Documenti di Didattica
Documenti di Professioni
Documenti di Cultura
Case Study - Intestinal Resection and Anastomosis in A Dog
Caricato da
api-301746262Descrizione originale:
Titolo originale
Copyright
Formati disponibili
Condividi questo documento
Condividi o incorpora il documento
Hai trovato utile questo documento?
Questo contenuto è inappropriato?
Segnala questo documentoCopyright:
Formati disponibili
Case Study - Intestinal Resection and Anastomosis in A Dog
Caricato da
api-301746262Copyright:
Formati disponibili
Running head: CANINE INTESTINAL SURGERY
Case Study:
Intestinal Resection and Anastomosis in a Dog
Rebecca M. England
4325 Companion Animal Anesthesia and Surgical Nursing
Tarleton State University
CANINE INTESTINAL SURGERY
Elf is a one-year-old spayed female Beagle cross that presented with acute vomiting for
less than 24 hours (Rockett & Christensen, 2010). The owner was concerned that it may be due
to the ingestion of part of a pillow that she had recently torn apart. When the owner returned
home two days ago, she was unable to find locate most of the stuffing. Elf started vomiting about
24 hours later (Rockett & Christensen, 2010).
Her physical exam findings:
BCS: 3/5 at 44 lbs.
Temperature: 102.1 F
HR 120 bpm
RR panting
Mucous membranes: pale pink and dry
Respiratory: no abnormalities heard
Gastrointestinal: Resistance during abdominal palpation, discomfort seen, no F.B. detected
Dehydration assessment: 6% deficit
Due to the clinical history and presentation of Elf at this examination, the veterinarian
recommended abdominal radiographs be performed to assist in ruling out the possibility of a
foreign body obstruction. The owner consented. Survey radiographs, in addition to contrast
radiographs, were taken. In looking at the study, delayed gastric emptying was noted (Rockett &
Christensen, 2010). The veterinarian diagnosed vomiting secondary to foreign body ingestion, so
an abdominal exploratory was scheduled (Rockett & Christensen, 2010).
The veterinarian requested an ECG prior to induction of anesthesia (Rockett &
Christensen, 2010). A technician started the procedure, and it was quickly noted that it was being
performed incorrectly. The pictures in this text show two techniques, and Figure 5-9a is
obviously correct, shown on the next page (Rockett & Christensen, 2010). This placement is
correct in that the dog is placed in right lateral recumbency (not left) and the leads are placed
appropriately, slightly proximal to the elbows and stifles and on the correct limbs (Thomas &
Lerche, 2011). A fifth lead may sometimes be used at the apex of the heart on the left side of the
CANINE INTESTINAL SURGERY
thorax to obtain additional information. It appears to be appropriately placed in this picture. The
lead placement is often remembered by a common rhyme used among technicians, white on the
right (or snow over grass), and smoke over fire. This allows quick placement of the leads using
the colors of the leads. There is also a color photograph for comparison, but without the chest
and green leads, as it is difficult to determine if the leads are placed on the proper limbs due to
the black and white nature of the photograph.
Figure 5-9a: Obtaining an ECG on a dog. Correct positioning shown.
From Rockett & Christensen, 2010
Standard 3-lead ECG commonly used in veterinary patients, retrieved from personal photography library.
CANINE INTESTINAL SURGERY
In Figure 5-9b below, positioning of the patient and application of the leads is incorrect.
The patient is in left lateral recumbency and the leads have been placed distal to the elbows and
stifles. Again, there is also a color photograph for comparison, but without the chest and green
leads, as it is difficult to determine if the leads are placed on the proper limbs due to the black
and white nature of the photograph.
Figure 5-9b: Obtaining an ECG on a dog. Incorrect positioning shown.
From Rockett & Christensen, 2010
Standard 3-lead ECG commonly used in veterinary patients, retrieved from personal photography library.
CANINE INTESTINAL SURGERY
After the ECG is performed, the technician starts to gather equipment and supplies
needed for the induction and monitoring of anesthesia (Rockett & Christensen, 2010). The
technician gets out an esophageal stethoscope, shown below. An esophageal stethoscope is used
to manually auscultate and monitor heart rate and rhythm (Tear, 2012). If the end of the tubing is
adjacent to the lung fields, breath sounds may also be auscultated (Tear, 2012).
