Examination in Chemistry 2014: QP Code C 203Documento15 pagineNational Standard Examination in Chemistry 2014: QP Code C 203Karan TejwaniNessuna valutazione finora
- Carbonyl CompoundDocumento197 pagineCarbonyl CompoundAmitNessuna valutazione finora
- Halogen DerivativesOrganic Chem Class XIIDocumento32 pagineHalogen DerivativesOrganic Chem Class XIIDwaipayan Pradhan100% (2)
- Jee 2014 Booklet5 HWT HalidesDocumento12 pagineJee 2014 Booklet5 HWT Halidesvarunkohliin100% (2)
- Section-I (Single Correct Choice) : HC CH 1.1eq Nanh Nanh Nanh X XDocumento14 pagineSection-I (Single Correct Choice) : HC CH 1.1eq Nanh Nanh Nanh X XPriyansh YadavNessuna valutazione finora
- WORK BOOK - Exercise in ChemistryDocumento28 pagineWORK BOOK - Exercise in ChemistryTikeshwar SharmaNessuna valutazione finora
- Chemistry Jee MainDocumento15 pagineChemistry Jee MainAt TanwiNessuna valutazione finora
- (Xii) Haloalkanes and HaloarenesDocumento11 pagine(Xii) Haloalkanes and HaloarenessitaramroyalNessuna valutazione finora
- Quiz Organic 1Documento6 pagineQuiz Organic 1ronakgupta332005Nessuna valutazione finora
- Aldehydes and KetonesDocumento29 pagineAldehydes and KetonesJiya singhNessuna valutazione finora
- Halogen Derivatives PDFDocumento32 pagineHalogen Derivatives PDFRaju Singh100% (1)
- Notes Chapter 882Documento107 pagineNotes Chapter 882notime ReactionNessuna valutazione finora
- Goc & Eas Test-IiDocumento7 pagineGoc & Eas Test-IiAniket GuptaNessuna valutazione finora
- Hydrocarbons A 1Documento4 pagineHydrocarbons A 1REJA MUKIB KHANNessuna valutazione finora
- Alkanes Alkenes Alkynes ObjectiveDocumento7 pagineAlkanes Alkenes Alkynes ObjectiveVishal_93Nessuna valutazione finora
- Hydrocarbon TestDocumento7 pagineHydrocarbon TestRajeev GangwarNessuna valutazione finora
- (Xii) Alcohol, Phenol, EtherDocumento10 pagine(Xii) Alcohol, Phenol, EtherAbhishek SharmaNessuna valutazione finora
- Aldehyde and Ketone (DPP)Documento12 pagineAldehyde and Ketone (DPP)Shreeyansh Bhardwaj0% (1)
- CC 13Documento6 pagineCC 13Deepak SethyNessuna valutazione finora
- Unit 11 MCQDocumento7 pagineUnit 11 MCQJay VermaNessuna valutazione finora
- Alcohol, Phenol & Ether: A O CH C HDocumento6 pagineAlcohol, Phenol & Ether: A O CH C HJitendra KumarNessuna valutazione finora
- Hydrocar SHEET3Documento4 pagineHydrocar SHEET3Aayush SaxenaNessuna valutazione finora
- Answers To ROH Tutorial PDFDocumento12 pagineAnswers To ROH Tutorial PDFCorvo Haosen Al-Han0% (1)
- Resonance Chemistry DPP 6 (Advanced)Documento11 pagineResonance Chemistry DPP 6 (Advanced)Anurag1210701067% (6)
- JMS-5 Paper - 2Documento7 pagineJMS-5 Paper - 2janmanchiNessuna valutazione finora
- Practice Makes Perfect in Chemistry: Oxidation-ReductionDa EverandPractice Makes Perfect in Chemistry: Oxidation-ReductionValutazione: 5 su 5 stelle5/5 (1)
- Chirality in Supramolecular Assemblies: Causes and ConsequencesDa EverandChirality in Supramolecular Assemblies: Causes and ConsequencesF. Richard KeeneNessuna valutazione finora
- VLSI ALU VerilogDocumento19 pagineVLSI ALU VerilogAbuturab Mohammadi100% (1)
- Oc and SC Tests On A Single-Phase Transformer: Experiment No. 1Documento5 pagineOc and SC Tests On A Single-Phase Transformer: Experiment No. 1Abuturab MohammadiNessuna valutazione finora
- Mathworks Placement ProcessDocumento1 paginaMathworks Placement ProcessAbuturab Mohammadi100% (2)
- Electrical Machines Lab Manual - 2014-15 - Cycle I-11!08!2014Documento52 pagineElectrical Machines Lab Manual - 2014-15 - Cycle I-11!08!2014Abuturab MohammadiNessuna valutazione finora
- Tutorial 1Documento3 pagineTutorial 1Abuturab MohammadiNessuna valutazione finora
- 07 Spiral MatrixDocumento3 pagine07 Spiral MatrixAbuturab MohammadiNessuna valutazione finora
- Optics Iit MaterialDocumento27 pagineOptics Iit MaterialAbuturab MohammadiNessuna valutazione finora
- Class 11 Diwali Assignment Physics SolutionDocumento25 pagineClass 11 Diwali Assignment Physics SolutionHarshitShuklaNessuna valutazione finora
