Documenti di Didattica
Documenti di Professioni
Documenti di Cultura
Dynamic Resetting of The Human Circadian Pacemaker by Intermittent Bright Light
Caricato da
api-308065432Descrizione originale:
Titolo originale
Copyright
Formati disponibili
Condividi questo documento
Condividi o incorpora il documento
Hai trovato utile questo documento?
Questo contenuto è inappropriato?
Segnala questo documentoCopyright:
Formati disponibili
Dynamic Resetting of The Human Circadian Pacemaker by Intermittent Bright Light
Caricato da
api-308065432Copyright:
Formati disponibili
Am J Physiol Regulatory Integrative Comp Physiol
279: R1574R1579, 2000.
Dynamic resetting of the human circadian pacemaker by
intermittent bright light
DAVID W. RIMMER,1 DIANE B. BOIVIN,1 THERESA L. SHANAHAN,1
RICHARD
E. KRONAUER,2 JEANNE F. DUFFY,1 AND CHARLES A. CZEISLER1
1
Circadian, Neuroendocrine and Sleep Disorders Section, Division of Endocrinology, Department of
Medicine, Harvard Medical School, Brigham and Womens Hospital, Boston 02115; and 2Division of
Engineering and Applied Sciences, Harvard University, Cambridge, Massachussets 02138
Received 15 March 2000; accepted in final form 16 June 2000
Rimmer, David W., Diane B. Boivin, Theresa L.
Shanahan, Richard E. Kronauer, Jeanne F. Duffy, and
Charles A. Czeisler. Dynamic resetting of the human circadian pacemaker by intermittent bright light. Am J Physiol
Regulatory Integrative Comp Physiol 279: R1574R1579,
2000.In humans, experimental studies of circadian resetting typically have been limited to lengthy episodes of exposure to continuous bright light. To evaluate the time course of
the human endogenous circadian pacemakers resetting response to brief episodes of intermittent bright light, we studied 16 subjects assigned to one of two intermittent lighting
conditions in which the subjects were presented with intermittent episodes of bright-light exposure at 25- or 90-min
intervals. The effective duration of bright-light exposure was
31% or 63% compared with a continuous 5-h bright-light
stimulus. Exposure to intermittent bright light elicited almost as great a resetting response compared with 5 h of
continuous bright light. We conclude that exposure to intermittent bright light produces robust phase shifts of the endogenous circadian pacemaker. Furthermore, these results
demonstrate that humans, like other species, exhibit an
enhanced sensitivity to the initial minutes of bright-light
exposure.
in understanding photic
resetting of the human circadian pacemaker since it
was initially recognized that light is the principal circadian synchronizer in humans (9, 12, 14, 18, 27, 34,
41). Photic induction of circadian phase shifts has been
characterized extensively and is known to depend on
several variables including the timing, duration, and
intensity of retinal light exposure. Techniques of bright
light administration gleaned from these principles
have been implemented successfully not only in the
laboratory setting but also in the treatment of a variety
of disorders including maladaptation to night shift
work, delayed and advanced sleep phase syndrome,
circadian rhythm sleep disorders, dyssomnia associated with transmeridian travel and spaceflight, as well
as age-related early morning awakening (3, 4, 7, 8, 10,
13, 17, 23, 34, 38, 40, 41). However, the use of brightlight therapy in these settings typically has consisted
of 2- to 8-h continuous exposures. The imposition of
time constraints involved in such extended exposures
obviates the need to develop, on the one hand, more
efficient means of delivering bright-light therapy,
whereas on the other hand, retaining the ability to
induce phase shifts of comparable magnitude to those
obtained through extended exposures.
Much of the evidence supporting the notion that the
endogenous circadian pacemaker may be sensitive to
shorter durations of light exposure comes from animal
models. In 1991, Nelson and Takahashi (35) demonstrated that the action of light as a synchronizer of the
circadian clock in the golden hamster Mesocricetus
auratus was most efficient during the first few minutes
of the application and that further extension of the
stimulus produced little additional phase shift. This
finding was consistent with photic resetting properties
observed in the mosquito (37). Furthermore, it has
been reported that the circadian system of mice can
integrate a chain of 60 very brief (2 ms) light pulses to
cause circadian phase shifts of up to 2 h (42). Yet, the
temporal dynamics of the circadian phase resetting
response to light in humans remains unquantified.
This is particularly notable because exposure to bright
light is typically intermittent in everyday life (21, 26,
36, 39). Therefore, to characterize whether humans,
like rodents and mosquitoes, have enhanced sensitivity
to the initial minutes of intermittent light exposure, we
undertook experiments to examine whether repeated
brief exposures to 9,500 lx of light produce robust
phase shifts of the circadian timing system. We compared the phase shifts produced by those brief, repeated light exposures to those elicited by continuous
light exposure and by darkness to estimate the temporal dynamics of the human circadian response to light.
Our results reveal that the endogenous circadian timing system of humans is far more sensitive to shorterduration light exposure than was previously recognized.
Address for reprint requests and other correspondence: C. A.
Czeisler, Circadian, Neuroendocrine and Sleep Disorders Section,
Div. of Endocrinology, Harvard Medical School, Brigham and Womens Hospital, 221 Longwood Ave., Boston, MA 021155817.