Esophageal stethoscope. From Tear, 2012
The veterinarian also wants Elfs percent oxygen saturation (Rockett & Christensen,
2010). The monitoring equipment needed to obtain this data is called a pulse oximeter (Tear,
2012), as seen below.
CANINE INTESTINAL SURGERY
The assistant prepared to use a non-rebreathing anesthetic set-up for this patient, seen
below. This tubing, however, is not appropriate for a patient of Elfs size, being that she is 44
pounds. Non-rebreathing circuits are for use in patients weighing less than 15 pounds.
A rebreathing circuit such as a universal F rebreathing circuit is appropriate for Elf, seen below.
It is ideal for patients larger in size weighing more than 15 pounds.
The size of rebreathing reservoir bag to be used must be calculated based on the patients weight
and tidal volume of the lungs. See the formula and calculation below:
CANINE INTESTINAL SURGERY
Tidal volume = body weight in pounds x 30 = bag size in milliliters (mL)
44 pounds x 30 = 1,320 mL
Tidal volume > 1,000 mL but < 2,000 mL = 2-liter bag
A 2-liter rebreathing reservoir bag is appropriate for Elf. A picture is shown below:
The veterinarian wants to use Olsen-Hegar needle drivers for the surgical procedure
(Rockett & Christensen, 2010). In the text (Rockett & Christensen, 2010), this instrument is
labeled A, seen below. This instrument in particular has scissor blades set behind the jaws,
whereas the Mayo-Hegar needle drivers have no scissor blades.
CANINE INTESTINAL SURGERY
The veterinary assistant heats up 0.9% sodium chloride in the incubator. Is this type of
fluid appropriate for flushing the abdomen? Yes, it is. 0.9% sodium chloride is an isotonic fluid,
which closely matches the bodys fluids. It is primarily used in the event that intestinal contents
leaked into the abdominal cavity (Tear, 2012). Flushing the abdomen will decrease the chance of
peritonitis from occurring.
The veterinarian needs a specific type of towel clamp that would be appropriate for holding the
suction tubing in place. The other clamp choices are not appropriate as they are sharp and could
potentially puncture the drape material or the suction tubing. This clamp is identified as C in the text
(Rockett & Christensen, 2010), and shown below. It is blunt and will not penetrate materials.
CANINE INTESTINAL SURGERY
Prior to anesthetizing Elf for her surgical procedure, an IV catheter is placed so that she
can receive fluids to replace her dehydration deficit. She is considered to be 6% dehydrated. The
volume of fluid she will need to replace the deficit is calculated below:
Dehydration deficit calculation:
Dehydration % x BWlb x 500
6% x 44 lbs. x 500 = 1,320 ml to replace the deficit.
After receiving fluids, Elf was induced using propofol at a dose of 2 mg/lb. She received
8.8 ml of propofol IV for induction. See the calculation below:
Propofol is 10 mg/ml (Plumb, 2011)
44 lbs. * 2 mg/lb. = 88 mg
88 mg divided by 10 mg/ml = 8.8 ml propofol
CANINE INTESTINAL SURGERY
10
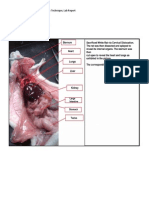
A large amount of pillow batting was removed from the proximal portion of the jejunum
(Rockett & Christensen, 2010). The assistant asks about the location of the jejunum in
relationship to the stomach. Here is a drawing below, depicting the gastrointestinal system of a
dog:
Once the surgery is complete, Elf is monitored in the recovery room. Extubation should
be performed until the patients primary reflexes return. These include a palpebral reflex
response and the ability to swallow. The ability to swallow is most crucial for the patient. If the
patient is unable to swallow, and then subsequently extubated too early, the patient will not be
able to protect the airway. This could lead to aspiration, an inability to breath, and even death.
Therefore, a patient should not be extubated until swallowing well. Five minutes after
extubation, Elf starts to vomit. It is important in a situation like this that the patient be propped
up in a sternal position to minimize the chances of aspirating fluid or other contents from the
CANINE INTESTINAL SURGERY
11
gastrointestinal tract. If severe or the patient has complications breathing, reintubation may be
necessary. Otherwise, monitor and keep the patient sternal until the vomiting has subsided. It
may be necessary to administer antinausea medication per the veterinarians orders if it persists.
