Documenti di Didattica
Documenti di Professioni
Documenti di Cultura
Boiler Water Chemistry
Caricato da
Bhargav ChaudhariCopyright
Formati disponibili
Condividi questo documento
Condividi o incorpora il documento
Hai trovato utile questo documento?
Questo contenuto è inappropriato?
Segnala questo documentoCopyright:
Formati disponibili
Boiler Water Chemistry
Caricato da
Bhargav ChaudhariCopyright:
Formati disponibili
Boiler Water Management
Water Characteristics and Quality
Fundamental Mission of Boiler Power House Management
Reliable production of steam
continuous & quality steam (no unplanned outages)
Safe production of steam
no injuries. no loss of capital equipment Food safety: FDA. Kosher
Protect the capital investment
minimize corrosion and scaling
Environmental/Regulatory Compliance Cost effective operation
efficient as possible
Water characteristics and quality Characteristics
pH Hardness Conductivity Total Dissolved Solids Alkalinity p-alkalinity m-alkalinity Total Suspended Solids
important parameter (acidity. alkalinity) combination of Ca and Mg uS/cm (relationship with dissolved solids) amount of dissolved ions (mg/l) amount of buffering titration until pH=8.3 total alkalinity (pH=4.3) everything that is not dissolved and can be filtered out
Water characteristics and quality Impurities
Cations Sodium Potassium Calcium Magnesium Ammonium Iron Manganese
Na+ K+ Ca2+ Mg2+ NH4+ Fe2+ Mn2+
MINERALS
AIR
EARTH
Anions Bicarbonate Chloride Sulphate Nitrate Silicate
HCO3ClSO42NO3HSiO3-
Cations Anions
Oxygen Carbon Monoxide Carbon Dioxide
Clay Silt Sand
a. Dissolved solids
b. Dissolved gases
c. Suspended matter
Water characteristics and quality pH Scale
ACIDIC 1 2 3 4 5 6 7 pH Hydrogen Ions (H+) decrease pH; Hydroxyl (OH-) increase pH 8 9 BASIC 10 11 12 13 14
Water Characteristics and quality Three battles
Presence of troublesome water components requires
Deposition Control Preventing overheat failures resulting from waterside deposition Corrosion Control Maintaining materials of construction. dealing with general and localized corrosion mechanisms Steam Purity Preventing carryover of boiler water salts into steam phase
Deposition
Deposition What is
Result of insolubility of inorganic scale formers Hardness Based Deposits Metal Oxide Based Deposits Can occur as a result of contaminated condensate or feedwater components Unlikely with demineralized makeup water Good pretreatment may limit deposition potential Develops at areas of highest heat input Issue becomes more critical as boiler pressure goes up
Deposition Problems caused
Loss of Boiler Efficiency
Scale reduces heat transfer Increased fuel/energy consumption
Boiler Tube Failure
Scale elevates tube temperature - causing tube overheating Partial destroyed boiler sections and even boiler explosion due to overheating of tube metal over critical temperature for steel
Under-deposit Corrosion
Caused by high localized concentration of corrosive molecules
Deposition Efficiency reduction
20
Efficiency Loss in %
18 16 14 12 10 8
6
4 2 0.4 1.6 2.8 3.6 0.8 1.2 2.4 3.2 4.4 4.8 0 2
Deposit in mm
Water tube boiler, 95 % of Scale CaCO3. No heat recovery 20 bar [290 psi], 11.3 ton/h
Deposition Increased tube temperature
Boiler tube metal wall
Boiler tube Deposit metal wall
Boiler tube Deposit metal wall Increased wall T4
Wall T2
Wal T2 T3
Boiler water T1
Boiler water T1 needed Resulting boiler water T0
Boiler water T1
A Clean internal heat transfer surface. T1 for needed steam production
B Scaled surface resulting in lower T0 while needed T1
C Scaled surface Fire side T2 is increased to T4 to reach water T1 T3 is critical for metal structure
Deposition Effect of scale on heat transfer
120
240 200
Temperature [F]
115
Tube metal temperature increase
100
95
Temperature [C]
80
160 120 80 40 0 0 0.002 0.004 0.006 0.008 0.01 Deposit thickness [mm]
70
60
50
40
25
20
Deposition Factors
Factors that contribute in waterside deposition:
Presence of ionic scale formers in feedwater (calcium, magnesium, barium, strontium, silica, iron) Presence of scale formers treatment derived (phosphate, alkalinity) Presence of particulate contaminants Interactions with existing deposition Heat input Flow parameters Microclimates Location in riser circuit
Deposition Solubilities
Compound Calcium Bicarbonate Carbonate Sulfate Magnesium Bicarbonate Carbonate Sulfate Sodium Bicarbonate Carbonate Chloride Hydroxide Sulfate
ppm as CaCO3 0 C [32 F] 1620 15 1290 37.000 101 170.000 30.700 61.400 225.000 370.000 33.600 100 C [212 F] Decomposes 13 1250 Decomposes 75 356.000 Decomposes 290.000 243.000 970.000 210.000
Deposition Hardness precipitation
Precipitation of insoluble hardness 2 HCO3- CO32- + CO2+ H2O CO32- + H2O CO2 + 2OH Ca2+ + CO32- CaCO3
100% 60 - 80% boiler scale
Mg+2 + OH- MgOH+ H2SiO3 H+ + HSiO3 MgOH+ + HSiO3- MgSiO3 + H2O
Scaling & Deposits Heat transfer limitation Boiler tube failure
Exceeding saturation through evaporation, resulting in crystallization eg. CaSO4.SiO2
Deposition Why Hardness is Such an Issue
Soluble Amount
Most materials are more soluble at increasing temperature
Ca. Mg. Ba. Sr solubility decreases at increasing temperature
Temperature
Deposition Boiler deposits