The costs of publication of this article were defrayed in part by the
payment of page charges. The article must therefore be hereby
marked advertisement in accordance with 18 U.S.C. Section 1734
solely to indicate this fact.
circadian rhythms; core body temperature; phototherapy
MANY ADVANCES HAVE BEEN MADE
R1574
0363-6119/00 $5.00 Copyright 2000 the American Physiological Society
http://www.ajpregu.org
INTERMITTENT LIGHT RESETS THE HUMAN CIRCADIAN PACEMAKER
R1575
MATERIALS AND METHODS
Subjects. Sixteen healthy men (24 2.1 yr, means SD)
were recruited for study using newspaper advertisements
and billboard flyers. All subjects were free from any acute or
chronic physical or psychiatric disorders. Medical suitability
was determined on the basis of medical history, physical
examination, ophthalmologic examination, and blood and
urine chemistries. Psychological screening questionnaires,
including the Minnesota Multiphasic Personality Inventory
and the Beck Depression Inventory and State Anxiety Scale
were used to exclude subjects with evidence of psychopathology from participation in the study. Individuals with a history of drug dependency, psychiatric illness, or affective disorder such as major depression or manic-depressive illness,
were excluded from study. Subjects were asked to refrain
from using alcohol, nicotine, prescription drugs, dietary supplements, and caffeine throughout the course of the study;
urinary toxicological analysis was performed to ensure that
subjects were drug free on admission to the laboratory. All
subjects studied reported no recent history of regular nightshift work, and none reported crossing more than one time
zone in the previous 3 mo. Subjects were asked to maintain a
regular sleep-wake schedule for 3 wk at home before the
study, such that their habitual bedtimes and wake times
occurred at the same time (30 min) each day and were 8 h
apart. Compliance was verified by sleep-wake logs and wrist
actigraphy monitoring for the week before the study. The
protocols used were reviewed and approved by the Human
Research Committee at the Brigham and Womens Hospital,
and each subject gave written informed consent before the
start of the inpatient portion of the protocol.
Experimental protocol. Subjects underwent an experimental protocol designed to measure the phase-shifting effects of
intermittent light on the endogenous core body temperature
cycle. All studies were performed in the Environmental
Scheduling Facility of the General Clinical Research Center
of the Brigham and Womens Hospital.
On empanelment in the 2-wk study, each subject underwent 3 adaptation days, with the time of their 8-h sleep
episode schedule based on their habitual bedtime and wake
time obtained from the prestudy sleep-wake logs. Beginning
at wake time on the fourth day, subjects underwent a constant-routine (CR) procedure (detailed below) that consisted
of 3033 h of enforced wakefulness to assess the phase of the
endogenous circadian core body temperature rhythm. The
CR continued until 9.5 h after the fitted minimum of the core
body temperature rhythm on day 5, at which time subjects
were allowed to sleep for 8 h. For the next 3 consecutive days,
subjects were then scheduled to undergo one of two intermittent bright-light conditions (detailed below) followed by a
final CR assessment of endogenous circadian phase that
lasted 4050 h (Fig. 1).
From admission to the laboratory until 1.5 h before bedtime
on day 3, subjects lived in 150 lx of light during waking hours.
From that point forward, room light intensity was lowered to
1015 lx during waking hours (except during bright lightexposure episodes) for the remainder of the study. All sleep
episodes were conducted in 0.02 lx (all reported light intensities were measured in the angle of gaze).
CR. We used the CR procedure to reveal the endogenous
component of the circadian core body temperature rhythm.
This methodology is a modification of the original technique
used by Mills et al. (32) and has been successfully used to
minimize or distribute evenly the potential masking effects
associated with changes in activity, posture, sleep-wake
state, diet, and ambient temperature across the circadian
Fig. 1. Double raster plot of study protocol. Successive days are
plotted both beneath each other as well as side by side. Solid bars
indicate scheduled sleep episodes, open bars represent the constant
routines (CR), and open circles represent the estimated endogenous
circadian phase of the core body temperature minimum (CBTmin).
Open boxes (sun) represent the 3-cycle, 5-h/day bright-light stimuli.
For the first 3 days, subjects 8-h sleep episodes were scheduled to
occur at their habitual bedtimes and were conducted in darkness
(0.03 lx). Ambient light intensity during waking hours was 150 lx
until 1.5 h before bedtime on day 3. Thereafter, all waking hours
were conducted in 1015 lx for the remainder of the study (gray
area). Bright-light stimuli were centered 1.5 h after the CBTmin
determined during the first CR. Subsequent sleep episodes were
scheduled to begin 9.5 h after the initial CBTmin, the midpoint of
which corresponded to 12 h opposite the midpoint of the bright-light
stimuli. The phase-shifting effect of the bright-light treatments was
assessed during the second CR.
cycle (9, 12, 15, 33). When successfully employed, this technique affords an assessment of the output of endogenous
circadian pacemaker, as reflected by the core body temperature rhythm. The CR procedure in these studies consisted of
a regimen of enforced semirecumbent wakefulness with activity restricted to prevent changes in body posture. Daily
caloric intake was calculated using the Wilmore nomogram
(44) that was adjusted upward by an activity factor of 10% to
maintain a consistent metabolic state across circadian
phases. Sustenance was provided in the form of hourly
snacks and liquid aliquots containing 150 mmol of sodium
and 100 mmol of potassium per 24-h period. A technician was
present throughout the CRs to ensure compliance with the
posture, activity level, and constant wakefulness aspects of
the protocol.
Lighting regimen. Subjects were assigned to one of two
intermittent lighting conditions that consisted of a threecycle, 5-h episode of exposure to bright light (9,500 lx in the
angle of gaze) interrupted by episodes of complete darkness.
This episode was centered on average 1.51 0.08 h (means
SD) after the initial endogenous temperature minimum and
administered against a background of dim light (1015 lx).
Within each 5-h episode, darkness episodes were repeated at
90- or 25-min intervals (duty cycle; Fig. 2, c and d ). Subject
comfort required that the light-dark transitions be effected
with moderate stepwise changes that were spaced uniformly
in time. As a result, the subjects were in darkness for 44
min of the 90-min duty cycle or 19.7 min of the 25-min duty
cycle. The effective duration of bright-light exposure for each
5-h intermittent stimulus together with the 25-min transitions before and after the 5-h stimulus was 63% or 31%,
respectively, compared with the continuous bright-light protocol (see below). Results obtained from these two groups of
subjects were compared with historical data from 15 volunteers (21.67 3.54 yr, mean SD) who participated in the
R1576
INTERMITTENT LIGHT RESETS THE HUMAN CIRCADIAN PACEMAKER
Fig. 2. Lighting regimen used to study the phase-shifting effect of 3
cycles of 5-h bright-light exposures on the endogenous circadian
pacemaker. Exposures were centered 1.5 h after the endogenous
circadian core body temperature minimum (time 0 on axis) to induce
phase advances of the endogenous circadian pacemaker (a). Each
stimulus consisted of exposure to either 3 cycles of 5 h of continuous
bright (10,000 lx) light (100% stimulus; b), 3 cycles of 5 h of bright
light consisting of 46 min of light within each 90-min duty cycle
(63% stiumulus; c), 3 cycles of 5 h of bright light consisting of 5.3
min of bright light within each 25-min duty cycle (31% stimulus; d),
or complete darkness (0% stimulus; e). Each 5-h stimulus was preceded and followed by 25-min transitions during which the intensities were increased and decreased in 5-min intervals. Similarly,
within the 90- and 25-min duty cycles, light intensities were progressively increased and decreased in 1-min intervals and 10-s intervals,
respectively. The effective duration of bright-light exposure producing drive on the endogenous pacemaker for all transitions are represented by the dashed lines (28).
same experimental protocol but were exposed to a 5-h stimulus consisting of either continuous (100%) bright light (Fig.