Now that the surgery room is no longer being used, it must be prepared for the next
patient. There are several things that should be done to clean and maintain the room properly
(Tear, 2012):
1. The O.R. should be cleaned daily, even if it is not used
2. Equipment used to clean the O.R. should only be used in the O.R to prevent crosscontamination
3. All surfaces of the room should be damp-dusted and wiped down before and after
each patient
4. Any equipment and furniture that is visibly soiled needs to be cleaned at the end of
each procedure
5. All instruments should be removed from the O.R. and washed/cleaned/repackaged in
a designated area
6. Surfaces should be cleaned with an appropriate disinfectant (this will vary depending
on the hospitals need and exposure to diseases)
7. When cleaning surfaces, a scrubbing motion should be used
8. Examples of surfaces to clean include:
a. Surgical lights
b. Tables
c. Glass sliding cabinets
d. Wheels, step stools, foot pedals, wall telephones, light switches
e. All handles (doors, drawers, and cabinets)
f. Anesthesia machine
g. Ventilation faceplates and doorway ledges
9. The floor should be swept and mopped with a fresh bucket of appropriate disinfectant
and a clean mop head
10. The surgery door should be kept closed at all times to reduce the chances of
contamination
Based on hospitals standards, there may be additional things to perform. Overall, however, the
concept is the same.
This case study reveals the wide variety of knowledge that is necessary to carry out a
surgical procedure such as this one. A variety of equipment is needed, and one must be familiar
with its function and how to apply it to patient use. Several calculations are also necessary, from
CANINE INTESTINAL SURGERY
12
induction to the post-operative period. Patient anatomy is especially important to understand, not
only from a procedural standpoint, but also in order to educate owners. Maintaining the O.R. is
also an vital daily task.
CANINE INTESTINAL SURGERY
13
References
Colville, T. and Bassert, J. (2007). Clinical Anatomy and Physiology for Veterinary Technicians, 2nd ed.
St. Louis, MO: Mosby.
Plumb, D. C. (2011). Plumbs Veterinary Drug Handbook, 7th ed. PharmVet Inc.
Rockett and Christensen (2010). Case Studies in Veterinary Technology: A Scenario-Based Critical
Thinking Approach. Heyburn, ID: Rockett House Publishing LLC.
Sonsthagen, T. (2005). Veterinary Instruments and Equipment: A Pocket Guide. St. Louis, MO: Mosby.
Tear, M. (2012). Small Animal Surgical Nursing: Skills and Concepts, 2nd ed. St. Louis, MO: Mosby
Elsevier.
Thomas, J. A. and Lerche, P. (2011). Anesthesia and Analgesia for Veterinary Technicians, 4th ed. St.
Louis, MO: Elsevier.
Potrebbero piacerti anche
- Human Anatomy Physiology Laboratory Manual Fetal Pig Version 12th Edition Marieb Solution ManualDocumento5 pagineHuman Anatomy Physiology Laboratory Manual Fetal Pig Version 12th Edition Marieb Solution Manualsritrader90% (10)
- Case Study - Seizuring DogDocumento8 pagineCase Study - Seizuring Dogapi-301746262Nessuna valutazione finora
- Case Study - Seizuring DogDocumento8 pagineCase Study - Seizuring Dogapi-301746262Nessuna valutazione finora
- J Exp Med-1922-Warren-187-202Documento16 pagineJ Exp Med-1922-Warren-187-202Anggun PermatasariNessuna valutazione finora
- Evaluation of A Protocol For Fast Localised Abdominal Sonography of Horses (FLASH) Admitted For ColicDocumento6 pagineEvaluation of A Protocol For Fast Localised Abdominal Sonography of Horses (FLASH) Admitted For Colicludiegues752Nessuna valutazione finora