Name Acmite Analcite Anhydrite Aragonite Basic magnesium phosphate Brucite Calcium hydroxide Calcite Copper Cuprite Ferrous oxide Goetnite Gypsum Hematite Hydroxyapatite Magnetite Serpentine (magnesium silicate) Sodium ferrous phosphate Tenorite Thenardite Xonotlite Formula Na2OFe2O34SiO2 Na2OAl2O34SiO22H2O CaSO4 CaCO3 (gamma form) Mg3(PO4)2Mg(OH)2 Mg(OH)2 Ca(OH)2 CaCO3 (beta form) Cu Cu2O FeO Fe2O3 H2O (alpha form) CaSO42 H2O Fe2O3 Ca10(PO4)6(OH)2 Fe3O4 3MgOSiO22 H2O NaFePO4 CuO Na2SO4 5 CaO5 SiO2 H2O
Deposition Silica
Forms deposits in boilers Occurs as magnesium silicate or silicic acid
Selective silica carryover Silica is selectively dissolved into the steam Controlled by limiting the silica concentration in the boiler water Controlled by limiting boiler pressure Controlled by maintaining high pH Cannot be controlled mechanically by steam separators Not usually a problem with boilers with less than 40 bar (600 psig) pressure
Deposition Iron scale
Iron is usually found in a boiler as one or more of the following:
A complex with calcium A complex with phosphate Hematite - Fe2O3 Magnetite - Fe3O4
Corrosion
Corrosion What is
Definition
The deterioration of a material by the interaction with its environment
Impact
Feedwater piping Feedwater heaters Economizer Boiler Condensate system
Oxygen corrosion in flame pipe (6 bar boiler)
Corrosion Problems caused
Impact corrosion on boiler reliability
Premature replacement need of boiler boilers should last very long upon correct use Water side corrosion should be nihil and not a factor contributing to the life time of the boiler Unscheduled maintenance to boiler or feed water lines Unscheduled maintenance to the condensate system Filthy steam and condensate traps can cause leaking of steam into condensate system due to malfunctioning valves
Oxygen corrosion in flame pipe (6 bar boiler)
Corrosion Boiler metal passivation
Natural passivation:
Formation of a protective barrier on metal surface by reaction with water Fe + 2 H2O Fe(OH)2 + H2 3Fe(OH)2 Fe3O4 + H2 + 2H2O Black magnetite film - Fe3O4 Rate of Magnetite formation is Temperature dependent Spontaneous above 180 C (360 F) Reduces general corrosion Difficult to quantify results
Oxygen scavenging Metal passivation
SHIKORR REACTION
T < 180 C
3 Fe + 6 H2O 3 Fe(OH)2 + 3 H2
2 Fe(OH)2 + 2 H2O
Fe(OH)2 + 2 Fe(OH)3
2 Fe(OH)3 + H2
Fe3O4 + 4 H2O
T > 180 C
3 Fe + 4 H2O Fe3O4 + 4 H2
Corrosion Solubility of magnetite in water
Corrosion Effect of pH
Do we have a better picture ?? Check advanced training series
Corrosion Types
Types of Corrosion
Oxygen corrosion Alkalinity concentration Caustic corrosion Acid corrosion Chelant corrosion Erosion/Corrosion
Condensate return pipe with deep attack due to the combined effect of CO2 and O2
Corrosion Oxygen Corrosion
Oxygen. O2
Hydroxide OH-
Corrosion products
Metal ions dissolve
CATHODE
Electron transport
ANODE
ANODE:
Fe0 Natural Metal 2eElectrons +
Fe+2 Metal ion +
2eMetal electrons 2(OH-) Charged Ion
CATHODE:
1/2 O2 Oxygen
H2O Water
Corrosion Factors Oxygen corrosion
Can be found throughout the system Mechanism same as other oxygen corrosion cells Corrosion mechanisms affected by: pH
Minimize corrosion: pH >9.0
Temperature
Higher temperatures reduce O2 solubility, but significantly increase corrosivity
Dissolved oxygen concentration
Higher O2 concentrations increase corrosivity
Fluid velocity
Enhances effect of other corrodents More diffusion - better mass transport
Corrosion Oxygen Corrosion
Dissolved oxygen as function of temperature at atmospherical pressure
5.5 ppm O2
2.5 ppm O2
50C (122 F)
85C (185 F)
Corrosion Oxygen Corrosion
Problems associated with oxygen:
Crack formation in degassers - deaerator cracking Corrosion pits that require immediate remedy to obtain approval after periodic inspections by authorities Corrosion damage to gray cast iron in feed water pumps Leaking feed water pipes, leaking economisers, pits and craters in boiler tubes (low pressure boilers)
Oxygen corrosion in feed water line
Corrosion Oxygen Corrosion
Economical impact of oxygen corrosion
Increased energy consumption Oxygen corrosion introduces additional iron and copper oxides in the water
Deposition on heated surfaces (limiting heat transfer)
Oxygen corrosion in feed water line (pH 7.5. 50 C)
Increased blowdown due to turbidity water Increased conductivity due to addition of oxygen scavengers (e.g. sulfite) requires increased blowdown
Corrosion Caustic Damage
There are two forms of damage caused by caustic soda to high pressure boilers, namely:
Do we have a picture Steef??
Caustic corrosion Caustic embrittlement
Corrosion Caustic Corrosion
Do we have a picture Steef??
Two conditions are necessary for caustic corrosion to occur: Presence of a corrosive material in the boiler water (caustic soda) Mechanism for concentrating this material, usually due to deposits Usually found only in high pressure boilers
Fe3O4 Fe2O3 + FeO (Magnetite dissolution) Fe2O3 + 2 NaOH 2 NaFeO2 + H2O (soluble) FeO + 2 NaOH Na2FeO2 + H2O (soluble)
Localized in boiler Also called crater attack or caustic gouging No embrittlement of metal
Corrosion Caustic Embrittlement
Steef, kan deze slide eruit? Zo nee, do we have a picture Steef?
Embrittlement is a special form of stress corrosion cracking
Should not be confused with caustic corrosion Three conditions must be present Concentrating mechanism present Metal under high stress Must contain silica Inhibited by improved fabrication techniques and by organic and nitrate-based inhibitors
Check last remark!! Nitrate??!?