2b) or continuous (0%) darkness (control; Fig. 2e) (16).
Physiological monitoring. The bright-light stimuli were
administered using ceiling-mounted cool-white fluorescent
lamps (North American Philips Lighting, Bloomfield, NJ).
Light measurements were performed using a research photometer [model 1700 and SED (SEL) 038/Y 7859/W 3339
detector; International Light, Newburyport, MA] held at the
forehead and directed in the line of gaze. Subjects were
outfitted with clear goggles throughout the 5-h intermittent
light episodes (model #S379; Uvex Safety, Smithfield, RI) to
exclude ultraviolet exposure.
Throughout the entire study, subjects were maintained in
an environment free from time cues, and social contact was
limited to the laboratory personnel who were specially
trained to apply the physiological monitoring devices, administer light stimuli, ensure wakefulness, and shield the subjects from temporal information.
Core body temperature was recorded via disposable rectal
thermistors (Yellow Springs Instrument, Yellow Springs,
OH), and room temperature was recorded by a surface thermistor and maintained at 29.6 0.9C (mean SD). Both
the core body temperature and room temperature were recorded at 1-min intervals by means of a real-time, online
data-acquisition system using IBM PC-compatible, Pentiumbased computers.
Statistical analysis. Circadian phase was estimated from
the core body temperature data derived from the CRs by a
dual-harmonic regression plus correlated noise model fit to
the data by a nonlinear least-squares procedure (6). The first
5 h of CR temperature data were excluded from analysis to
minimize the masking effects resulting from the transition
from sleep to wake and postural changes associated with
beginning the CR. Phase shifts were calculated as the difference (in hours) in the time of the fitted core body temperature
minima between the first and second CRs. By convention,
phase advances (to an earlier hour) are represented as positive phase shifts, whereas phase delays (to a later hour) are
negative phase shifts. An ANOVA, with planned tests for
linear and quadratic trends, as well as post hoc pairwise
comparisons using Tukeys method, were used to evaluate
the effect of the intervention regimen on phase shifts between groups of subjects. In addition, the data were compared with the resetting responses predicted for the intermittent light stimulus by a mathematical model of the effect
of light on the human circadian pacemaker (22), in which it
was presumed that any given duration of light exposure
exerts an intensity-dependent direct drive on the pacemaker,
independent of the duration of prior light exposure (30).
RESULTS
As previously reported at that phase of light administration (12), all light-exposed groups demonstrated
phase advances of their endogenous circadian rhythm
of core body temperature. The median phase-advance
shift observed was 2.87 h in the group of subjects
exposed to the intermittent 31% bright-light duty
schedule; 3.90 h in the group exposed to the intermittent 63% bright-light duty schedule; and 4.52 h in the
group exposed to the 100% continuous bright-light
stimulus, respectively (Fig. 3). A median phase delay of
0.97 h was observed in the 0% group of subjects
exposed to continuous darkness, consistent with the
slightly longer-than-24-h period of the endogenous circadian pacemaker (11, 16). Thus, compared with the
Fig. 3. Resetting responses of 31 individual subjects to the 0%
(continuous darkness), 31% (5-min bright light, 20-min darkness
duty cycle), 63% (46-min bright light, 44-min darkness duty
cycle), and 100% (continuous bright light) conditions. Phase-advance
shifts are plotted as positive values, whereas phase-delay shifts are
plotted as negative values. Horizontal bars represent the median
phase shifts observed in each condition. For clarity, resetting responses of similar magnitude have been slightly offset horizontally.
Data from the continuous darkness and continuous bright-light
groups from Duffy et al. (16).
INTERMITTENT LIGHT RESETS THE HUMAN CIRCADIAN PACEMAKER
control 0% condition (darkness), the 31% intermittent
bright-light duty schedule, the 63% intermittent
bright-light duty schedule, and the 100% continuous
bright-light stimulus duty schedule produced median
phase shifts of 3.84, 4.87, and 5.51 h (individual
phase shifts are reported in Table 1). The planned
ANOVA revealed both a significant linear trend (P
0.0001) and a significant quadratic trend (P 0.0104),
indicating that there was a significant nonlinear relationship between the relative duration of the brightlight exposure and the resetting response. The post hoc
ANOVA for pairwise comparisons using Tukeys
method revealed that 1) the phase advance shifts in all
light-exposed groups were significantly different from
the change in phase observed for the 0% control group
of subjects exposed to darkness (P 0.01 in all cases);
2) the phase advances observed for the 63% intermittent bright-light group were not significantly different
from those observed in the group exposed to 100%
continuous bright light; and 3) the phase-advances
observed for the 31% intermittent bright-light group
were significantly less than from those of the 100%
continuous bright-light group (P 0.05) but were not
significantly different from the 63% bright-light group.
Figure 4 illustrates how these resetting responses
compare with those predicted for the intermittent light
stimulus by the direct drive model of the effect of light
on the human circadian pacemaker (22).