- How To Anaesthetize A GiraffeDocumento4 pagineHow To Anaesthetize A Giraffeltrathie3087100% (2)
- Anaesthesia NotesDocumento61 pagineAnaesthesia NotessuggaplumNessuna valutazione finora
- The Yale Preoperative Anxiety Scale How Does It.Documento6 pagineThe Yale Preoperative Anxiety Scale How Does It.Karen ValdezNessuna valutazione finora
- Canadian C-Spine Rule Could Help Trauma Patients, Ease Overcrowding in Emergency Departments, Study FindsDocumento4 pagineCanadian C-Spine Rule Could Help Trauma Patients, Ease Overcrowding in Emergency Departments, Study FindsAmrie HeussaffNessuna valutazione finora
- Group B: Romero D. Romero P. Rufino L. Saddi E. Sanchez D. Santos ADocumento19 pagineGroup B: Romero D. Romero P. Rufino L. Saddi E. Sanchez D. Santos APam RomeroNessuna valutazione finora
- Anestesia em Animais Silvestres Capitulo 1Documento24 pagineAnestesia em Animais Silvestres Capitulo 1Daniel CostaNessuna valutazione finora
- An Alternative Method of Endotracheal Intubation of Common MarmosetsDocumento6 pagineAn Alternative Method of Endotracheal Intubation of Common Marmosetssolxmar tmNessuna valutazione finora
- Endotracheal SuctionDocumento11 pagineEndotracheal SuctionKeshav Singhmaar AryaNessuna valutazione finora
- Case Scenario ArfDocumento3 pagineCase Scenario ArfPam RomeroNessuna valutazione finora
- Total Injectable Anesthesia of Dogs and Cats For Remote Location Veterinary Sterilization Clinic BMC 2020Documento6 pagineTotal Injectable Anesthesia of Dogs and Cats For Remote Location Veterinary Sterilization Clinic BMC 2020Daniel DanielNessuna valutazione finora
- Rapid Sequence Intubation W Checklist 2022Documento4 pagineRapid Sequence Intubation W Checklist 2022drulilalbbNessuna valutazione finora
- Reeves 2001Documento11 pagineReeves 2001Luis José Hume QuirozNessuna valutazione finora
- Ultrasonography For The Veterinary Nurse - Frank BuschDocumento2 pagineUltrasonography For The Veterinary Nurse - Frank BuschFrank Busch100% (1)
- Table 1 Indications, Contraindications and Hazards of Suctioning Indications of SuctioningDocumento4 pagineTable 1 Indications, Contraindications and Hazards of Suctioning Indications of SuctioningRika RukmanaNessuna valutazione finora
- Animal Facility Videoendoscopic Intubation Station: Tips and Tricks From Mice To RabbitsDocumento4 pagineAnimal Facility Videoendoscopic Intubation Station: Tips and Tricks From Mice To RabbitsJavier Martin Bartolo GarciaNessuna valutazione finora
- Linsenmeier 2020Documento6 pagineLinsenmeier 2020Novita ApramadhaNessuna valutazione finora
- Nursing Facts in Brief Bio Physical Concept in Nursing and Rehabilitation/laboratory TestsDocumento9 pagineNursing Facts in Brief Bio Physical Concept in Nursing and Rehabilitation/laboratory TestsDhine Dhine ArguellesNessuna valutazione finora
- Histo Tech Lab Report 1Documento4 pagineHisto Tech Lab Report 1Hendry RaoNessuna valutazione finora
- Monitoring Peri Ajnestesi DepiDocumento19 pagineMonitoring Peri Ajnestesi DepijoyfullNessuna valutazione finora
- Anesthesia, Chemical Restraint and Pain Management in Snakes (Serpentes) - A Review Seven Mustafa, Nadya ZlatevaDocumento8 pagineAnesthesia, Chemical Restraint and Pain Management in Snakes (Serpentes) - A Review Seven Mustafa, Nadya ZlatevaFirdaus BillyNessuna valutazione finora
- Tabije, Arvie Jayselle PDocumento8 pagineTabije, Arvie Jayselle PJaysellePuguonTabijeNessuna valutazione finora