Corrosion Concentration mechanism
Steam blanketing
Under deposit / concentration to dryness
Excessive heat input Insufficient coolant flow Deposits
Elongated gouge caused by steam blanketing
Magnetite NaOH
Steam Out NaOH NaOH NaOH Water In
Fe3O4 porous deposit
NaOH
Corrosion Concentrating Mechanisms
The following conditions can result in dangerously high localized caustic soda concentrations
(Porous) metal oxide deposits Operation above rated capacity Excessive rate of load increase Excessive localized heat input Localized pressure differentials Restrictions in generating tube(s)
Corrosion Acid corrosion
Causes of acidity
Feedwater acid contamination Acid leaks from demineralizer, e.g. acid regenerant Breakdown of organic materials Condenser leaks MgCl2 + 2 H2O Mg(OH)2 + 2 HCl Organic chlorides R-X + 2 H2O R-OH + HCl
Corrosion Acid corrosion
% Conversion
Special form of acid corrosion can be found in condensate system:
Alkalinity of BFW breaks down in Boiler
2 NaHCO3 Na2CO3 + CO2 + H2O Na2CO3 + H2O 2 NaOH + CO2
CO2 leaves with steam and reacts with condensed water droplets to Carbonic Acid
CO2 + H2O H2CO3
The H2CO3 acid attacks metals by reducing pH
Steam purity
Steam Purity What is
Defined as the amount of non-water components of the steam
Priming Foaming Misting Selective carry-over
Normally measured in terms of ppb sodium ion and ppb silica.
Sodium Limits to turbines Old industrial turbines - max. of 20 ppb New industrial turbines - max. 5 ppb Silica Limits to Turbines - 20 ppb
Steam Purity Problems caused
Effects of low steam purity
Process Contamination Loss of Turbine Efficiency Turbine Imbalance Operational Problems Plant Shut-down
Water Quality guidelines
Water quality guidelines
Drum-type boilers using softened (not deionized) feedwaters
Boiler Water component [ppm] Drum Pressure. psig (bar)
150 (10) 300 (20) 600 (40) 900 (60) 1200 (80) 1500 (100)
Check Etienne source???
TDS (max) Phosphate (as PO4) Hydroxide (as CaCO3)
4000 30-60 300-400
3500 30-60 250-300
3000 20-40 150-200
2000 15-20 120-150
500 10-15 100-120
300 5-10 80-100
Sulfite
30-60
30-40
20-30
15-20
Not recommende d
5 2 50-70
Not recommende d
3 1
Silica (as SiO2. max) Total Iron (as Fe. max) Organics
100 10 70-100
50 5 70-100
30 3 50-70
10 2 50-70
Different for all boiler types. feed water quality and region ASME: USA Krachtwerktuigen: Holland TUV: Germany British Standard: UK
Water quality guidelines High purity Feed water
Drum-type boilers using high purity (deionized) feedwaters
Boiler Water Component Drum pressure psig (bar)
Check Etienne source???
TDS (max) Phosphate (as PO4) pH Silica (as SiO2. max) Total iron (as Fe. max) Hydrazine
Upto 600 (40)
900 (60)
120 (80)
1500 (100)
1800 (120)
2400 (160)
500 15-25 9.8-10.2 10 2 Idem soft water
500 15-25 9.8-10.2 10 2 0.04-0.06
300 15-25 9.8-10.2 5 2 0.04-0.06
200 5-10 9.4-9.7 2 1 0.04-0.06
100 5-10 9.4-9.7 1 0.5 0.04-0.06
50 5-10 9.4-9.7 0.25 0.25 0.04-0.06
Different for all boiler types. feed water quality and region ASME: USA Krachtwerktuigen: Holland TUV: Germany British Standard: UK
Water quality guidelines general remarks
Limits on TDS will vary with the design of the boiler and with the needs of the system Despite TDS maximums. industrial type boilers (as opposed to utility type) should not be operated above 100 cycles of concentration Silica may be carried at higher levels if there are no condensing turbines in the cycle. In any case. maintain an "O"/SiOratio of at least 3/1 to inhibit silica deposition. Iron levels shown are theoretical levels based on feedwater iron multiplied by cycles of feed water concentration
ASME Guidelines for Feedwater Quality
Water tube
Boiler Pressure [bar] 0 - 20
Boiler Pressure [psi] 0 300 301 450 451 600 601 750 751 900 901 1500
Hardness [ppm] 0.300 0.300 0.200 0.200 0.100 N/D
Iron [ppm] 0.100 0.050 0.030 0.025 0.020 0.010
Copper [ppm] 0.050 0.025 0.020 0.020 0.015 0.010
Make again in PPT format
21 - 31 32 - 41 42 51 52 62 63 - 103
Fire tube 0 - 20 0 300 1.0 0.100 0.050
Feedwater Quality requirements France - Softened Feedwater
Operating pressure Feedwater pH Total Hardness Oxygen Huil
bar
< 15
15 - 25
25 - 35
35 45
8.5 [F] < 0.5 remove absent
8.5 < 0.2 remove absent
8.5 < 0.2 Remove absent
8.5 < 0.1 Remove Absent
Boiler water TAC TA Silica SiO2 / TAC Total dissoloved solids Chlorine Phosphate pH [Mg/L] [Mg/L] [Mg/L] [F] [F] [Mg/L] 100 0.7 TAC 200 2.5 < 4000 800 30 - 100 10.5 - 12 80 0.7 TAC 150 2 < 3000 600 30 - 100 10.5 - 12 60 0.7 TAC 90 1.5 < 2000 400 20 - 80 10.5 - 12 40 0.7 TAC 40 1 < 1500 300 20 80 10.5 - 12