R1577
Fig. 4. Fractional median resetting responses of the circadian phaseshifting effect in response to 3 cycles of exposure to 2 different
regimen of 5 h intermittent bright-light stimuli on the output of the
endogenous circadian pacemaker. The relative duration of brightlight exposure for each nominal 5-h stimulus represents the percentage of the 5-h episode that subjects were exposed to bright light in
each of the intermittent light groups. The response fractions represent the ratios of the resetting responses observed in the endogenous
circadian rhythm of core body temperature in each of the intermittent light groups (solid black bars) to the resetting response of
subjects in the continuous light group. The response fractions predicted by the direct-drive model of the effect of the bright light on the
endogenous circadian pacemaker for the intermittent light conditions are plotted in open bars (22).
DISCUSSION
This is the first laboratory-based study designed to
quantify the temporal dynamics of circadian phase
resetting by intermittent bright light in humans. Our
findings demonstrate that the response of the human
Table 1. Individual and mean phase shifts of the
endogenous circadian temperature rhythm
in the intermittent light groups
Subject Code
Phase Shift, h
63% Bright-Light Group
1341
1343
1347
1324
1328
1329
1321
1338
Mean
SE
3.55
3.51
0.07
3.57
4.67
5.14
5.72
4.22
3.81
0.07
31% Bright-Light Group
1251
1384
14a4
1502
1506
1517
1518
1521
Mean
SE
2.87
1.06
3.15
0.36
0.27
4.19
2.86
4.79
2.44
0.65
circadian pacemaker to short-duration light pulses is
not different from those observed in other organisms
(35, 37, 42) and necessitate the refinement of previous
models describing human sensitivity to light.
Our results suggest that an intermittent bright-light
stimulus, interrupted by intervals of complete darkness that substantially exceed the duration of the light
exposure, can significantly phase shift the human circadian pacemaker. Surprisingly, when the bright-light
exposure occupied only 31% of the stimulus, 70% of the
median resetting response was preserved compared
with the continuous stimulus. Furthermore, when the
bright-light exposure occupied only 63% of the stimulus, nearly 90% of the median resetting response was
preserved compared with the continuous stimulus (Fig.
4). The results from the latter group are especially
revealing. Although 37% of light was eliminated, only a
10% price was paid in the phase-shifting response.
Such results are inconsistent with the direct-drive
model that we had proposed, in which the effectiveness
of bright light in phase shifting the endogenous circadian pacemaker was homogenous throughout the duration of the stimulus.
We have thus now proposed two alternative modifications to the direct-drive model to reconcile these new
findings. The first alternative is the maintained-drive
model, which embodies two rate constants, representing the turn-on process (when the lights are turned
on) and representing the persistence after the turnoff process (when the lights are turned off) (28, 29).
This model postulates that light produces a drive on
R1578
INTERMITTENT LIGHT RESETS THE HUMAN CIRCADIAN PACEMAKER
the endogenous pacemaker that persists or is maintained for a period of time after it is turned off, decaying only gradually with an extended time course. Similarly, when a light stimulus is initiated, a period of
time elapses before full phase-shifting strength is attained. The second alternative, the dynamic-stimulus
model, postulates that during dark episodes, a potential for photic drive is stored and that this potential is
tapped at the start of light exposure with rate constant
and is reconstituted (restored) with a rate constant
(28, 29). During an extended light episode, the phaseshifting drive potential declines (with a time constant
of 20 min) to a low asymptotic level (/) corresponding to a balance between utilization and reconstitution. That is, the drive rate onto the circadian
pacemaker is limited by the restoration process. Thus,
when light is turned off, the phase-shifting drive stops
completely, but the restoration of drive potential continues at the rate, thereby priming the system for the
next light episode.
We have shown mathematically (28, 29) that the
temporal pattern of drive for the maintained-drive
model can be determined by passing the drive pattern
from the dynamic-stimulus model through a leaky
integrator of time constant 1. Thus, in the sense of
cumulative drive, the two new models are equivalent,
but important distinctions remain. Compared with the
dynamic-stimulus temporal-drive pattern, the pattern
of the maintained-drive model is significantly delayed
by the time 1 2.2 h, too long to account for the
initial strong phase-shifting response presently observed (29). Because the dynamic-stimulus model is
more consistent with the prompt biochemical responses (Fos protein) (20) to light observed in mammalian suprachiasmatic nuclei, it is the one we adopted.
The current results, as anticipated by the dynamicstimulus model, demonstrate that the initial strength
of the photic drive produced by the bright-light stimulus is very strong after several hours of dim light
exposure. As the bright-light exposure is maintained,
the drive strength declines (with a time constant of
20 min) to an asymptotic level of only about one-sixth
of its initial value. Thus the initial strength of the drive
after several hours of dim light is approximately six
times the strength of the drive after 1 h of bright-light
exposure (28, 29). These results suggest that, as in the
golden hamster and mosquito, there is a strong phaseresetting response to the initial minutes of a light
stimulus followed by a much weaker continuing response as the light duration is increased. This concept
is supported by both Brainard et al. (5), who demonstrated that intermittent bright light, which is conveyed to the pineal gland through the circadian pacemaker (25, 31), is capable of inducing significant
suppression of plasma melatonin levels in humans and
by Baehr et al. (1), who in a lab/field study reported
significant phase-delay shifts in groups exposed to intermittent bright light during simulated night shifts
compared with those exposed to dim light.
Perspectives
Our findings demonstrate that humans are much
more sensitive to the phase-resetting effects of brief
intermittent exposures to light than was previously
recognized (26, 43) and reinforce the concept that the
mechanisms by which human circadian rhythms are
entrained by light are not qualitatively different from
those of other mammals (14). Our study provides evidence that intermittent light exposure, even to as little
as repeated 5-min intervals of bright light, can produce
circadian resetting effects nearly as great as those
observed with a continuous 5-h bright-light exposure.
These findings, together with the recognition that humans are sensitive to the phase-resetting effects of
light throughout the subjective day (24), indicate that
the brief intermittent exposures to bright light that we
typically experience in everyday life (21, 36, 39) may
have a much greater impact on circadian entrainment
than was previously recognized (19, 26, 36, 39, 43).