- English For Professional Nurse 2Documento7 pagineEnglish For Professional Nurse 2Anonymous HWsv9pBUhNessuna valutazione finora
- Nursing Management in Abdominal SurgeryDocumento19 pagineNursing Management in Abdominal Surgeryejguy7777100% (2)
- IV-C Safety Guidelines For Emergency Drugs, Endotracheal Set-Up and Defibrillator UseDocumento21 pagineIV-C Safety Guidelines For Emergency Drugs, Endotracheal Set-Up and Defibrillator UseRio DanteNessuna valutazione finora
- Safe Recovery From AnaesthesiaDocumento7 pagineSafe Recovery From AnaesthesiaKarthikNessuna valutazione finora
- Necropsy Guide for Dogs, Cats, and Small MammalsDa EverandNecropsy Guide for Dogs, Cats, and Small MammalsSean P. McDonoughValutazione: 4 su 5 stelle4/5 (1)
- Chapter 7 HandoutDocumento5 pagineChapter 7 HandoutFrance KennethNessuna valutazione finora
- Et IntubationDocumento3 pagineEt IntubationCherry L. M. JaramilloNessuna valutazione finora
- Operasi Pada TikusDocumento8 pagineOperasi Pada Tikuslathifa_nurNessuna valutazione finora
- LaryngectomyDocumento7 pagineLaryngectomyŞahin ŞahinNessuna valutazione finora
- Anesthesia Rabbits PreDocumento4 pagineAnesthesia Rabbits PreAgungLailatulKurniawanNessuna valutazione finora
- Resume Nasogastric Tube InstallationDocumento17 pagineResume Nasogastric Tube InstallationRedmy LasmanaNessuna valutazione finora
- Ateneo de Zamboanga University: Nursing Skills Output (Nso)Documento9 pagineAteneo de Zamboanga University: Nursing Skills Output (Nso)Mitzi BelamideNessuna valutazione finora
- Pronation Therapy: DR Ahmed Abdelhady MD Critical Care ICU ConsultantDocumento30 paginePronation Therapy: DR Ahmed Abdelhady MD Critical Care ICU Consultantlayla alyamiNessuna valutazione finora
- Rodent Euthanasia Adult NIHDocumento2 pagineRodent Euthanasia Adult NIHgabriela_mariangela5929Nessuna valutazione finora
- Case ReportDocumento9 pagineCase ReportAnanta IpoNessuna valutazione finora
- Goligorsky M.S. - Renal Disease. Techniques and Protocols (2003)Documento485 pagineGoligorsky M.S. - Renal Disease. Techniques and Protocols (2003)NMC NEPHROLOGYNessuna valutazione finora
- 4 Pre-Eclampsia & Eclampsia - Eclampsia - Scenario 1Documento4 pagine4 Pre-Eclampsia & Eclampsia - Eclampsia - Scenario 1rhtyehNessuna valutazione finora
- QS EndotrachealIntubationDocumento4 pagineQS EndotrachealIntubationChandra HalimNessuna valutazione finora
- Ja Alas 2009000292Documento4 pagineJa Alas 2009000292MihEugenNessuna valutazione finora
- Part 1. The Erlanger Endo-Trainer.Documento4 paginePart 1. The Erlanger Endo-Trainer.Alberto Kenyo Riofrio PalaciosNessuna valutazione finora
- Als ProceduresDocumento3 pagineAls Proceduresapi-296884857Nessuna valutazione finora
- Assisting For Endotracheal IntubationDocumento16 pagineAssisting For Endotracheal IntubationSREEDEVI T SURESH100% (1)
- OSCE Marksheet 1Documento7 pagineOSCE Marksheet 1Elias PetrouNessuna valutazione finora
- Dwnload Full Human Anatomy and Physiology Laboratory Manual Fetal Pig Version Update 10th Edition Marieb Solutions Manual PDFDocumento36 pagineDwnload Full Human Anatomy and Physiology Laboratory Manual Fetal Pig Version Update 10th Edition Marieb Solutions Manual PDFpeiwenhlmel100% (9)
- Adult Rodents - Chemical Methods 1. CO Asphyxiation: EUTHANASIA in Laboratory AnimalsDocumento3 pagineAdult Rodents - Chemical Methods 1. CO Asphyxiation: EUTHANASIA in Laboratory Animalsjekapu simwingaNessuna valutazione finora