Feedwater Quality requirements France - Demineralized Feedwater
Operating pressure Feedwater pH Total Hardness Oxygen Huil Iron Copper
bar
40 - 60
60 - 75
75 - 100
8.5 [F] < 0.05 remove Mg/l Mg/l Mg/L < 0.05 < 0.05 < 0.03
8.5 < 0.05 remove < 0.05 < 0.05 < 0.03
8.5 < 0.05 Remove < 0.05 < 0.03 < 0.01
Boiler water TAC TA Silica SiO2 / TAC Total dissoloved solids Free NaOH Phosphate pH [Mg/L] [Mg/L] [Mg/L] [F] [F] [Mg/L] 25 0.5 TAC 15 <1 < 500 < 20 10 - 60 10 - 11 10 0.5 TAC 10 <1 < 300 < 300 10 - 40 10 - 11 5 0.5 TAC 5 <1 < 100 < 100 5 20 9.5 10.5
Feedwater Quality requirements France Fire tube boilers
Operating pressure Feedwater pH Total Hardness Oxygen Huil
bar
10
10 - 15
15 25
8.5 [F] < 0.5 remove Mg/l absent
8.5 < 0.5 remove absent
8.5 < 0.2 Remove Absent
Boiler water TAC TA Silica SiO2 / TAC Total dissoloved solids [Mg/L] [F] [F] [Mg/L] 120 0.7 TAC 200 2.5 5000 100 0.7 TAC 200 2.5 4000 80 0.7 TAC 150 2 3000
Free NaOH
Phosphate pH
[Mg/L]
[Mg/L]
< 20
30 - 100 10.5 - 12
< 300
30 - 100 10.5 - 12
< 100
30 - 100 10.5 - 12
Feedwater Quality requirements Germany - VGB
Scope Continuous operation Fully demineralized feedwater Continuous operation Demineralized feedwater Alkalized boiler water Na3PO4 recommended Pressure area PH (25 C) Neutral operation Combined operation Alkaline operation Conductivity (25 C) [uS/cm] [bar] > 68 7-8 8-9 9-10 < 0.25 < 50 < 50 < 5 (for < 250 kW/m2) < 3 (for > 250 kW/m2) < 0.2 68 - 136 9.8 10.2 Continuous operation Demineralized feedwater Alkalized boiler water Na3PO4 recommended > 136 9.3 9.7 Alkaline Continuous operation Demineralized feedwater Volatile agents Continuous operation Steam for condensing turbines
Oxygen Neutral operation Combined operation Alkaline operation Iron Copper
[mg/l] 0.050 - 0.250 0.030 0.150 < 0.100 [mg/l] [mg/l] [mg/l] [mg/l] [mg/L] < 0.02 < 0.03 < 0.01 < 0.02 <6 <3 NA < 0.020 < 0.030 < 0.010 < 0.020
Na Silica If Na3PO4 is dosed PO4
Boiler water Quality requirements Germany - TV
Scope Pressure area Appearance Conductivity (25 C) Hardness Oxygen Phosphate PH (25 C) p-alkalinity CO2 fixed Iron Copper Silica Organic carbon Oil [ppm CaCO3] [mg/l ] ppm Ppm [mg/l] [ppm as KMnO4] ppm [S/cm] [ppm CaCO3] ppm [mg/l ] [bar]
Softened feedwater 1 <> 68
Boiler water on softened feedwater 1 <> 22 22 <> 44 44 <> 68
Demin. feedwater < 68
Boiler water based on demin. feedwater < 68 < 68
Colourless, free from suspended matter < 10,000 < 1.0 < 0.02 10 20 10.5 12 50 600 < 25 < 0.03 < 0.005 NR < 10 <1 Graph Graph < 10 < 0.03 < 0.005 < 0.02 <3 <1 <4 <4 5 - 15 10 11.8 25 - 300 5 - 15 10 11 5 50 >9 < 0.1 <6 9.5 10.5 >7 < 5,000 < 2,500 < 0.2 < 150 <3
Boiler water Quality requirements Italy
Scope Pressure area Appearance Conductivity (25 C) Hardness Oxygen Phosphate [S/cm] [ppm CaCO3] ppm [mg/l ] [bar]
PH (25 C)
p-alkalinity CO2 fixed Iron Copper Silica Organic carbon [ppm CaCO3] [mg/l ] ppm Ppm [mg/l] [ppm as KMnO4]
Oil
ppm
Boiler water Quality requirements Spain Firetube: UNE 9-075-92
Feedwater 0.5 bar Appearance Colourless and without suspended solids [ppm CaCO3] [mg/L] 10 8-9
Feedwater > 0.5 bar
Boiler water 0.5 bar
Boiler water 0.5 < 13 bar
Boiler water > 13 bar
Hardness Oxygen PH (20 C)
5 0.2 8-9 10.5 12.5 10 - 12 10 12
CO2 fixed
Organic carbon Oil Conductivity ( 40 kg/m2) Conductivity (> 40 kg/m2 p-alkalinity Phosphate Silica
[mg/l ]
[ppm as KMnO4] ppm [mg/L] [mg/L] [ppm CaCO3] [mg/l P2O5 ] [mg/l SiO2]
25
10 3
25
10 1 6,000 5,000 1,000 30 250 6,000 5,000 800 25 200 4,000 3,000 600 20 150
Boiler water Quality requirements Spain - water tube: UNE 9-075-92
Maximum allowed values
Hardness [mg/L]
Alkalinity [mg/L] 700 600 500 400 300 250 100 40 10 2
TSS [mg/L] 150 100 80 60 40 20 10
pH (20 C)
Phosphate [Mg/L P2O5]
Silica [mg/l] 140 50 50 50 10 10 4 4 4 4 4 140 50
< 20 21- 32 33 40 41 53 54 64 65 70 71 126 127 165 166 180 181 203 > 98 < 20 20 - 40
Natural circulation Natural circulation Natural circulation Natural circulation Natural circulation Natural circulation Natural circulation Natural circulation Natural circulation Natural circulation Forced circulation Forced circulation with continuous steam formation Forced circulation with continuous steam formation
3,500 3,000 2,500 2,000 1,500 1,250 100 50 25 15 0.05 2,000 1,000
9.5 - 11 9.5 - 11 9.5 - 11 9.5 - 11 9.5 - 11 9.5 - 11 9.5 10.5 9.5 10.5 9.5 10.5 9.5 10.5 9.5 10.5
25 25 10 10 10 3 3 3 3 3 3 -
400 200
100 50
Boiler water Quality requirements Netherlands
Parameters Krachtwerktuigen KEMA
Waterpijp
CHECK I do not have the original!