Given that photic resetting responses can also be
achieved in humans at much lower light intensities (3,
45), these data suggest that sporadic nocturnal light
exposure, such as that which occurs to a wide variety of
people ranging from hospitalized patients to astronauts orbiting the Earth, may have more of an adverse
circadian phase-shifting effect than previously anticipated. These data also have practical implications and
suggest that a less-restrictive bright-light treatment
regimen can be used to reset the circadian pacemaker
in populations such as night workers or transmeridian
travelers and to treat patients with seasonal affective
disorders or circadian rhythm sleep disorders.
The authors acknowledge the contribution of B. Vu to the initial
protocol development, implementation, and supervision of studies in
the 63% intermittent light condition. We also thank the subject
volunteers, research technicians, dieticians, and nurses at the General Clinical Research Center; J. Kao, K. Foote, and G. Jayne for
subject recruitment and technical supervision; L. Rosenthal for illustrations; S. Hurwitz for consultation on the statistical analyses;
and D.-J. Dijk for critical review of the manuscript.
This research was supported in part by Grants NAGW-4033 and
NAG53952 from the National Aeronautics and Space Administration, 1-R01-AG06072 from the National Institute on Aging, 1-R01MH-45130 from the National Institute of Mental Health, and
F49620940398 from the Air Force Office of Scientific Research.
These studies were performed in a General Clinical Research Center
supported by National Center for Research Resources Grant M01RR-02635.
This work was presented, in part, at the Second International
Congress of the World Federation of Sleep Research Societies, Nassau, Bahamas 1995.
Present address of D. W. Rimmer: Dept. of Kinesiology, Pennsylvania State University, University Park, PA 16802; present address
for D. B. Boivin: Douglas Hospital Research Centre, Verdun, Quebec
H4H 1R3, Canada.
REFERENCES
1. Baehr EK, Fogg LF, and Eastman CI. Intermittent bright
light and exercise to entrain human circadian rhythms to night
work. Am J Physiol Regulatory Integrative Comp Physiol 277:
R1598R1604, 1999.
2. Boivin DB, Duffy JF, Kronauer RE, and Czeisler CA. Sensitivity of the human circadian pacemaker to moderately bright
light. J Biol Rhythms 9: 315331, 1994.
INTERMITTENT LIGHT RESETS THE HUMAN CIRCADIAN PACEMAKER
3. Boivin DB, Duffy JF, Kronauer RE, and Czeisler CA. Doseresponse relationships for resetting of human circadian clock by
light. Nature 379: 540542, 1996.
4. Boulos Z, Campbell SS, Lewy AJ, Terman M, Dijk D-J, and
Eastman CI. Light treatment for sleep disorders: consensus
report. VII. Jet lag. J Biol Rhythms 10: 167176, 1995.
5. Brainard GC, Baumholtz MA, Hanifin JP, and Rollag MD.
Constant versus intermittent ocular light exposure for melatonin
regulation in humans: is there temporal summation of photic
stimuli (Abstract)? Soc Res Biol Rhythms 6: 236, 1998.
6. Brown EN and Czeisler CA. The statistical analysis of circadian phase and amplitude in constant-routine core-temperature
data. J Biol Rhythms 7: 177202, 1992.
7. Campbell SS, Terman M, Lewy AJ, Dijk D-J, Eastman CI,
and Boulos Z. Light treatment for sleep disorders: consensus
report. V. Age-related disturbances. J Biol Rhythms 10: 151
154, 1995.
8. Czeisler CA. The effect of light on the human circadian pacemaker. In: Circadian Clocks and Their Adjustment, edited by
Waterhouse JM. Chichester: Wiley, 1995, p. 254302.
9. Czeisler CA, Allan JS, Strogatz SH, Ronda JM, Sanchez R,
Ros CD, Freitag WO, Richardson GS, and Kronauer RE.
Bright light resets the human circadian pacemaker independent
of the timing of the sleep-wake cycle. Science 233: 667671, 1986.
10. Czeisler CA, Johnson, MP, Duffy JF, Brown EN, Ronda
JM, and Kronauer RE. Exposure to bright light and darkness
to treat physiologic maladaptation to night work. N Engl J Med
322: 12531259, 1990.
11. Czeisler CA, Duffy JF, Shanahan TL, Brown EN, Mitchell
JF, Rimmer DW, Ronda JM, Silva EJ, Allan JS, Emens JS,
Dijk D-J, and Kronauer RE. Stability, precision, and near-24hour period of the human circadian pacemaker. Science 284:
21772181, 1999.
12. Czeisler CA, Kronauer RE, Allan JS, Duffy JF, Jewett ME,
Brown EN, and Ronda JM. Bright light induction of strong
(type 0) resetting of the human circadian pacemaker. Science
244: 13281333, 1989.
13. Czeisler CA, Kronauer RE, Johnson MP, Allan JS, Johnson TS, and Dumont M. Action of light on the human circadian
pacemaker: treatment of patients with circadian rhythm sleep
disorders. In: Sleep 88, edited by Horne J. Stuttgart, Germany:
Gustav Fischer Verlag, 1989, p. 4247.
14. Czeisler CA and Wright KP Jr. Influence of light on circadian
rhythmicity in humans. In: Regulation of Sleep and Circadian
Rhythms, edited by Turek FW and Zee PC. New York: Dekker,
1999, p. 149180.
15. Duffy JF. Constant Routine In: Encyclopedia of Sleep and
Dreaming, edited by Carskadon MA. New York: Macmillan,
1993, p. 134136.
16. Duffy JF, Kronauer RE, and Czeisler CA. Phase-shifting
human circadian rhythms: influence of sleep timing, social contact and light exposure. J Physiol (Lond) 495.1: 289297, 1996.
17. Eastman CI, Boulos Z, Terman M, Campbell SS, Dijk D-J,
and Lewy AJ. Light treatment for sleep disorders: consensus
report. VI. Shift work. J Biol Rhythms 10: 157164, 1995.
18. Eastman CI and Martin SK. How to use light and dark to
produce circadian adaptation to night shift work. Ann Med 31:
8798, 1999.
19. Espiritu RC, Kripke DF, Ancoli-Israel S, Mowen MA, Mason WJ, Fell RL, Klauber MR, and Kaplan OJ. Low illumination experienced by San Diego adults: association with atypical depressive symptoms. Biol Psychiatry 35: 403407, 1994.