- The Development, Concept and Use of Medical Equipment As Science and Technology AdvancesDocumento34 pagineThe Development, Concept and Use of Medical Equipment As Science and Technology Advancesmatt_21_luiNessuna valutazione finora
- Solution Manual For Human Anatomy Laboratory Manual With Cat Dissections 8Th Edition Marieb 9780134255583 9780134255583 Full Chapter PDFDocumento29 pagineSolution Manual For Human Anatomy Laboratory Manual With Cat Dissections 8Th Edition Marieb 9780134255583 9780134255583 Full Chapter PDFleo.kitchen421100% (11)
- Ovrio Salpingo Histerectomiavideolaparoscpicaemgatosdomsticos TcnicacomdoisportaisDocumento6 pagineOvrio Salpingo Histerectomiavideolaparoscpicaemgatosdomsticos TcnicacomdoisportaisDra MeiNessuna valutazione finora
- HP May03 CapsuleDocumento7 pagineHP May03 CapsuleManeesh RainaNessuna valutazione finora
- ThyroidectomyDocumento51 pagineThyroidectomyPopey Moore100% (1)
- Bennett - 2011 - The Chelonian Respiratory SystemDocumento15 pagineBennett - 2011 - The Chelonian Respiratory SystemdsouzapeixotoNessuna valutazione finora
- Icu Design and SetupDocumento9 pagineIcu Design and SetupNorfaidah AliNessuna valutazione finora
- Single Incision, Laparoscopic-Assisted Ovariohysterectomy For Mucometra and Pyometra in DogsDocumento5 pagineSingle Incision, Laparoscopic-Assisted Ovariohysterectomy For Mucometra and Pyometra in Dogsalfi fadilah alfidruNessuna valutazione finora
- Neonatal IntubationDocumento7 pagineNeonatal Intubationdrsalil82Nessuna valutazione finora
- Respiratory EmergencyDocumento6 pagineRespiratory Emergencyapi-235947247Nessuna valutazione finora
- Laparoscopic Colopexy in Dogs: Full Paper SurgeryDocumento6 pagineLaparoscopic Colopexy in Dogs: Full Paper SurgeryrianperoNessuna valutazione finora
- FleasDocumento27 pagineFleasapi-301746262100% (6)
- Retrogade CystographyDocumento16 pagineRetrogade Cystographyapi-301746262Nessuna valutazione finora
- Educational Program 2 - Importance of Post-Operative CareDocumento9 pagineEducational Program 2 - Importance of Post-Operative Careapi-301746262Nessuna valutazione finora
- DetoxificationDocumento20 pagineDetoxificationapi-301746262100% (2)
- Midterm Case Study PPT - Heartworm Infection in A DogDocumento33 pagineMidterm Case Study PPT - Heartworm Infection in A Dogapi-301746262Nessuna valutazione finora
- Week 11 Case Study - Dog With PruritusDocumento9 pagineWeek 11 Case Study - Dog With Pruritusapi-301746262Nessuna valutazione finora
- 9 The Post Anesthesia Care UnitDocumento77 pagine9 The Post Anesthesia Care Unitsanjivdas100% (1)
- New York Medicaid APG CrosswalkDocumento6.734 pagineNew York Medicaid APG CrosswalksdpetesrhsNessuna valutazione finora
- Pathophysiology: Cardiovascular System Dr. Maen DweikDocumento38 paginePathophysiology: Cardiovascular System Dr. Maen DweikshoibyNessuna valutazione finora
- Quiz orDocumento2 pagineQuiz orJo-Mar Arellano Avena0% (3)
- Infiltration Anaesthesia For Tympanoplasty: A Comparison of Dexmedetomidine and Clonidine As Adjuvants To Lignocaine and AdrenalineDocumento7 pagineInfiltration Anaesthesia For Tympanoplasty: A Comparison of Dexmedetomidine and Clonidine As Adjuvants To Lignocaine and AdrenalineIJAR JOURNALNessuna valutazione finora
- Dexmed Mag SufDocumento5 pagineDexmed Mag SufAshiyan IrfanNessuna valutazione finora
- Principles of Neurophysiological Assessment, Mapping, and MonitoringDocumento28 paginePrinciples of Neurophysiological Assessment, Mapping, and Monitoringmlslm202380Nessuna valutazione finora