Low pressure boilers
<80 <15
Pressure area [bar] Phosphate [mg/l ] PH (25 C) p-alkalinity [ppm CaCO3]
0.5-20 30-80
9.8-10.2 250-750
Chloride [mg/l] Silica [mg/l]
<2 <5
TDS Conductivity [S/cm] Organic carbon [ppm CZV] Iso ascorbic acid [mg/l] < 6000 (pH=8.3) <150 <2500 (after cation filter)
Boiler water Quality requirements UK Shell tube upto 30 bar
Parameter
Unit
Feedwater
Boiler water
Boiler water Demineralized feedwater
Heat flux
300 Clear, no foam 8.5 9.5 Mg/kg CaCO3 Mg/kg Mg/kg CaCO3 Mg/kg CaCO3 Mg/kg Mg/kg 30 70 0.1 1.0 120 160 15 30 0.1 1.0 (FW) Mg/kg Mg/kg Mg/kg Mg/kg uS/cm 30 60 150 200 3500 7000 1 1000 350 2 10.5 12.0
> 300
CHECK Is there an update, have seen the draft!
Appearance pH (25 C) Total Hardness Oxygen Total M-alkalinity O- and P-alkalinity Oil and grease Oxygen scavenger Sulfite, or Hydrazine, or Tannin, or Iso ascorbic acid, or DEHA Phosphate Silica Suspended solids Dissolved solids Conductivity (25 C)
9.5 10.5
100 20
Assess 0.1 1.0 Assess Assess 0.1 1.0 (FW) 10 30 5 20 1000 2000
Boiler water Quality requirements UK Fired Water tube
CHECK - Is there an update, have seen the draft!
Parameter Unit 0 - 20 21 40 41 - 60 61 - 80 81 - 100 101 - 120 121 Feedwater pH (25 C) Total Hardness Oxygen Fe, Cu, and Ni Mg/kg CaCO3 Mg/kg Mg/kg 8.5 9.5 2 0.02 0.05 8.5 9.5 1 0.02 0.05 8.5 9.5 ND 0.01 0.03 8.5 9.5 ND 0.005 0.02 8.5 9.5 ND 0.005 0.02 8.5 9.5 ND 0.005 0.02 8.5 9.5 ND 0.005 0.02
Boiler water O- and P-alkalinity Oxygen scavenger Sulfite, or Hydrazine, or Tannin, or Iso ascorbic acid, or DEHA in FW Chloride Phosphate Silica Dissolved solids Conductivity (25 C) Mg/kg Mg/kg Mg/kg uS/cm 30 70 0.4 x O & P alkalinity 3000 6000 20 50 0.4 x O & P alkalinity 2500 5000 20 40 20 1000 2000 Mg/kg CaCO3 Mg/kg 50 - 300 50 - 150 25 50 10 20 5 - 10 2-5 1-5
30 50 0.1 10 120 160 15 30 0.1 0.25
20 40 0.1 0.5 NR 15 30 0.1 0.25
15 - 20 0.1 0.2 NR 15 30 0.1 0.25
NR 0.05 0.1 NR NR 0.1 0.25
Assess 15 30 5 200 450
NR 0.05 0.1 NR NR 0.1 0.25
Assess 10 20 2 50 150
NR 0.05 0.1 NR NR 0.1 0.25
Assess 3 10 1.5 20 60
NR 0.05 0.1 NR NR 0.1 0.25
Assess 35 0.5 10 35
Boiler water Quality requirements UK Other boilers
CHECK - Is there an update, have seen the draft!
Non-fired Water Tube Parameter Unit 0 - 40 Non-fired Water Tube 41 - 80 Non-fired Water Tube > 80 Once through Coil boiler < 40 Coil boiler > 41 Electrode boiler 101 120
Feedwater
pH (25 C) Total Hardness Oxygen Fe, Cu, and Ni Mg/kg CaCO3 Mg/kg Mg/kg 9.3 9.8 ND 0.02 0.05 9.3 9.8 ND 0.01 0.03 9.3 9.8 ND 0.005 0.02 0.005 0.005 0.02 8.5 9.5 8.5 9.5 1 8.5 9.5 ND 7.5 9.5 1 Nil 0.2 Cond 400 Boiler water
O- and P-alkalinity Total
Oxygen scavenger Sulfite, or Hydrazine, or Tannin, or Iso ascorbic acid, or DEHA in FW Phosphate Silica Dissolved solids Conductivity (25 C)
Mg/kg CaCO3
Mg/kg
25 - 50
10 - 50
25
300
150
300 600
50 100 pH > 9.5 Hardness 0 - 10
15 - 20 0.1 0.5 120 160 15 30 0.1 0.25 Mg/kg Mg/kg Mg/kg uS/cm 20 - 40 20 1000 2000
0.05 0.1 NR 15 30 0.1 0.25 15 30 5 200 400
0.05 0.1 NR 15 30 0.1 0.25 3 10 1-5 50 100 0.02 200 0.2
10 - 20 0.05 0.1 NR NR 0.1 0.25 Assess < 0.4 X O&P alk 8000
NR 0.02 0.05 NR NR 0.02 0.05 3-5 < 0.4 X O&P alk 5000
400 2000
Boiler water Quality requirements UK Fired Water tube
Feedwater
Parameter Unit 0 - 20 21 40 41 - 60 61 - 80 81 - 100 101 - 120 121
Pressure Appearance
bar Clear, no foam
300
pH (25 C)
Total Hardness Oxygen Fe, Cu, and Ni Mg/kg CaCO3 Mg/kg Mg/kg
8.5 9.5
2 0.02 0.05
8.5 9.5
1 0.02 0.05
8.5 9.5
ND 0.01 0.03
8.5 9.5
ND 0.005 0.02
8.5 9.5
ND 0.005 0.02
8.5 9.5
ND 0.005 0.02
8.5 9.5
ND 0.005 0.02
Potrebbero piacerti anche
- Boiler Water BasicsDocumento63 pagineBoiler Water BasicsTin Aung Kyi100% (4)
- Boiler Water AnalysisDocumento44 pagineBoiler Water AnalysisDileep MishraNessuna valutazione finora
- Thermal Power Plant Water ChemistryDocumento33 pagineThermal Power Plant Water ChemistryNAITIK100% (4)
- Control Water Quality BoilersDocumento48 pagineControl Water Quality BoilerssamettekNessuna valutazione finora
- Boiler Tube Failures: Pradip PatelDocumento69 pagineBoiler Tube Failures: Pradip PatelVishnu ParvandaNessuna valutazione finora
- Boiler Water Chemistry: Korba Simulator 339Documento12 pagineBoiler Water Chemistry: Korba Simulator 339Rakesh Kiran100% (1)
- Boiler Water TreatmentDocumento37 pagineBoiler Water Treatmentrajendick59% (17)
- Chemical Cleaning NPTIDocumento39 pagineChemical Cleaning NPTISaravana Kumar100% (1)
- Phosphate Hideout PDFDocumento4 paginePhosphate Hideout PDFUsama JawaidNessuna valutazione finora