20. Hastings MH, Best JD, Ebling FJP, Maywood ES, McNulty
S, Schurov I, Selvage D, Sloper P, and Smith KL. Entrainment of the circadian clock. In: Progress in Brain Research,
edited by Buijs RM, Kalsbeek A, Romijn HJ, Pennartz CMA, and
Mirmiran M. Amsterdam: Elsevier Science, 1996, p. 147174.
21. Hebert M, Dumont M, and Paquet J. Seasonal and diurnal
patterns of human illumination under natural conditions. Chronobiol Int 15: 5970, 1998.
22. Jewett ME and Kronauer RE. Refinement of a limit cycle
oscillator model of the effects of light on the human circadian
pacemaker. J Theor Biol 192: 455465, 1998.
R1579
23. Jewett ME, Kronauer RE, and Czeisler CA. Light-induced
suppression of endogenous circadian amplitude in humans. Nature 350: 5962, 1991.
24. Jewett ME, Rimmer DW, Duffy JF, Klerman EB, Kronauer
RE, and Czeisler CA. Human circadian pacemaker is sensitive
to light throughout subjective day without evidence of transients. Am J Physiol Regulatory Integrative Comp Physiol 273:
R1800R1809, 1997.
25. Klein DC, Moore RY, and Reppert SM. Suprachiasmatic Nucleus: The Minds Clock. New York: Oxford University Press, 1991.
26. Kripke DF and Gregg LW. Circadian effects of varying environmental light. In: Medical Monitoring in the Home and Work
Environment, edited by Miles LE and Broughton RJ. New York:
Raven, 1990, p. 187195.
27. Kronauer RE and Czeisler CA. Understanding the use of
light to control the circadian pacemaker in humans. In: Light
and Biological Rhythms in Man, edited by Wetterberg L. Oxford,
UK: Pergamon, 1993, p. 217236.
28. Kronauer RE, Forger D, and Jewett ME. Quantifying human
circadian pacemaker response to brief, extended, and repeated light
stimuli over the photopic range. J Biol Rhythms 14: 500515, 1999.
29. Kronauer RE, Forger DB, and Jewett ME. Erratum: quantifying human circadian pacemaker response to brief, extended,
and repeated light stimuli over the photopic range. J Biol
Rhythms 15: 184186, 2000.
30. Kronauer RE, Jewett ME, and Czeisler CA. Modeling human circadian phase and amplitude resetting. In: Biological
Clocks, Mechanisms and Applications, edited by Touitou Y. Amsterdam: Elsevier Science, 1998, p. 6372.
31. Li Y, Jiang DH, Wang ML, Jiao DR, and Pang SF. Rhythms of
serum melatonin in patients with spinal lesions at the cervical,
thoracic or lumbar region. Clin Endocrinol (Oxf) 30: 4756, 1989.
32. Mills JN, Minors DS, and Waterhouse JM. Adaptation to
abrupt time shifts of the oscillator[s] controlling human circadian rhythms. J Physiol (Lond) 285: 455470, 1978a.
33. Mills JN, Minors DS, and Waterhouse JM. The effect of sleep
upon human circadian rhythms. Chronobiologia 5: 1427, 1978b.
34. Minors DS, Waterhouse JM, and Wirz-Justice A. A human
phase-response curve to light. Neurosci Lett 133: 3640, 1991.
35. Nelson DE and Takahashi JS. Sensitivity and integration in
a visual pathway for circadian entrainment in the hamster
(Mesocricetus auratus). J Physiol (Lond) 439: 115145, 1991.
36. Okudaira N, Kripke DF, and Webster JB. Naturalistic studies of human light exposure. Am J Physiol Regulatory Integrative
Comp Physiol 245: R613R615, 1983.
37. Peterson EL. A limit cycle interpretation of a mosquito circadian oscillator. J Theor Biol 84: 281310, 1980.
38. Rosenthal NE, Joseph-Vanderpool JR, Levendosky AA,
Johnston SH, Allen R, Kelly KA, Souetre E, Schultz PM, and
Starz KE. Phase-shifting effects of bright morning light as treatment for delayed sleep phase syndrome. Sleep 13: 354361, 1990.
39. Savides TJ, Messin S, Senger C, and Kripke DF. Natural
light exposure of young adults. Physiol Behav 38: 571574, 1986.
40. Terman M, Lewy AJ, Dijk D-J, Boulos Z, Eastman CI, and
Campbell SS. Light treatment for sleep disorders: consensus
report. IV. Sleep phase and duration disturbances. J Biol
Rhythms 10: 135147, 1995.
41. Van Cauter E, Sturis J, Byrne MM, Blackman JD, Leproult R, Ofek G, LHermite-Baleriaux M, Refetoff S, Turek
FW, and Van Reeth O. Demonstration of rapid light-induced
advances and delays of the human circadian clock using hormonal phase markers. Am J Physiol Endocrinol Metab 266:
E953E963, 1994.
42. Van den Pol AN, Cao V, and Heller HC. Circadian system of
mice integrates brief light stimuli. Am J Physiol Regulatory
Integrative Comp Physiol 275: R654R657, 1998.
43. Wever RA, Polasek J, and Wildgruber CM. Bright light affects
human circadian rhythms. Pflugers Arch 396: 8587, 1983.
44. Wilmore DW. The Metabolic Management of the Critically Ill.
New York: Plenum, 1977.
45. Zeitzer JM, Dijk D-J, Kronauer RE, Brown EN, and
Czeisler CA. Sensitivity of the human circadian pacemaker to
nocturnal light: melatonin phase resetting and suppression.
J Physiol (Lond). 526: 695702, 2000.