- R No.1 Dhara Aparnathi e No-2104321001Documento14 pagineR No.1 Dhara Aparnathi e No-2104321001Lal NandaniNessuna valutazione finora
- Reiki Manuals 1 + 2 - Leonie Faye PDFDocumento100 pagineReiki Manuals 1 + 2 - Leonie Faye PDFvictory v100% (17)
- Test Bank For Local Anesthesia For The Dental Hygienist 2nd Edition by LogothetisDocumento12 pagineTest Bank For Local Anesthesia For The Dental Hygienist 2nd Edition by Logothetistracywilsonjftmrqknys100% (29)
- Book 2 (74-143)Documento70 pagineBook 2 (74-143)Shaik NisaNessuna valutazione finora
- LD Gingival RetractionDocumento39 pagineLD Gingival RetractionRohan GroverNessuna valutazione finora
- ParacentesisDocumento3 pagineParacentesisVenus April LimonNessuna valutazione finora
- Guidelines in Practice: Hypothermia Prevention: Terri Link, MPH, BSN, Cnor, Cic, Caip, FapicDocumento14 pagineGuidelines in Practice: Hypothermia Prevention: Terri Link, MPH, BSN, Cnor, Cic, Caip, Fapicputu rusmiathiNessuna valutazione finora
- AQI Is The Definition of QualityDocumento3 pagineAQI Is The Definition of QualityeryxspNessuna valutazione finora
- As3000 Service Manual PDFDocumento180 pagineAs3000 Service Manual PDFsambadeeNessuna valutazione finora
- Filamer Christian Institute Vs CADocumento4 pagineFilamer Christian Institute Vs CADustin NitroNessuna valutazione finora
- Computer Based Patient Record For Anaesthesia (Dec 1997)Documento3 pagineComputer Based Patient Record For Anaesthesia (Dec 1997)Sirisha CkvNessuna valutazione finora
- ROGELIO E. RAMOS and ERLINDA RAMOS, in their own behalf and as natural guardians of the minors, ROMMEL RAMOS, ROY RODERICK RAMOS and RON RAYMOND RAMOS, petitioners, vs. COURT OF APPEALS, DELOS SANTOS MEDICAL CENTER, DR. ORLINO HOSAKA and DRA. PERFECTA GUTIERREZ, respondents.Documento26 pagineROGELIO E. RAMOS and ERLINDA RAMOS, in their own behalf and as natural guardians of the minors, ROMMEL RAMOS, ROY RODERICK RAMOS and RON RAYMOND RAMOS, petitioners, vs. COURT OF APPEALS, DELOS SANTOS MEDICAL CENTER, DR. ORLINO HOSAKA and DRA. PERFECTA GUTIERREZ, respondents.Magne Joy Binay - AlcantaraNessuna valutazione finora
- Physical Exam: Anesthetic Plan: Asa #Documento1 paginaPhysical Exam: Anesthetic Plan: Asa #Jeremy-ann HamNessuna valutazione finora
- Articaine 30 Years LaterDocumento27 pagineArticaine 30 Years LaterChristianEdwarZuñigaNessuna valutazione finora
- Cattle Dehorning - Questions and AnswersDocumento5 pagineCattle Dehorning - Questions and AnswersAfan Syafi'iNessuna valutazione finora
- Congenital Diaphragmatic HerniaDocumento29 pagineCongenital Diaphragmatic Herniapaddy9980Nessuna valutazione finora
- SW Presentation Kroening Medical GasDocumento116 pagineSW Presentation Kroening Medical GasYudi Wahyudi WahyudiantoNessuna valutazione finora
- LA Nerve DamageDocumento6 pagineLA Nerve DamagebabukanchaNessuna valutazione finora
- Typhon Case LogDocumento3 pagineTyphon Case LogJeremy HallNessuna valutazione finora
- Temperature Regulation and Monitoring-Samar TahaDocumento57 pagineTemperature Regulation and Monitoring-Samar TahapaulaNessuna valutazione finora
- Guidelines For Perioperative Care in Esophagectomy Enhanced Recovery After Surgery (ERAS®) Society Recommendations.Documento32 pagineGuidelines For Perioperative Care in Esophagectomy Enhanced Recovery After Surgery (ERAS®) Society Recommendations.FlorNessuna valutazione finora
- 1 s2.0 S0883540321005842 MainDocumento6 pagine1 s2.0 S0883540321005842 MainSavNessuna valutazione finora