- Boiler Water TreatmentDocumento30 pagineBoiler Water TreatmentDeo Anugrah Utomo100% (6)
- Boiler Water ChemistryDocumento60 pagineBoiler Water Chemistryنيرمين احمدNessuna valutazione finora
- Boiler Phosphate TreatmentDocumento4 pagineBoiler Phosphate TreatmentSilvina C. Gómez Correa0% (1)
- Phosphate Hideout: Questions and AnswersDocumento4 paginePhosphate Hideout: Questions and AnswersSivakumar Rajagopal100% (3)
- All You Need To Know About Boiler Feed Water Treatment E-Book-1 PDFDocumento37 pagineAll You Need To Know About Boiler Feed Water Treatment E-Book-1 PDFHazem Alsharif100% (1)
- Water ChemistryDocumento13 pagineWater ChemistrynivasssvNessuna valutazione finora
- Boiler Water TrainingDocumento113 pagineBoiler Water Trainingaramkhosrove92% (13)
- Boiler Water Chemistry Analysis (PB QBook)Documento8 pagineBoiler Water Chemistry Analysis (PB QBook)Ehsan Pappu100% (1)
- Boiler Tube Failures Causes and ControlsDocumento20 pagineBoiler Tube Failures Causes and ControlsParmeshwar Nath Tripathi100% (1)
- Boiler Water Treatment - WhyDocumento53 pagineBoiler Water Treatment - Whymechetron100% (8)
- Best Practices in Boiler Water TreatmentDocumento5 pagineBest Practices in Boiler Water Treatmenthiyt4amNessuna valutazione finora
- Boiler-Water ChemistryDocumento94 pagineBoiler-Water ChemistryPRAG100% (2)
- BFW Chemistry ControlDocumento61 pagineBFW Chemistry ControlSteve WanNessuna valutazione finora
- Co-Ordinated and Congruent Phosphate Treatment of Boiler WaterDocumento3 pagineCo-Ordinated and Congruent Phosphate Treatment of Boiler WaterRavi Shankar0% (1)
- Boiler Preservation NalcoDocumento4 pagineBoiler Preservation NalcoSushil Kumar71% (7)
- Boiler Corrosion & Feedwater TreatmentDocumento24 pagineBoiler Corrosion & Feedwater Treatmentmohan4175Nessuna valutazione finora
- Carbohydrazide vs Hydrazine Oxygen Scavenger StudyDocumento43 pagineCarbohydrazide vs Hydrazine Oxygen Scavenger StudyVinod SindhavNessuna valutazione finora
- Feed Water ManagementDocumento31 pagineFeed Water ManagementAnmol AgarwalNessuna valutazione finora
- Role of Chemistry in Power PlantDocumento42 pagineRole of Chemistry in Power Plantsevero97100% (1)
- Boiler Tube LeakagesDocumento33 pagineBoiler Tube LeakagesVIBHAV0% (1)
- Basic Guide To Boiler Water TreatmentDocumento8 pagineBasic Guide To Boiler Water TreatmentjewettwaterNessuna valutazione finora
- Power Plant ChemistryDocumento136 paginePower Plant ChemistryGajender Singh RajputNessuna valutazione finora
- Nalco Cooling Tower PresentationDocumento115 pagineNalco Cooling Tower PresentationAnonymous ffje1rpa80% (10)
- Boiler Water TreatmentDocumento50 pagineBoiler Water Treatmentak_thimiriNessuna valutazione finora
- Oxygenated Boiler Water Chemistry-15.06.2016Documento25 pagineOxygenated Boiler Water Chemistry-15.06.2016tek_surinderNessuna valutazione finora
- Boiler Chemical Cleaning Guidelines - Case Studies000000000001012756 PDFDocumento92 pagineBoiler Chemical Cleaning Guidelines - Case Studies000000000001012756 PDFraosudh100% (1)
- Stator Water Chemistry R0Documento6 pagineStator Water Chemistry R0vel muruganNessuna valutazione finora
- Oxygenated Feed Water TreatmentDocumento5 pagineOxygenated Feed Water Treatmentsenthil031277100% (1)
- HRSG Water ChemistryDocumento83 pagineHRSG Water ChemistrynivasssvNessuna valutazione finora
- Cooling Water BasicsDocumento163 pagineCooling Water BasicsTin Aung Kyi94% (16)
- Boiler Coal Combustion: Operation PerspectiveDocumento42 pagineBoiler Coal Combustion: Operation PerspectiveDevendra Singh DhakreyNessuna valutazione finora
- Boiler Water Treatment: Thermax Limited Chemical DivisionDocumento53 pagineBoiler Water Treatment: Thermax Limited Chemical Divisionkcp1986100% (2)
- Condensate Water Polishing SystemDocumento257 pagineCondensate Water Polishing SystemHamza RaoNessuna valutazione finora
- Chemical Cleaning of Boilers With EDTADocumento33 pagineChemical Cleaning of Boilers With EDTASACHIN PETKARNessuna valutazione finora
- Alkali BoiloutDocumento31 pagineAlkali BoiloutDuggineni Ramakrishna0% (1)
- Boiler Pressure Parts & Tube FailureDocumento76 pagineBoiler Pressure Parts & Tube Failurekvsagar100% (4)
- Cycle Water Chemistry by Sudhanshu DashDocumento31 pagineCycle Water Chemistry by Sudhanshu DashSudhanshu Sekhar Dash100% (1)
- Silica Carryover in BoilerDocumento3 pagineSilica Carryover in BoilermnasiroleslamiNessuna valutazione finora