Potrebbero piacerti anche
- A Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryDa EverandA Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryValutazione: 3.5 su 5 stelle3.5/5 (231)
- The Sympathizer: A Novel (Pulitzer Prize for Fiction)Da EverandThe Sympathizer: A Novel (Pulitzer Prize for Fiction)Valutazione: 4.5 su 5 stelle4.5/5 (119)
- Never Split the Difference: Negotiating As If Your Life Depended On ItDa EverandNever Split the Difference: Negotiating As If Your Life Depended On ItValutazione: 4.5 su 5 stelle4.5/5 (838)
- Devil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaDa EverandDevil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaValutazione: 4.5 su 5 stelle4.5/5 (265)
- The Little Book of Hygge: Danish Secrets to Happy LivingDa EverandThe Little Book of Hygge: Danish Secrets to Happy LivingValutazione: 3.5 su 5 stelle3.5/5 (399)
- Grit: The Power of Passion and PerseveranceDa EverandGrit: The Power of Passion and PerseveranceValutazione: 4 su 5 stelle4/5 (587)
- The World Is Flat 3.0: A Brief History of the Twenty-first CenturyDa EverandThe World Is Flat 3.0: A Brief History of the Twenty-first CenturyValutazione: 3.5 su 5 stelle3.5/5 (2219)
- The Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeDa EverandThe Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeValutazione: 4 su 5 stelle4/5 (5794)
- Team of Rivals: The Political Genius of Abraham LincolnDa EverandTeam of Rivals: The Political Genius of Abraham LincolnValutazione: 4.5 su 5 stelle4.5/5 (234)
- Shoe Dog: A Memoir by the Creator of NikeDa EverandShoe Dog: A Memoir by the Creator of NikeValutazione: 4.5 su 5 stelle4.5/5 (537)
- The Emperor of All Maladies: A Biography of CancerDa EverandThe Emperor of All Maladies: A Biography of CancerValutazione: 4.5 su 5 stelle4.5/5 (271)
- The Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreDa EverandThe Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreValutazione: 4 su 5 stelle4/5 (1090)
- Her Body and Other Parties: StoriesDa EverandHer Body and Other Parties: StoriesValutazione: 4 su 5 stelle4/5 (821)
- The Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersDa EverandThe Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersValutazione: 4.5 su 5 stelle4.5/5 (344)
- Hidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceDa EverandHidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceValutazione: 4 su 5 stelle4/5 (894)
- Elon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureDa EverandElon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureValutazione: 4.5 su 5 stelle4.5/5 (474)
- Multi OrgasmDocumento29 pagineMulti Orgasm4gen_7100% (2)
- The Unwinding: An Inner History of the New AmericaDa EverandThe Unwinding: An Inner History of the New AmericaValutazione: 4 su 5 stelle4/5 (45)
- The Yellow House: A Memoir (2019 National Book Award Winner)Da EverandThe Yellow House: A Memoir (2019 National Book Award Winner)Valutazione: 4 su 5 stelle4/5 (98)
- On Fire: The (Burning) Case for a Green New DealDa EverandOn Fire: The (Burning) Case for a Green New DealValutazione: 4 su 5 stelle4/5 (73)
- Speech in ActionDocumento160 pagineSpeech in ActionSplashXNessuna valutazione finora
- Trailblazer 2019Documento75 pagineTrailblazer 2019Georgiana CiaraNessuna valutazione finora
- (TCM) Foods and ElementsDocumento25 pagine(TCM) Foods and ElementsKevin FerreiraNessuna valutazione finora
- Thinking About StrategyDocumento14 pagineThinking About StrategyMehdi FaizyNessuna valutazione finora
- Artifical Light Annotated Bibliography v3Documento7 pagineArtifical Light Annotated Bibliography v3api-308065432Nessuna valutazione finora
- Circadian Rhythms in Liver Metabolism and DiseaseDocumento10 pagineCircadian Rhythms in Liver Metabolism and Diseaseapi-308065432100% (1)
- Shah Peer Review FormDocumento2 pagineShah Peer Review Formapi-308065432Nessuna valutazione finora
- Shah CommentsDocumento14 pagineShah Commentsapi-308065432Nessuna valutazione finora
- Sleep Deficiency and Deprivation Leading To Cardiovascular DiseaseDocumento5 pagineSleep Deficiency and Deprivation Leading To Cardiovascular Diseaseapi-308065432Nessuna valutazione finora
- IntroductionDocumento7 pagineIntroductionapi-308065432Nessuna valutazione finora
- The Impact of Light From Computer Monitors On Melatonin Levels in College StudentsDocumento11 pagineThe Impact of Light From Computer Monitors On Melatonin Levels in College Studentsapi-308065432Nessuna valutazione finora
- OutlineDocumento2 pagineOutlineapi-308065432Nessuna valutazione finora
- The Mammalian Circadian Timing System - Synchronization of Peripheral ClocksDocumento9 pagineThe Mammalian Circadian Timing System - Synchronization of Peripheral Clocksapi-308065432Nessuna valutazione finora
- Artifical Light Rough DraftDocumento3 pagineArtifical Light Rough Draftapi-308065432Nessuna valutazione finora
- Uncovering Residual Effects of Chronic Sleep Loss On Human PerformanceDocumento16 pagineUncovering Residual Effects of Chronic Sleep Loss On Human Performanceapi-308065432Nessuna valutazione finora
- Maternal Sleep Deprivation at Different Stages of Pregnancy Impairs The Emotional and Cognitive Functions and Suppresses Hippocampal Long-Term Potentiation in The Offspring RatsDocumento10 pagineMaternal Sleep Deprivation at Different Stages of Pregnancy Impairs The Emotional and Cognitive Functions and Suppresses Hippocampal Long-Term Potentiation in The Offspring Ratsapi-308065432Nessuna valutazione finora
- High Sensitivity of The Human Circadian Melatonin Rhythm To Resetting by Short Wavelength LightDocumento4 pagineHigh Sensitivity of The Human Circadian Melatonin Rhythm To Resetting by Short Wavelength Lightapi-308065432Nessuna valutazione finora
