Documenti di Didattica
Documenti di Professioni
Documenti di Cultura
DC
Caricato da
Darran Cairns0 valutazioniIl 0% ha trovato utile questo documento (0 voti)
513 visualizzazioni3 pagineTitolo originale
dc
Copyright
© Attribution Non-Commercial (BY-NC)
Formati disponibili
PDF o leggi online da Scribd
Condividi questo documento
Condividi o incorpora il documento
Hai trovato utile questo documento?
Questo contenuto è inappropriato?
Segnala questo documentoCopyright:
Attribution Non-Commercial (BY-NC)
Formati disponibili
Scarica in formato PDF o leggi online su Scribd
0 valutazioniIl 0% ha trovato utile questo documento (0 voti)
513 visualizzazioni3 pagineDC
Caricato da
Darran CairnsCopyright:
Attribution Non-Commercial (BY-NC)
Formati disponibili
Scarica in formato PDF o leggi online su Scribd
Sei sulla pagina 1di 3
6
Q3__ A Binary Peritectic Alloy of iridium and osmium of average composition 90 wt %
Osmium is melted to 3000° C. What phases are present at (a) 2600° C, (b)
2665+A T° C and (c) 2665-A T° C. What are the compositions of each phase at
these temperatures? What is the amount of each phase? Identify any three phase
invariant reactions with composition and Temperature.
Atomie percentage osmium
r 10 20 «30 40SssCiaSCSC«CSCs
Weight percentage osmium s) s
. eg —CeS) 1
famarat- 5 epee: \ 60 = as
@ ré00' (42% Coeurrm') auETaT
eee B (6157 0mm) tewrt
wat = LS 2 8%
@ 265 -AT 2(A3e AS og
@ (615%) Amont= AD %
fant = 6628 = 4%
@ 2065+ AT L (23%) BIS -23
iano on GO-23 = 4th,
g (615%) Ausount S33 i:
yy
wt%
Q4 A Binary Eutectic Alloy of copper and silver of average composition 80% is
melted to 1000° C. What phases are present at (a) 1000° C, (b) 800° C and (c)
600° C. What are the compositions of each phase at these temperatures? What is
the amount of each phase?
7 Atomie percent silver on
c lo 20 30 eae 10 ees 308160 e700 es Olney
1100 T T T T T T T T
1084.5°
+ 1900
1000
1700
900
7 1600
800
| 1400,
700
1200
600
1000
500
850
400. 1 1 i i 1 L L 1 1
Cu
@ 006 ce
@ oc
10) os 206 oa 30 ee eee 50 ete CO) ett 70 me sO OO Ag
Weight percent silver ==>
Compaitin — 80% Aq Amount 1S [002% ligued
. 42-80) .w < 12 . 31%
L campitin — FS% Ag — x 2 oF
B composition 42, a #35 piv = 5 = 20%
42-46 ze
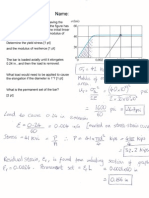
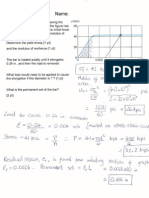
Q2 From the steel phase diagram below: (a) a 3% C Austenite Steel is cooled slowly
from 1000° C toa temperature of 800° C determine the weigh percent austenite in the
steel. (b) What structural change occurs in iron at 723° C. (c) Define the following,
phases in Steel (i) austenite, (ii) 0. Ferrite(iii) cementite,
Comentle
7 Feces = O26 GPEC
i
i wer y = (G6A3) 36
. eoF-l2 5-47
400) whey -
' ‘ = 62%
beth ==).
te (6-64 -[-2) 6
oo 7 ferrite
Pustente
) oo Ferrite
| & @ gg (ec ce a
oO youstentte FCC ren ty)
BCL
- . &C
a) conwibi te cS fe.
Potrebbero piacerti anche
- SKMBT 50110042115310Documento3 pagineSKMBT 50110042115310Darran CairnsNessuna valutazione finora
- SKMBT 50110043017390Documento2 pagineSKMBT 50110043017390Darran CairnsNessuna valutazione finora
- M EvlAA'L S P //.H-2 0 L 0Documento5 pagineM EvlAA'L S P //.H-2 0 L 0Darran CairnsNessuna valutazione finora
- Cairns 002Documento2 pagineCairns 002Darran CairnsNessuna valutazione finora
- DCDocumento1 paginaDCDarran CairnsNessuna valutazione finora
- DC 1Documento1 paginaDC 1Darran CairnsNessuna valutazione finora
- DC 1Documento1 paginaDC 1Darran CairnsNessuna valutazione finora
- DCDocumento2 pagineDCDarran CairnsNessuna valutazione finora
- DC 001Documento3 pagineDC 001Darran CairnsNessuna valutazione finora
- DCDocumento4 pagineDCDarran CairnsNessuna valutazione finora
- DC 23Documento2 pagineDC 23Darran CairnsNessuna valutazione finora
- DcweibullDocumento2 pagineDcweibullDarran CairnsNessuna valutazione finora
- CairnsDocumento3 pagineCairnsDarran CairnsNessuna valutazione finora
- Cairns 001Documento1 paginaCairns 001Darran CairnsNessuna valutazione finora
- Example 343 Midterm 1Documento6 pagineExample 343 Midterm 1Darran CairnsNessuna valutazione finora
- Example 343 Midterm 1Documento6 pagineExample 343 Midterm 1Darran CairnsNessuna valutazione finora
- MAE243 Spring 2009 Quiz 2BDocumento1 paginaMAE243 Spring 2009 Quiz 2BDarran CairnsNessuna valutazione finora
- MAE243 Spring 2009 Quiz 3Documento1 paginaMAE243 Spring 2009 Quiz 3Darran CairnsNessuna valutazione finora
- CairnsDocumento6 pagineCairnsDarran CairnsNessuna valutazione finora
- DcairnsDocumento2 pagineDcairnsDarran CairnsNessuna valutazione finora
- Fall 2006 Midterm 1 Q1Documento1 paginaFall 2006 Midterm 1 Q1Darran CairnsNessuna valutazione finora
- MAE243 Spring 2009 Quiz 2BDocumento1 paginaMAE243 Spring 2009 Quiz 2BDarran CairnsNessuna valutazione finora
- MAE243 Spring 2009 Quiz 2BDocumento1 paginaMAE243 Spring 2009 Quiz 2BDarran CairnsNessuna valutazione finora
- TorsionDocumento1 paginaTorsionDarran CairnsNessuna valutazione finora