- Flow Accelerated Corrosion and Its Prevention Through AVT (O) and OTDocumento10 pagineFlow Accelerated Corrosion and Its Prevention Through AVT (O) and OTthanhhuyenNessuna valutazione finora
- Boiler Internal & External TreatmentDocumento62 pagineBoiler Internal & External Treatmentmdnor100% (1)
- Wet Lay Up in BoilerDocumento5 pagineWet Lay Up in BoilersprotkarNessuna valutazione finora
- Boiler Water Treatment FundamentalsDocumento8 pagineBoiler Water Treatment FundamentalssauravsinhaaNessuna valutazione finora
- Boiler Feed Water and Boiler Water - EWT Water TechnologyDocumento2 pagineBoiler Feed Water and Boiler Water - EWT Water TechnologyEdhi SutantoNessuna valutazione finora
- EPRI Phosphat & Caustic Treatment GuidelinesDocumento404 pagineEPRI Phosphat & Caustic Treatment Guidelinesrizal100% (7)
- WTPDocumento33 pagineWTPSam100% (2)
- Demineralization by Ion Exchange: In Water Treatment and Chemical Processing of Other LiquidsDa EverandDemineralization by Ion Exchange: In Water Treatment and Chemical Processing of Other LiquidsValutazione: 4 su 5 stelle4/5 (2)
- Boiler Water ChemistryDocumento48 pagineBoiler Water ChemistryR.S.Sankarasubramanian100% (1)
- Boiler Water TreatmentDocumento80 pagineBoiler Water TreatmentRAJINDER HANSNessuna valutazione finora
- Boiler Tube FailuresDocumento83 pagineBoiler Tube Failuressen_subhasis_5867% (3)
- DSS Boiler Water Treatment Technologyh1Documento64 pagineDSS Boiler Water Treatment Technologyh1عزت عبد المنعمNessuna valutazione finora
- LG Optimus L9Documento6 pagineLG Optimus L9Bhargav ChaudhariNessuna valutazione finora
- Boiler OH Intervals-EPRIDocumento93 pagineBoiler OH Intervals-EPRIBhargav Chaudhari100% (1)
- Solid Waste-1Documento19 pagineSolid Waste-1Bhargav ChaudhariNessuna valutazione finora
- 190 Sample ChapterDocumento55 pagine190 Sample ChapterJayit SahaNessuna valutazione finora
- NTPC's Greener FutureDocumento89 pagineNTPC's Greener FutureKanhaiya Kumar SinghNessuna valutazione finora
- Handout AADocumento79 pagineHandout AABhargav ChaudhariNessuna valutazione finora
- Life Assesment of BoilersDocumento53 pagineLife Assesment of BoilersBhargav ChaudhariNessuna valutazione finora
- Gsecl Tender For CBDocumento1 paginaGsecl Tender For CBBhargav ChaudhariNessuna valutazione finora
- 2a - Life Assesment of BoilersDocumento9 pagine2a - Life Assesment of BoilersBhargav ChaudhariNessuna valutazione finora
- EPPWT HandoutDocumento47 pagineEPPWT HandoutBhargav ChaudhariNessuna valutazione finora
- 05 - C&iDocumento120 pagine05 - C&iBhargav ChaudhariNessuna valutazione finora
- King Steam Generator Upgrades&MaintDocumento26 pagineKing Steam Generator Upgrades&MaintBhargav ChaudhariNessuna valutazione finora
- Handout AADocumento79 pagineHandout AABhargav ChaudhariNessuna valutazione finora
- 01 - Boiler Performance & DesignDocumento79 pagine01 - Boiler Performance & DesignBhargav ChaudhariNessuna valutazione finora
- Control TechnologiesDocumento58 pagineControl TechnologiesBabu AravindNessuna valutazione finora
- Boiler Reliabiliti Optimization 2001Documento178 pagineBoiler Reliabiliti Optimization 2001Bhargav ChaudhariNessuna valutazione finora
- ESP DesignReviewDocumento22 pagineESP DesignReviewssirupaNessuna valutazione finora
- Boiler Reliabiliti Optimization 2001Documento178 pagineBoiler Reliabiliti Optimization 2001Bhargav ChaudhariNessuna valutazione finora
- Presentation ON Efficiency Imprvement in Coal Fired Thermal Power StationDocumento29 paginePresentation ON Efficiency Imprvement in Coal Fired Thermal Power Stationprithvirajd20Nessuna valutazione finora
- Electrostatic Precipitator Modeling and SimulationDocumento37 pagineElectrostatic Precipitator Modeling and SimulationSelvanathan Murugaiyan100% (1)
- M14Cent Pump VibDocumento49 pagineM14Cent Pump VibBhargav Chaudhari100% (1)
- Electrostatic Precipitator (ESP)Documento20 pagineElectrostatic Precipitator (ESP)avmurugan87Nessuna valutazione finora
- 3.igen Ea BoilerDocumento64 pagine3.igen Ea BoilerBhargav ChaudhariNessuna valutazione finora
- Thermal Plant LayoutDocumento71 pagineThermal Plant LayoutBhargav Chaudhari100% (1)
- Air BechtelDocumento34 pagineAir BechtelBhargav ChaudhariNessuna valutazione finora
- SOFA BurnersDocumento20 pagineSOFA BurnersBhargav ChaudhariNessuna valutazione finora
- Equipment BoilerDocumento135 pagineEquipment Boilertvpham123Nessuna valutazione finora