- Artifical Light Rough DraftDocumento13 pagineArtifical Light Rough Draftapi-308065432Nessuna valutazione finora
- Artifical Light Annotated BibliographyDocumento5 pagineArtifical Light Annotated Bibliographyapi-308065432Nessuna valutazione finora
- Protecting The Melatonin Rhythm Through Circadian Healthy Light ExposureDocumento53 pagineProtecting The Melatonin Rhythm Through Circadian Healthy Light Exposureapi-308065432Nessuna valutazione finora
- Breast Cancer and Circadian Disruption From Electric Lighting in The Modern WorldDocumento22 pagineBreast Cancer and Circadian Disruption From Electric Lighting in The Modern Worldapi-308065432Nessuna valutazione finora
- Lighting For The Human Circadian Clock - Recent Research Indicates That Lighting Has Become A Public Health IssueDocumento9 pagineLighting For The Human Circadian Clock - Recent Research Indicates That Lighting Has Become A Public Health Issueapi-308065432Nessuna valutazione finora
- Circadian Rhythms and Risk For Substance Use Disorders in AdolescenceDocumento13 pagineCircadian Rhythms and Risk For Substance Use Disorders in Adolescenceapi-308065432Nessuna valutazione finora
- The Role of Retinal Photoreceptors in The Regulation of Circadian RhythmsDocumento13 pagineThe Role of Retinal Photoreceptors in The Regulation of Circadian Rhythmsapi-308065432Nessuna valutazione finora
- OutlineDocumento2 pagineOutlineapi-308065432Nessuna valutazione finora
- Primary Paper PresentationDocumento7 paginePrimary Paper Presentationapi-308065432Nessuna valutazione finora
- Tbi Neurodegenerative Disease Annotated BibliographyDocumento2 pagineTbi Neurodegenerative Disease Annotated Bibliographyapi-308065432Nessuna valutazione finora
- Artifical Light Annotated BibliographyDocumento2 pagineArtifical Light Annotated Bibliographyapi-308065432Nessuna valutazione finora
- Sensitivity of The Human Circadian Pacemaker To Nocturnal Light - Melatonin Phase Resetting and SuppressionDocumento8 pagineSensitivity of The Human Circadian Pacemaker To Nocturnal Light - Melatonin Phase Resetting and Suppressionapi-308065432Nessuna valutazione finora
- Exposure To Room Light Before Bedtime Suppresses Melatonin Onset and Shortens Melatonin Duration in HumansDocumento10 pagineExposure To Room Light Before Bedtime Suppresses Melatonin Onset and Shortens Melatonin Duration in Humansapi-308065432Nessuna valutazione finora
- Histology AssignmentDocumento4 pagineHistology AssignmentNico LokoNessuna valutazione finora
- The Wolf and The LambDocumento4 pagineThe Wolf and The LambRaff Habibie RizzkhanbjmNessuna valutazione finora
- Science Level 4 - Animal CardsDocumento2 pagineScience Level 4 - Animal CardsClaudiaCarolinadeZeledónNessuna valutazione finora
- Kaylee Howlett ResumeDocumento2 pagineKaylee Howlett Resumeapi-353201001Nessuna valutazione finora
- Asexual Reproduction Worksheet: Name: . DateDocumento3 pagineAsexual Reproduction Worksheet: Name: . DateTamondong PaulNessuna valutazione finora
- NG TubeDocumento3 pagineNG TubeKenny JosefNessuna valutazione finora
- Dani's Odyssey or Some Commotion in The Hinterland FogDocumento228 pagineDani's Odyssey or Some Commotion in The Hinterland FogHanged Man PublishingNessuna valutazione finora
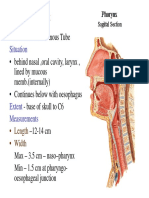
- H & N - Pharynx& Deglutition PDFDocumento17 pagineH & N - Pharynx& Deglutition PDFRamin AmirkhiziNessuna valutazione finora
- Plitvice Lakes, CroatiaDocumento7 paginePlitvice Lakes, CroatiaMirkoNessuna valutazione finora
- Imo 1.13 Elemelementary First AidDocumento22 pagineImo 1.13 Elemelementary First AidMuhammad TaufiqNessuna valutazione finora
- A Detailed Lesson Plan in Partial Fulfillment of The Course Requirements in (PED8)Documento11 pagineA Detailed Lesson Plan in Partial Fulfillment of The Course Requirements in (PED8)Angel Clarence BasilioNessuna valutazione finora
- Idoneth Deepkin Akhelian Thrallmaster v2Documento10 pagineIdoneth Deepkin Akhelian Thrallmaster v2Cerdeira HansNessuna valutazione finora
- Institute nephro-urology rate listDocumento9 pagineInstitute nephro-urology rate listVertika VermaNessuna valutazione finora
- Spiral FlapDocumento20 pagineSpiral FlapDiolanda De AbreuNessuna valutazione finora
- Food ChainsDocumento14 pagineFood ChainsShemaiah HoyteNessuna valutazione finora
- Catclass NotesDocumento3 pagineCatclass NotesIssac CaldwellNessuna valutazione finora
- ErizoDocumento36 pagineErizoEloy De Jesús SalazarNessuna valutazione finora
- Medical Parasitology: Human ParasitesDocumento30 pagineMedical Parasitology: Human ParasitesElycaNessuna valutazione finora
- Crochet Plush Bunny Neon Amigurumi Free PatternDocumento4 pagineCrochet Plush Bunny Neon Amigurumi Free PatternflechedestructriceNessuna valutazione finora
- Economically Feasible Fish Feed For GIFT Tilapia (Oreochromis LankaDocumento31 pagineEconomically Feasible Fish Feed For GIFT Tilapia (Oreochromis Lankasantosh kumarNessuna valutazione finora
- Cryo Fructo Qual KitDocumento3 pagineCryo Fructo Qual KitSuryakant HayatnagarkarNessuna valutazione finora
- Ex Situ Conservation: R.R.Bharathi 2 Yr BiochemistryDocumento20 pagineEx Situ Conservation: R.R.Bharathi 2 Yr BiochemistryVishal TakerNessuna valutazione finora
- The Market Square Dog: Name: ClassDocumento8 pagineThe Market Square Dog: Name: ClassAlejandra GonzálezNessuna valutazione finora
- Advanced Ocean Animals Activity BookDocumento5 pagineAdvanced Ocean Animals Activity BookRetno MaharaniNessuna valutazione finora
- Soal Vocabulary Part 2Documento3 pagineSoal Vocabulary Part 2Sastrawan PelawiNessuna valutazione finora