- ObjectiveDocumento15 pagineObjectiveIkhmal FirdausNessuna valutazione finora
- Ucl Chem 114aDocumento3 pagineUcl Chem 114aKelLYSNessuna valutazione finora
- Mini Polyatomic IonsDocumento2 pagineMini Polyatomic IonsJohann Daniel AlvarezNessuna valutazione finora
- Lesson Plan in Earth Science 1Documento5 pagineLesson Plan in Earth Science 1Gresel Gabawan100% (1)
- Easy Way To Understand Stewart DR George Handbook 2015 PDFDocumento48 pagineEasy Way To Understand Stewart DR George Handbook 2015 PDFAnonymous SMLzNANessuna valutazione finora
- IG2 Thermal Physics Practice TestDocumento5 pagineIG2 Thermal Physics Practice TestrehanNessuna valutazione finora
- Safety Data Sheet-Organic Exp.3Documento5 pagineSafety Data Sheet-Organic Exp.3Beste TırpanNessuna valutazione finora
- 5 - Estimation of Alkalinity of WaterDocumento4 pagine5 - Estimation of Alkalinity of WaterSai KumarNessuna valutazione finora
- Microbial Leaching 1Documento30 pagineMicrobial Leaching 1manmithNessuna valutazione finora
- Klyushin Ya.G. Electricity Gravity Heat PDFDocumento108 pagineKlyushin Ya.G. Electricity Gravity Heat PDFmuca110% (1)
- Thermofluids (1996, Springer US) PDFDocumento671 pagineThermofluids (1996, Springer US) PDFCristopher Aguaiza100% (1)
- Daily Lesson Log TemplateDocumento64 pagineDaily Lesson Log TemplateArlene ChavezNessuna valutazione finora
- A Design Procedure For High-Efficiency and Compact-Size 5-10-W MMIC Power Amplifiers in GaAs pHEMT TechnologyDocumento12 pagineA Design Procedure For High-Efficiency and Compact-Size 5-10-W MMIC Power Amplifiers in GaAs pHEMT Technologyln zNessuna valutazione finora
- Chapter 16 Acid Base Equilibria and Solubility Equilibria PDFDocumento120 pagineChapter 16 Acid Base Equilibria and Solubility Equilibria PDFlorrainebarandonNessuna valutazione finora
- Tathagata M.: Master of Sciences, Mechanical Engineering and Management September 2023 - PresentDocumento1 paginaTathagata M.: Master of Sciences, Mechanical Engineering and Management September 2023 - Presentkihic74264Nessuna valutazione finora
- Projectile MotionDocumento3 pagineProjectile MotionMark Leo Delos ReyesNessuna valutazione finora
- PS ReviewerDocumento3 paginePS ReviewerIt’s yanaNessuna valutazione finora
- Do Not Use Tables in Appendix A or B, Unless Stated OtherwiseDocumento2 pagineDo Not Use Tables in Appendix A or B, Unless Stated Otherwisesamala sonuNessuna valutazione finora
- Investigating The PH of The 11 Basic SubstancesDocumento3 pagineInvestigating The PH of The 11 Basic Substancesapi-29584515033% (3)
- NR21792 OcrDocumento475 pagineNR21792 Ocrmehrdad khoeiNessuna valutazione finora
- Particle Density Ecg 428Documento4 pagineParticle Density Ecg 428Aiman SyafiqNessuna valutazione finora
- Plate TheoryDocumento27 paginePlate TheoryDedex PutraNessuna valutazione finora
- The Index of Refraction: ConsequencesDocumento45 pagineThe Index of Refraction: ConsequencesSiddharth RajamohananNessuna valutazione finora
- 5.2.3 Redox and Electrode Potentials MSDocumento39 pagine5.2.3 Redox and Electrode Potentials MSUbayd HussainNessuna valutazione finora
- Suspension Acoustics - Temkin S PDFDocumento418 pagineSuspension Acoustics - Temkin S PDFDaniela VillegasNessuna valutazione finora
- Answers To 2.8 Exercises 2.8 Exercise 1: CL CL Cl. CLDocumento3 pagineAnswers To 2.8 Exercises 2.8 Exercise 1: CL CL Cl. CLSsNessuna valutazione finora
- Physics 403. Modern Physics Laboratory: Eugene V Colla, Anne SicklesDocumento56 paginePhysics 403. Modern Physics Laboratory: Eugene V Colla, Anne Sicklesally claveriaNessuna valutazione finora
- Disclosure To Promote The Right To InformationDocumento13 pagineDisclosure To Promote The Right To InformationVakkanthula SaikarthikNessuna valutazione finora
- The Subsystems of The EarthDocumento20 pagineThe Subsystems of The Earthcreate foxesNessuna valutazione finora