- Boiler Safety ValveDocumento12 pagineBoiler Safety ValveJaroslav Kuruc100% (2)
- Boiler Remaining Life AssessDocumento33 pagineBoiler Remaining Life AssessSSudhakar100% (1)
- F3 Chapter 4 Reactivity of MetalsDocumento11 pagineF3 Chapter 4 Reactivity of MetalsJue Hazea GoldshopNessuna valutazione finora
- AP Chemistry Solubility Rules Equations SheetDocumento8 pagineAP Chemistry Solubility Rules Equations SheetssNessuna valutazione finora
- Experiment 2 Analysis of An Unknown Vinegar Sample ObjectivesDocumento5 pagineExperiment 2 Analysis of An Unknown Vinegar Sample ObjectivesVirginia DukeNessuna valutazione finora
- Namma Kalvi 12th Chemistry Unit 1 Study Material em 215343Documento28 pagineNamma Kalvi 12th Chemistry Unit 1 Study Material em 215343HarishNessuna valutazione finora
- Membrane FinalDocumento7 pagineMembrane Finalpd shrmaNessuna valutazione finora
- Preparation of Buffer Solutions and Colorimetric Determination of pHDocumento8 paginePreparation of Buffer Solutions and Colorimetric Determination of pHscsa31619Nessuna valutazione finora
- Form 4 - Salts (+experiment)Documento4 pagineForm 4 - Salts (+experiment)kanryu_zonasNessuna valutazione finora
- 3322 Microgard Training Slides AbridgedDocumento47 pagine3322 Microgard Training Slides AbridgedVan SotNessuna valutazione finora
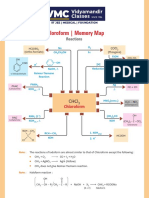
- Chloroform - Memory Map: ReactionsDocumento1 paginaChloroform - Memory Map: ReactionsAryan GuptaNessuna valutazione finora
- Extracting Copper From Its OresDocumento2 pagineExtracting Copper From Its Oresfarhathul afraaNessuna valutazione finora
- General Chemistry Laboratory Report: Experiment 2 PH and BuffersDocumento11 pagineGeneral Chemistry Laboratory Report: Experiment 2 PH and BuffersBùi Nhật MaiNessuna valutazione finora
- G9 Ste Conchem Q1 WK1Documento20 pagineG9 Ste Conchem Q1 WK1Breeza Marie VeralloNessuna valutazione finora
- K3M 2008 - Form 6Documento15 pagineK3M 2008 - Form 6SeanNessuna valutazione finora
- Plastic Film in Food IndustryDocumento1 paginaPlastic Film in Food IndustryNurul SyafiqahNessuna valutazione finora
- Lubricant Base OilsDocumento3 pagineLubricant Base OilssinhleprovietNessuna valutazione finora
- 5070 s11 QP 11Documento16 pagine5070 s11 QP 11chuasioklengNessuna valutazione finora
- Chemistry: Pearson Edexcel GCEDocumento28 pagineChemistry: Pearson Edexcel GCEJerome MosadaNessuna valutazione finora
- Cambridge IGCSE: CHEMISTRY 0620/42Documento12 pagineCambridge IGCSE: CHEMISTRY 0620/42Kohinoor BegumNessuna valutazione finora
- Shrikant FeSO4 ReportDocumento20 pagineShrikant FeSO4 ReportPrashant KiraveNessuna valutazione finora
- Chemical Formulae and EquationsDocumento3 pagineChemical Formulae and EquationsFatema KhatunNessuna valutazione finora
- ChemCatChem - 2020 - Seifert - Ethanol To Aromatics On Modified H ZSM 5 Part I Interdependent Dealumination ActionsDocumento11 pagineChemCatChem - 2020 - Seifert - Ethanol To Aromatics On Modified H ZSM 5 Part I Interdependent Dealumination ActionsBoby SixkillersNessuna valutazione finora
- Answers To End-Of-Chapter Questions: Chapter 9 Industrial Inorganic ChemistryDocumento2 pagineAnswers To End-Of-Chapter Questions: Chapter 9 Industrial Inorganic ChemistryMP3 MusicNessuna valutazione finora
- 08-Synthesize Ethyl AcetateDocumento11 pagine08-Synthesize Ethyl AcetateHamdan CheveuxleonNessuna valutazione finora
- Table of Standard Amino Acid Abbreviations and Side Chain PropertiesDocumento9 pagineTable of Standard Amino Acid Abbreviations and Side Chain PropertieschaitanyasakhNessuna valutazione finora
- Spectroscopic Determination of Amino AcidsDocumento7 pagineSpectroscopic Determination of Amino AcidsDeysi Suarez GomezNessuna valutazione finora
- NH Chemistry All 2019-DQP-SpellcheckonDocumento65 pagineNH Chemistry All 2019-DQP-SpellcheckonJohn TechNessuna valutazione finora
- Chloramine (Mono) : Indophenol Method Method 10171 0.04 To 4.50 MG/L CL (LR) Powder PillowsDocumento8 pagineChloramine (Mono) : Indophenol Method Method 10171 0.04 To 4.50 MG/L CL (LR) Powder PillowsDody PurmadaniNessuna valutazione finora
- Practice Exam I - Organic ChemsitryDocumento14 paginePractice Exam I - Organic ChemsitryDave KowalczykNessuna valutazione finora
- Types of Chemical Reactions LabDocumento15 pagineTypes of Chemical Reactions LabChitlet FrancheNessuna valutazione finora
- Isolation and Extraction of AlkaloidsDocumento24 pagineIsolation and Extraction of AlkaloidsPrateek Kanade89% (19)